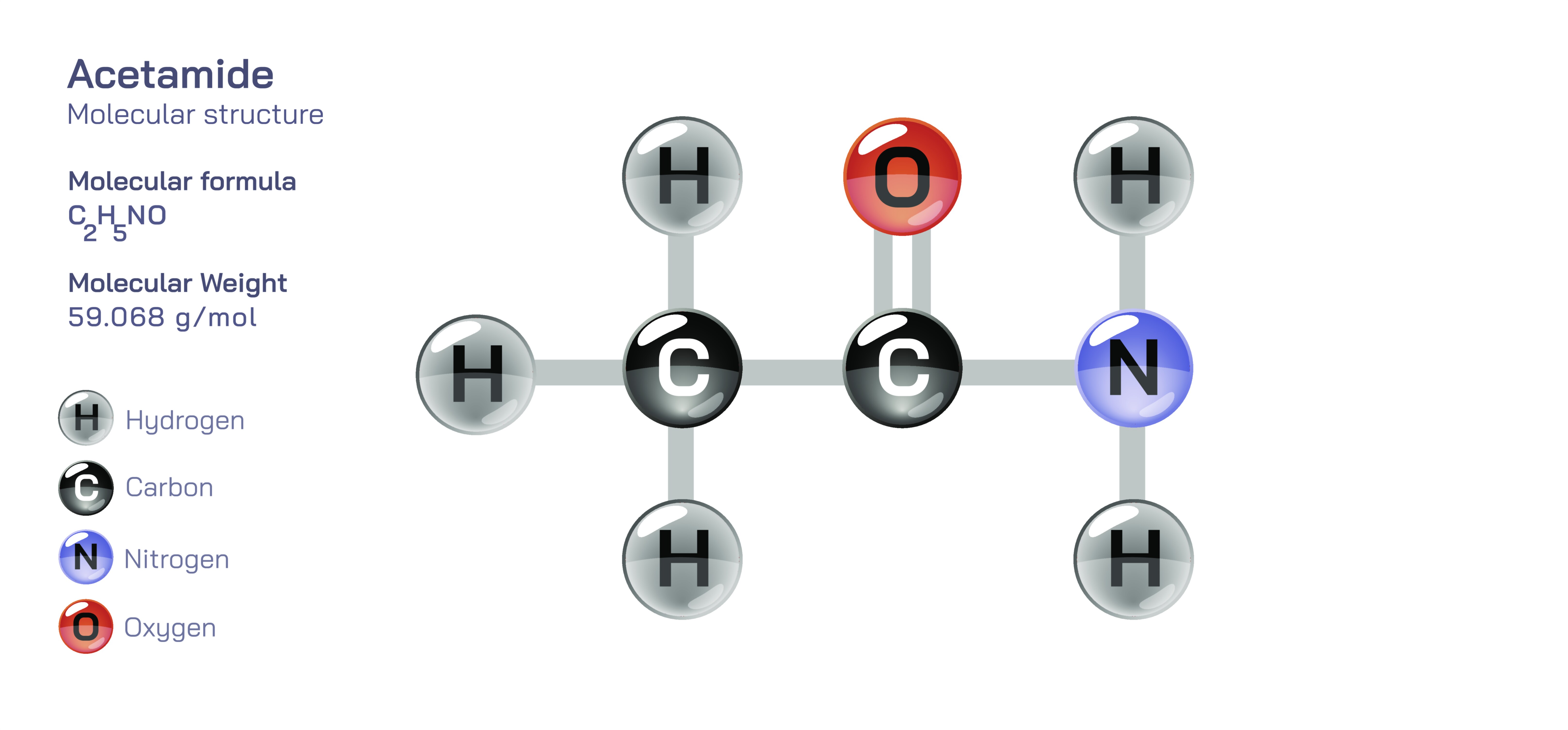
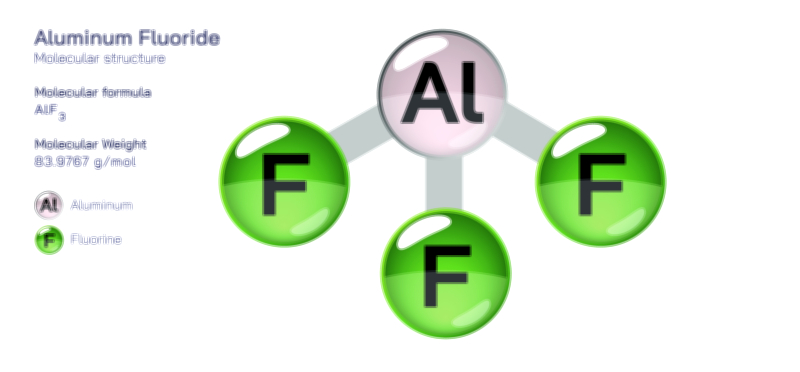
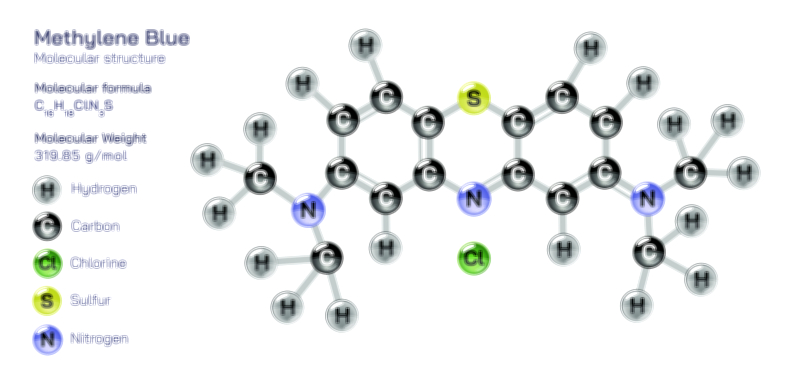
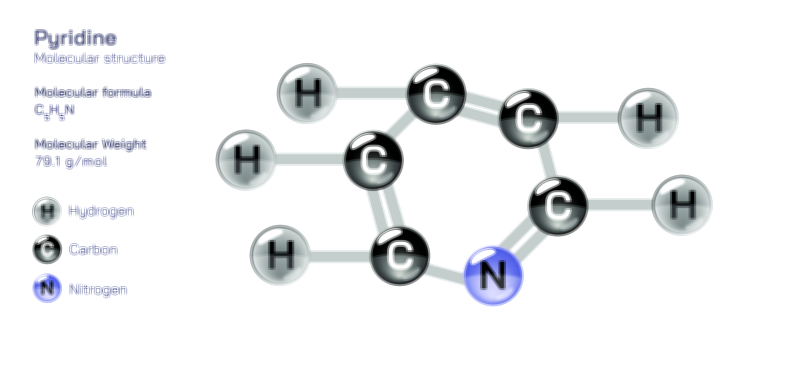
Acetamide Molecule — Chemical Structure and Organic Compound Illustration
Fundamental Structure and Molecular Identity of Acetamide
Acetamide, with the molecular formula CH₃CONH₂, stands as the simplest amide derived from acetic acid. Its structure embodies the essential features of the amide functional group, connecting an acetyl group to an amino group through a carbon–nitrogen bond that defines its chemical behavior. This combination forms a small yet deeply influential molecule in the study of organic chemistry, biochemistry, and industrial applications. The core of the acetamide molecule rests in the interplay between the carbonyl carbon and the attached nitrogen atom, creating a resonance-stabilized structure that blends characteristics of both amines and carboxylic acids. This resonance imparts a partial double-bond nature to the carbon–nitrogen connection, resulting in restricted rotation and significant stability. Understanding this molecular arrangement is crucial because acetamide serves as a fundamental reference compound for exploring peptide bonds, protein structures, and amide reactivity across organic synthesis. It occupies a unique position in scientific research as a model compound that bridges simple organic molecules with complex biological macromolecules.
