Acetate Ion — Chemical Structure and Molecular Composition Explained
The acetate ion is one of the most important and widely encountered organic ions in chemistry and biochemistry, serving as a fundamental building block in metabolic pathways, industrial manufacturing, and analytical chemistry. Represented chemically as CH₃COO⁻ or C₂H₃O₂⁻, the acetate ion originates from the deprotonation of acetic acid (CH₃COOH), meaning that when acetic acid loses a hydrogen ion (H⁺), the remaining species becomes an acetate ion. The transformation into acetate changes the behavior of the molecule dramatically: the neutral acid becomes a negatively charged conjugate base capable of forming ionic bonds, coordinating with metal ions, and participating in essential biochemical reactions. The acetate ion lies at the intersection of organic chemistry and biological processes, where its structure allows it to combine stability, reactivity, and solubility in a unique balance. This capacity for interaction forms the foundation of its importance in everything from industrial plastics production to the Krebs cycle that fuels living cells.
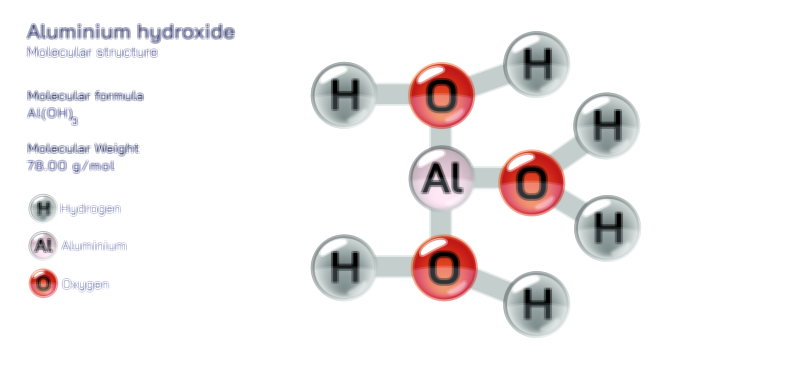
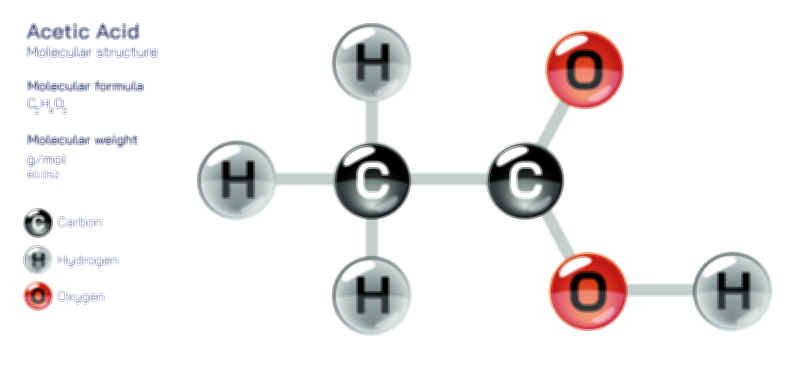
To understand acetate, one must first visualize its molecular structure, because its reactivity and chemical character arise from the arrangement of atoms within the molecule. The acetate ion consists of two carbon atoms, three hydrogen atoms, and two oxygen atoms arranged in the configuration CH₃–COO⁻. One carbon belongs to a methyl group (CH₃)—a saturated, non-polar fragment of the molecule—while the second carbon forms part of a carboxylate group (COO⁻). This carboxylate region is the functional core of the ion, containing a carbon atom double-bonded to one oxygen and single-bonded to another oxygen that carries the negative charge. Importantly, the two carbon–oxygen bonds in the carboxylate group are not simple single and double bonds in a fixed pattern; instead, the electrons are delocalized through resonance, meaning the negative charge is shared equally between the two oxygen atoms. This resonance gives acetate its remarkable stability: neither oxygen retains the full burden of the charge, and the electron distribution makes the ion less reactive than it would be with the charge concentrated in only one location.
This resonance stabilization explains why acetic acid is a weak acid—it dissociates only partially in water—but once acetate forms, the resulting ion is unusually stable for a conjugate base. Resonance also affects the geometric structure: the carbon in the carboxylate group displays planar trigonal geometry, with bond angles close to 120°, while the methyl carbon maintains a tetrahedral geometry. The contrast between the nonpolar methyl group and the negatively charged carboxylate group gives acetate an amphiphilic character: part of the molecule interacts strongly with polar molecules like water, while the other interacts favorably with hydrophobic environments. This dual nature explains why acetate dissolves readily in water but can also function in organic chemistry and biological systems where polarity varies.
