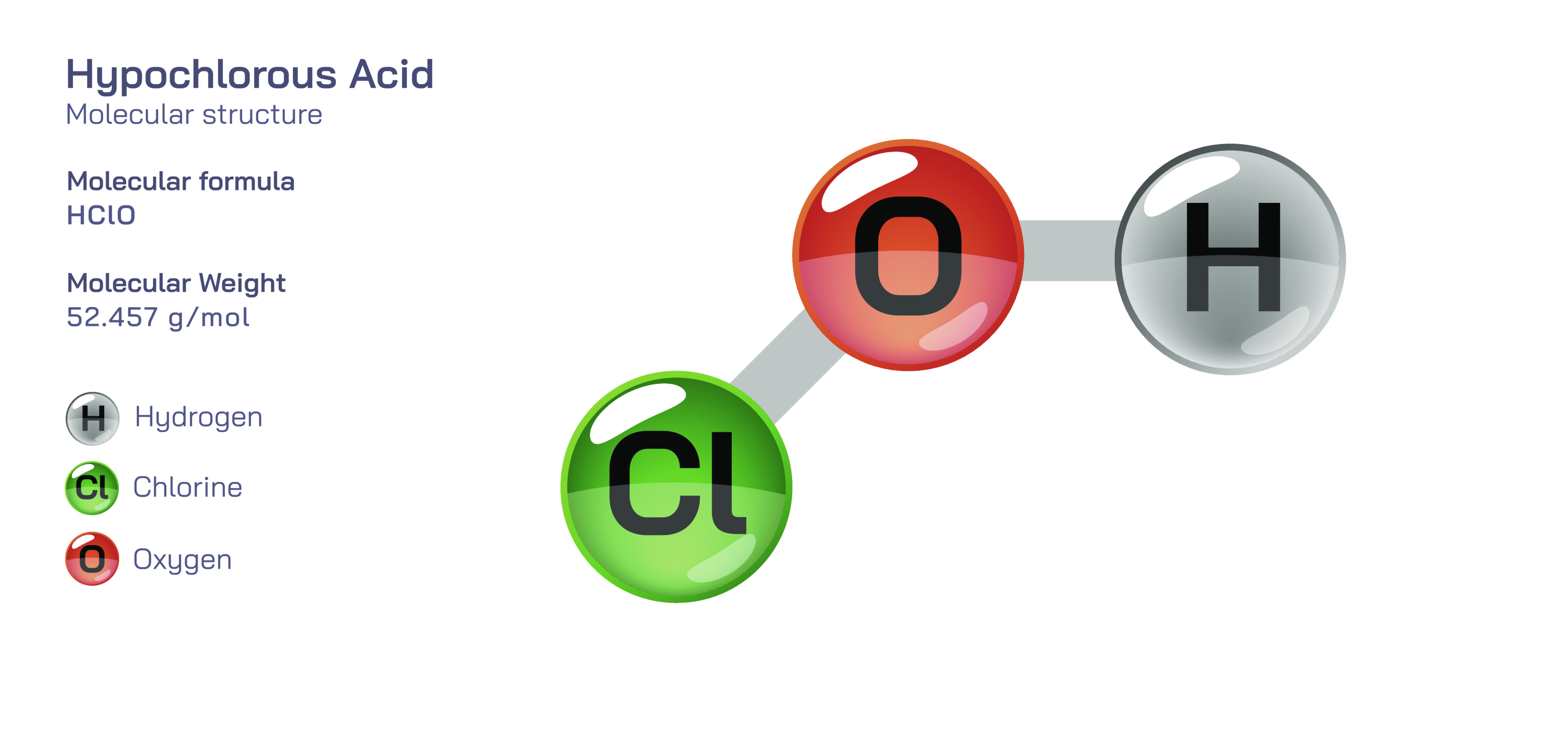
Hypochlorous Acid Chemical Structure — HOCl Molecule Diagram and Chemical Behavior Explained
Hypochlorous acid, expressed chemically as HOCl, is a molecule whose simplicity disguises one of the most important and versatile roles in both chemistry and biology. Formed from only three atoms — hydrogen, oxygen, and chlorine — the molecule embodies a structural balance that allows it to function as a powerful oxidizing agent, a selective antimicrobial substance, and a vital component of immune defense in living organisms. Although HOCl is often associated with disinfectants and sanitizing solutions, its deeper identity becomes clear only when the structure of the molecule and the distribution of electrons within its chemical bonds are examined. The arrangement of the atoms, rather than their number, determines every property HOCl demonstrates, including its ability to destroy pathogens, oxidize organic matter, and react dynamically with biomolecules. The molecule’s behavior is driven by the electronegativity contrast between oxygen and chlorine, the polarity of the O–Cl bond, and the ease with which hydrogen separates in aqueous environments. These features are responsible for the dual nature of hypochlorous acid: it is a weak acid in the traditional acid–base sense, yet one of the most potent oxidants and disinfectants in biological and industrial chemistry.
