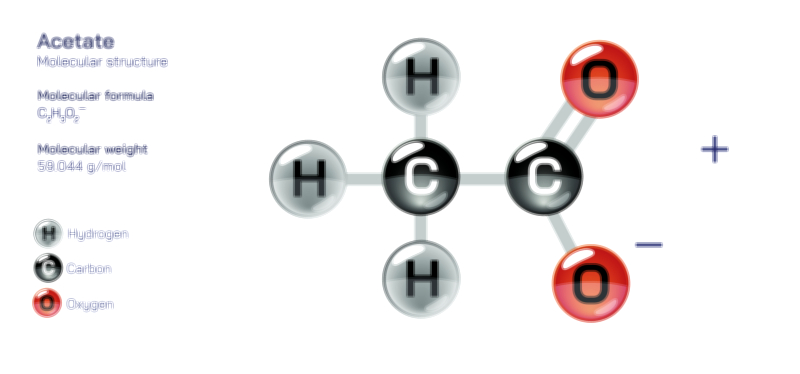
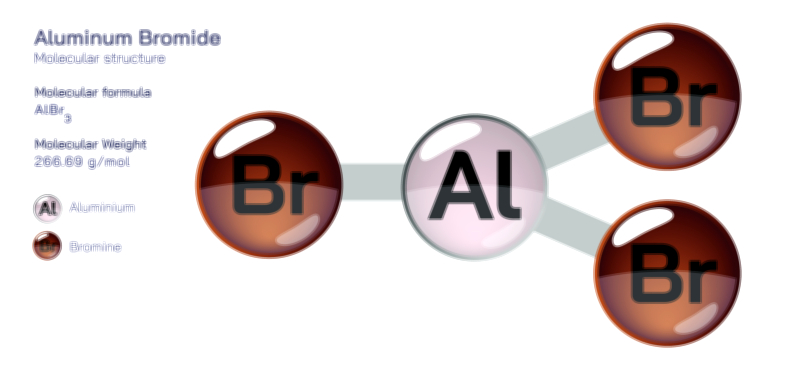
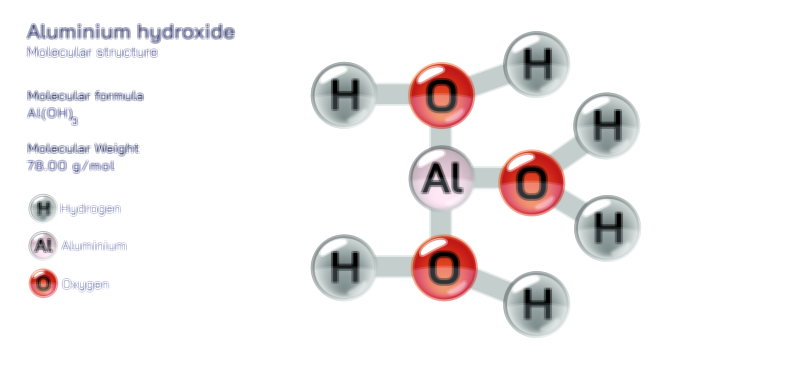
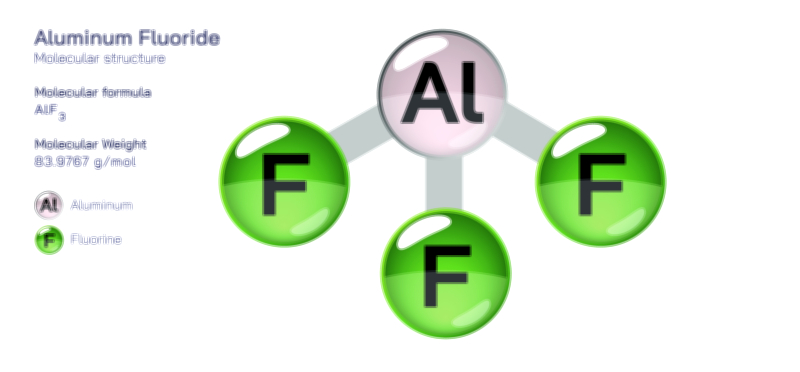
Acetylene Molecule — Chemical Structure and Hydrocarbon Illustration
Fundamental Structure and Identity of the Acetylene Molecule
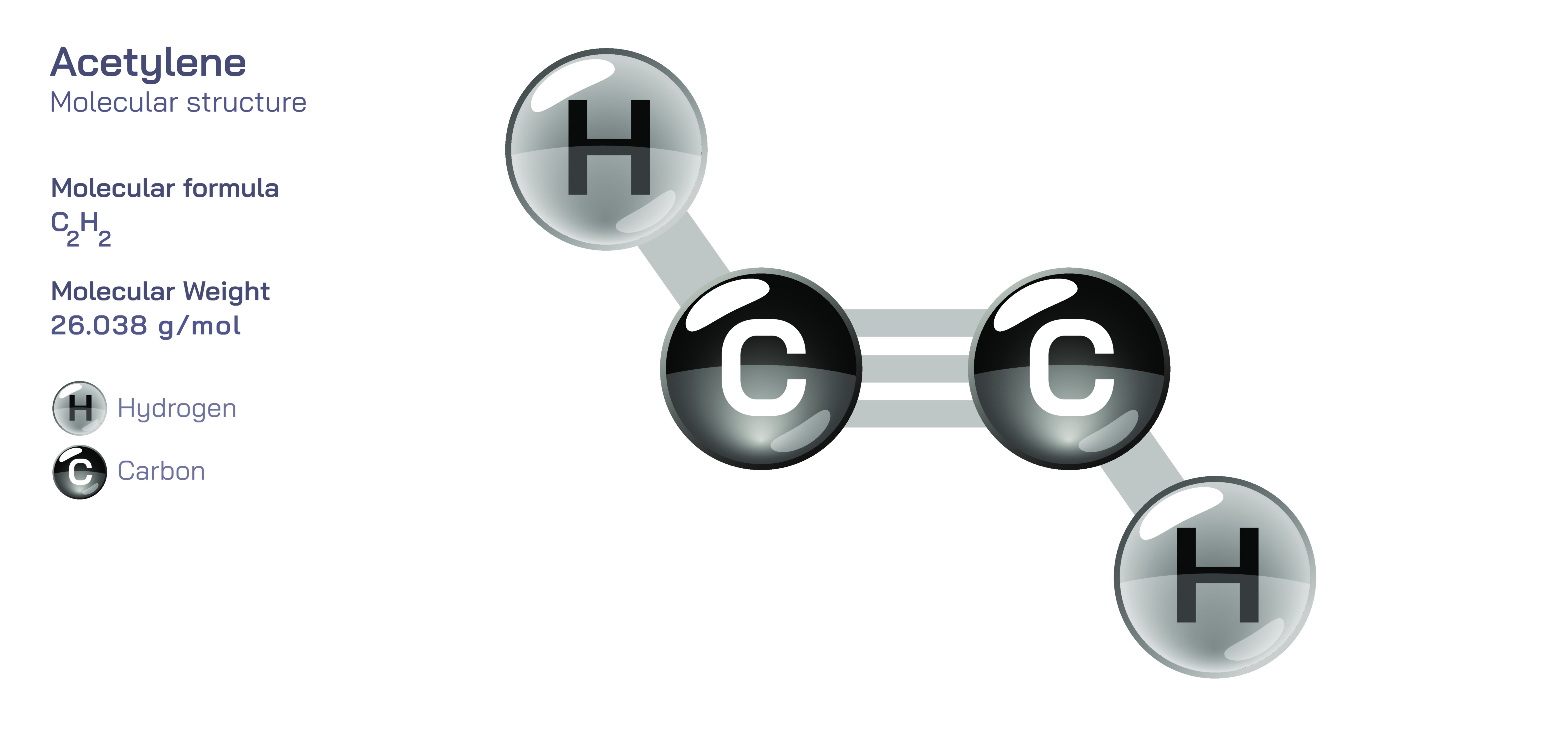
Acetylene, also known by its systematic name ethyne, is one of the simplest hydrocarbons yet among the most chemically intriguing. Represented by the formula C₂H₂, it belongs to the family of alkynes, a class defined by the presence of at least one carbon–carbon triple bond. This triple bond is the central feature of the acetylene molecule, creating a structure that is both highly reactive and remarkably linear. The molecule consists of two carbon atoms linked together through one sigma bond and two pi bonds, and each carbon atom is bonded to a single hydrogen. This combination results in a rigid, straight-line geometry that distinguishes acetylene from alkanes and alkenes, which offer more structural flexibility. Understanding the basic composition of acetylene provides insight into a wide range of chemical behaviors involving acidity, polymerization, addition reactions, combustion properties, and industrial synthesis. Despite its simplicity, acetylene plays a foundational role in organic chemistry, serving as both a building block for more complex molecules and a practical tool across numerous industrial applications.