The chemical structure determines how acetate behaves in solution. As an anion, acetate forms salts by pairing with positively charged ions known as cations. Salts such as sodium acetate, potassium acetate, calcium acetate, and ammonium acetate are highly soluble and widely used in laboratory, industrial, medical, and environmental settings. Sodium acetate, for example, is used in buffer solutions to control pH, and in heat packs that exploit crystallization energy. Ammonium acetate plays major roles in liquid chromatography and pharmaceutical formulation. Metal acetates—including copper(II) acetate, zinc acetate, and iron(II) acetate—are employed in catalysis, textile processing, and material science. The ability of acetate to coordinate with a broad array of cations stems from the negatively charged oxygen atoms, which act as electron-donating sites in coordination chemistry.
Beyond industrial chemistry, acetate occupies a central role in biochemistry and metabolism. In living organisms, acetate is incorporated into the acetyl group (CH₃CO–) that forms acetyl-CoA, one of the most crucial molecules for energy production, fat synthesis, and cellular respiration. Within the Krebs cycle (citric acid cycle), acetyl-CoA delivers two carbon units that eventually lead to the release of energy stored in carbohydrates, fats, and proteins. The reason acetate plays this metabolic role lies again in its structural balance: the carboxylate end provides reactivity while the methyl group supplies carbon for synthesis. Acetate also contributes to fatty acid synthesis, cholesterol generation, and biosynthesis of numerous hormones. In anaerobic conditions, some microorganisms produce acetate through fermentation as a survival strategy for energy extraction. These biological pathways highlight acetate not simply as a lab reagent but as a core molecule upon which life’s biochemical machinery depends.
Acetate’s significance expands when viewed as a participant in buffer systems. Acetic acid and acetate together form a conjugate acid–base pair that stabilizes pH in solutions by neutralizing added acids or bases. When hydrogen ions enter the system, acetate captures them to form acetic acid; when hydroxide ions appear, acetic acid donates hydrogen ions and regenerates acetate. This dynamic equilibrium enables precise pH control, especially in biochemical experiments, pharmaceutical formulations, and industrial processes. The acetate buffer system illustrates how chemical structure translates into macroscopic influence: acidity regulation relies on the predictability of acetate’s ability to accept protons due to its resonance-stabilized charge.
Everyday life contains multiple examples of acetate’s presence. Vinegar, widely used in cooking, consists mainly of acetic acid in water; once ingested, it converts to acetate in the body. Cellulose acetate—a polymer derived from acetate—is used in eyeglass frames, photographic film, filters, clothing fibers, and biodegradable plastics. Sodium acetate trihydrate is found in reusable heat packs that crystallize and release stored heat with a single click of a metal disk. Zinc acetate has medical applications in lozenges formulated for cold symptom relief. Although these uses span unrelated fields, they draw from the same molecular trait: acetate’s resonance-stabilized structure makes it chemically predictable, allowing it to respond consistently when incorporated into materials, solutions, or biological systems.
The behavior of the acetate ion also provides an instructive example of how chemistry links microscopic structure to macroscopic properties. The distribution of electrons across the two oxygen atoms determines solubility, acidity, buffering capacity, biological reactivity, and coordination chemistry. A single substitution in the methyl group or alteration of the carboxylate environment has the potential to transform the molecule into something new—an ester when combined with alcohols, an amide when paired with amines, a polymer precursor when activated, or an intermediate in metabolism depending on biochemical context. Acetate therefore acts as a structural platform, a versatile unit that chemistry manipulates into countless compounds.
In the end, the acetate ion is more than a simple anion: it is a dynamic molecular entity whose stability, reactivity, and resonance-driven structure influence organic chemistry, energy metabolism, materials science, medicine, and industrial technology. Its dual character — a nonpolar carbon backbone paired with a charged carboxylate head — enables it to participate in environments ranging from the watery cytoplasm of living cells to the high-temperature reactors of synthetic manufacturing. Through its ability to form salts, buffer systems, metabolic intermediates, polymers, and coordination complexes, the acetate ion demonstrates how a seemingly small molecular structure can become a cornerstone of both life and applied science.