The Polar Covalent Bonds of Water: Structure, Hydrogen Bonding, and Unique Properties
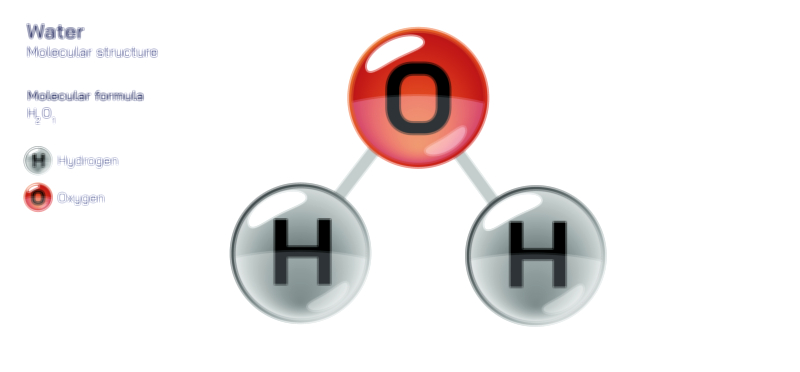
Water (H₂O) is a remarkable molecule essential for life, largely due to its polar covalent bonds. In a water molecule, the oxygen atom shares electrons with two hydrogen atoms, forming covalent bonds. However, because oxygen is more electronegative than hydrogen, it attracts the shared electrons more strongly, creating a partial negative charge on oxygen and partial positive charges on the hydrogen atoms. This unequal electron distribution gives water its molecular polarity, making it a polar molecule.
The polarity of water has significant implications. Polar molecules interact with other polar substances, allowing water to act as an excellent solvent for ionic and polar compounds. This property is critical for biological systems, where water facilitates chemical reactions, nutrient transport, and cellular processes. The polar covalent bonds in water also enable hydrogen bonding, a type of weak electrostatic attraction between the slightly positive hydrogen atom of one water molecule and the slightly negative oxygen atom of another. These hydrogen bonds are responsible for water’s high cohesion, surface tension, specific heat, and boiling point, which are vital for maintaining stable environments in both ecosystems and living organisms.
The molecular geometry of water contributes to its polar nature. Water has a bent or V-shaped structure, with a bond angle of approximately 104.5°. This geometry prevents the cancellation of dipole moments, ensuring a permanent polarity that supports hydrogen bonding networks. These interactions lead to unique properties such as ice floating on liquid water, allowing aquatic life to survive in freezing temperatures, and capillary action, which enables water to move through plant vessels against gravity.
Biological significance of polar covalent bonds in water is profound. Hydrogen bonds allow water to stabilize DNA and proteins, maintain cell turgor pressure, and regulate body temperature. They also play a role in biochemical reactions, facilitating molecular interactions in enzymatic activity and metabolism. Without polar covalent bonds, water would lack these vital characteristics, making life as we know it impossible.
In conclusion, the polar covalent bonds of water are fundamental to its molecular polarity, hydrogen bonding, and unique physical and chemical properties. These bonds underpin water’s role as a universal solvent, a temperature stabilizer, and a facilitator of biochemical reactions. Understanding these bonds provides deep insight into molecular chemistry, biology, and the essential nature of water for sustaining life on Earth.