Electronic Characteristics and the Nature of the Triple Bond
The defining element of acetylene’s structure is its carbon–carbon triple bond, which forms through sp hybridization of the carbon atoms. Each carbon uses one sp orbital to form a sigma bond with the other carbon, while the remaining sp orbital bonds with hydrogen. The two unhybridized p orbitals on each carbon overlap sideways to produce two pi bonds, completing the triple-bond framework. This arrangement compresses the molecule into a perfectly linear structure with a bond angle of 180 degrees. The triple bond carries significant electron density along the internuclear axis, making it shorter and stronger than double or single bonds. This strength contributes to acetylene’s characteristic reactivity: the molecule can undergo addition reactions where the triple bond is progressively reduced from three shared electron interactions to two and then one, allowing new atoms or groups to attach to the carbon chain. The linearity also produces a unique polarity distribution, leaving the internal carbon atoms relatively electron-rich while the terminal hydrogen atoms exhibit slight acidity. This acidity is unusual among hydrocarbons, making acetylene capable of forming metal acetylides when treated with strong bases or certain metal ions.
Physical Properties and Their Chemical Implications
Acetylene exists under normal conditions as a colorless gas with a faint, somewhat ether-like odor, although impurities often give it a sharper, garlic-like smell. It is highly flammable and capable of undergoing explosive decomposition under elevated pressure or temperature, which is why it is commercially stored dissolved in acetone or dimethylformamide within porous cylinders. Its combustion produces an extremely high-temperature flame, reaching over 3000°C, making it ideal for metal cutting and welding applications. The gas has slightly limited solubility in water but dissolves well in organic solvents due to its nonpolar hydrocarbon character combined with modest electron density around the triple bond. The linear compactness of acetylene enhances its reactivity toward electrophiles and transition metals while simultaneously reducing steric hindrance compared to bulkier organic molecules. These physical traits demonstrate why acetylene is both a reactive substrate in organic synthesis and a practical industrial material requiring careful handling.
Chemical Reactivity and Transformation Pathways
The reactivity of acetylene stems primarily from its triple bond, which serves as a site for nucleophilic, electrophilic, and catalytic addition reactions. Hydrogenation reduces acetylene sequentially to ethylene and then ethane. Halogens can add across the triple bond to produce dihalogenated or tetrahalogenated derivatives, while hydrogen halides form vinyl halides or geminal dihalides depending on reaction conditions. Acetylene’s acidic hydrogen atoms allow it to react with strong bases such as sodium amide, forming acetylide ions, which are highly useful intermediates in carbon–carbon bond formation. Metal acetylides also function as ligands in coordination complexes, making acetylene significant in organometallic chemistry. In polymer chemistry, acetylene can undergo chain-growth polymerization, forming polyacetylene, a conductive polymer that historically played a role in the discovery of organic conductors. These pathways reveal the molecule’s flexibility and its importance in constructing complex chemical structures from the simplest hydrocarbon building blocks.
Industrial, Environmental, and Practical Importance
Acetylene plays a prominent role in industrial processes, particularly in welding and cutting due to its exceptionally hot flame when burned with oxygen. Historically, it served as an illuminating gas, powering lamps before the widespread adoption of electricity. In modern chemical manufacturing, it acts as a precursor to a wide range of materials such as vinyl chloride, acrylonitrile, synthetic rubber components, solvents, and pharmaceutical intermediates. Its ability to form acetylide compounds also makes it valuable for synthesizing fine chemicals and specialty materials. In environmental chemistry, acetylene appears as a byproduct in combustion processes and certain natural reactions occurring in the atmosphere or soil microbial systems. Although it is not a major pollutant, its reactive nature allows it to influence local chemical environments where it accumulates. In biochemical contexts, some microorganisms can metabolize acetylene, converting it through enzymatic pathways that offer insights into nitrogen fixation and microbial evolution.
Illustrating the Structure and Chemical Characteristics of Acetylene
Scientific illustrations of acetylene typically highlight the linear geometry and the carbon–carbon triple bond, emphasizing the combination of sigma and pi bonding that defines its structure. Diagrams often show the overlapping p orbitals forming the pi bonds and the alignment of atoms into a straight line, reflecting the molecule’s simplicity and rigidity. These illustrations may also depict how electrophiles or catalysts approach the triple bond during addition reactions, providing a visual explanation of why acetylene is so reactive and versatile. Visual representations further clarify how the acidic hydrogen atoms can be removed to form acetylide ions, helping illustrate transformation pathways used in synthesis. By presenting the molecular features in clear structural formats, such illustrations assist in understanding how a seemingly simple molecule carries such a broad and impactful set of chemical behaviors across scientific, industrial, and environmental domains.