Electronic Characteristics and Structural Behavior
The electronic structure of acetamide is dominated by resonance stabilization between the carbonyl oxygen, the carbon atom of the carbonyl group, and the nitrogen atom of the amide linkage. This resonance distributes electron density across the C–N and C=O bonds, making the nitrogen less basic than in typical amines and reducing the reactivity of the carbonyl carbon compared to other carbonyl-containing compounds such as aldehydes or ketones. The partial double-bond nature of the C–N bond restricts its free rotation, resulting in a planar configuration around the amide functional group. This planarity is a key structural trait, forming the basis for the secondary structures observed in proteins and peptides, where thousands of amide bonds align into stable conformations. The hydrogen atoms attached to the nitrogen also play a role in stabilizing intermolecular interactions. Acetamide can engage in hydrogen bonding with itself and other molecules, which significantly influences its melting point, solubility, and ability to form crystalline structures. These hydrogen bonds also allow acetamide to interact effectively with biological molecules, making it relevant not only as a structural model but also as a compound with physiological and biochemical significance.
Physical Properties and Their Chemical Implications
The physical properties of acetamide reflect its intermediate position between amines, carboxylic acids, and other carbonyl-containing molecules. In its pure form, acetamide is a colorless, crystalline solid with a faint odor, exhibiting a relatively high melting point due to the extensive hydrogen bonding that stabilizes its structure in the solid phase. It is highly soluble in water, a trait attributed to the polarity of the amide group and its ability to form hydrogen bonds with water molecules. This solubility extends to alcohols and other polar solvents, allowing acetamide to serve as a versatile reagent and experimental medium in laboratory settings. Unlike many simple organic compounds, acetamide is thermally stable, resisting spontaneous decomposition under normal conditions. However, when heated to higher temperatures, it can undergo dehydration to form acetonitrile, a transformation important in industrial synthesis. The physical properties of acetamide also make it relevant in discussions of environmental behavior, as its water solubility and stability influence how it distributes in natural and industrial ecosystems.
Chemical Reactivity and Transformation Pathways
Although acetamide is a relatively stable compound compared to aldehydes, esters, or anhydrides, it retains the capacity to participate in several important chemical reactions. Hydrolysis is the most notable, where acetamide can break down into acetic acid and ammonia under acidic or basic conditions. This reaction highlights the reversibility and dynamic nature of the amide bond, which, although strong, can be cleaved through catalytic processes. Reduction reactions convert acetamide into ethylamine, offering a pathway to amine synthesis that is useful both academically and industrially. When subjected to dehydrating agents or elevated temperatures, acetamide transforms into acetonitrile, an important organic solvent and synthesis intermediate. These reaction pathways underscore acetamide’s role as a chemical bridge between functional groups, illustrating how small molecules can serve as templates for generating larger or more specialized organic structures. Additionally, the resonance stabilization of acetamide affects its behavior toward nucleophiles and electrophiles, often making it less reactive than structurally similar compounds, thereby offering controlled reactivity in synthetic schemes.
Biological and Industrial Relevance
Acetamide’s significance extends into biological and industrial fields where its molecular characteristics shape its utility. In biological studies, it is frequently used as a model compound for understanding peptide linkages in proteins, as the amide bond in acetamide closely mimics the fundamental structure of the peptide bond connecting amino acids. This connection to biological macromolecules makes acetamide a valuable reference in spectroscopy, protein chemistry, and structural biology. Industrially, acetamide is applied in the production of solvents, plasticizers, penetrating oils, and stabilizers due to its chemical stability and solubility properties. It is also used in pharmaceuticals, where its structural resemblance to peptide fragments allows it to serve as an intermediate in drug synthesis. Furthermore, its presence in environmental chemistry emerges from its use in various manufacturing processes, and understanding its behavior in water and soil systems is important for assessing potential impacts.
Illustrating the Structure and Behavior of Acetamide
Scientific illustrations of acetamide often emphasize the resonance stabilization and planar structure of the amide functional group. These visual representations typically highlight the carbonyl group, the attached nitrogen, and the distribution of electron density that explains the molecule’s stability and reactivity. Illustrations may show how the molecule forms hydrogen bonds, how the resonance contributes to bond length uniformity, and how the amide group aligns in biological systems. Such depictions help in understanding how and why acetamide behaves as it does across various chemical environments. By visualizing these structural principles, one gains a deeper appreciation of how a seemingly simple molecule embodies the essential characteristics of amide chemistry and influences both organic synthesis and biological structure.