At the structural level, hypochlorous acid is composed of a hydrogen atom bonded to an oxygen atom, which in turn is bonded to a chlorine atom, forming the sequence H–O–Cl. This asymmetric arrangement creates a highly polar molecule. Oxygen is the most electronegative of the three atoms, meaning it pulls electron density strongly toward itself. This leaves hydrogen partially positive and chlorine partly electron-deficient but surrounded by a diffuse electron cloud because of its large atomic radius. The result is a molecular configuration in which hydrogen is loosely attached and chlorine is primed to participate in electron-transfer reactions. When HOCl enters water, it can donate its proton to form the hypochlorite ion (ClO⁻), but even before dissociation, the molecule interacts aggressively with electron-rich biological and chemical targets. The oxygen–chlorine bond, weaker than the equivalent bond in oxygen–bromine or oxygen–iodine systems, becomes the functional pivot through which redox activity occurs. What defines HOCl most clearly is the presence of chlorine in a high oxidation state, which makes it hungry for electrons. Any contact with molecular structures capable of donating electrons — especially proteins, DNA bases, enzymes, cell membranes, and pigments — triggers oxidation that leads to molecular disruption.
The extraordinary antimicrobial power of hypochlorous acid originates from this structural ability to target electron-rich biological groups. HOCl reacts with sulfhydryl and thioether groups in proteins, neutralizing enzymes essential for microbial metabolism. It modifies membrane lipids by oxidative chlorination, compromising cell wall integrity and causing leakage of cellular contents. It attacks nucleic acids, disrupting genetic instructions needed for replication and repair. Unlike many disinfectants that require high concentrations or long exposure times, HOCl works efficiently because its molecular size allows it to penetrate microbial cell barriers, and its electrophilicity enables rapid reaction once inside. In living systems, this function is not artificial but natural: the human immune system relies on hypochlorous acid as a primary defense mechanism. When white blood cells known as neutrophils encounter invading microorganisms, they trigger an enzymatic process that converts chloride ions and hydrogen peroxide into HOCl inside a structure called the phagosome. The resulting burst of hypochlorous acid destroys bacteria and viruses within seconds, showing that this molecule serves not only technological sanitation but biological survival. The same chemical structure that allows HOCl to deactivate pathogens in disinfectants is at work inside the human body as a controlled biochemical weapon.
In industrial and environmental chemistry, hypochlorous acid plays a central role in water treatment, sanitation, and bleaching. When chlorine gas or sodium hypochlorite dissolves in water, HOCl forms as the primary active disinfecting species. The effectiveness of chlorinated water depends largely on the proportion of HOCl compared to its conjugate base, ClO⁻. This balance is governed by pH: at lower and neutral pH levels, HOCl dominates, while at higher pH levels the equilibrium shifts toward hypochlorite. Because HOCl is much more effective as a disinfectant than ClO⁻ — nearly 100 times more reactive toward biological contaminants — the regulation of pH becomes critical for water purification systems, swimming pools, medical sanitation, and industrial cleaning. The reason for this dramatic difference rests again in molecular structure. HOCl is uncharged and can penetrate biological membranes easily, while ClO⁻ carries a negative charge that limits penetration. Thus, the same molecule changes in potency simply by losing or retaining a proton, and the control of this equilibrium determines whether chlorine-based water systems function efficiently or weakly.
One of the most striking aspects of HOCl chemistry is the way its structure allows selective oxidation. Many oxidants break down all organic matter indiscriminately, but hypochlorous acid displays unusual preference toward amino acids that contain sulfur and nitrogen. This selectivity is precisely why it destroys pathogens while leaving surrounding environmental organic matter largely untouched under controlled concentrations. In physiological systems, selectivity prevents HOCl from damaging entire tissues during immune response by concentrating its reactivity on foreign cells rather than indiscriminately reacting with all biomolecules. Nonetheless, excessive or prolonged exposure can overwhelm biological defenses, which explains why strong HOCl solutions can irritate skin, eyes, and mucous membranes: they apply the same oxidative mechanism outside of the immune regulatory system.
The chemical stability of hypochlorous acid is tightly linked to its structure. At room temperature and in pure water, HOCl decomposes gradually into chloride and chlorate depending on conditions such as light, heat, metal ions, and organic contaminants. This instability is not a weakness but a consequence of being a high-energy molecule designed to oxidize and then neutralize itself. The O–Cl bond can break through redox reactions that release energy, and this property ensures that HOCl does not accumulate indefinitely in ecosystems or in water distribution systems. Instead, it exists long enough to exert disinfecting power and then decays into lower-energy species such as chloride, reducing chemical persistence in the environment. This short lifetime explains why HOCl must be generated on-site in many sanitation systems and cannot be stored for long periods without stabilizers.
Safety considerations surrounding hypochlorous acid derive from the same chemical traits responsible for its benefits. Concentrated HOCl or solutions rich in active chlorine can corrode skin and damage tissue by oxidizing biological molecules in an unregulated manner. Contact with acidic conditions causes the equilibrium to shift toward formation of chlorine gas, a highly toxic respiratory irritant. Mixing HOCl solutions with ammonia or amine-based cleaners produces chloramines and nitrogen trichloride, compounds harmful to eyes and lungs. These hazards are not anomalies of manufacturing but direct consequences of how HOCl behaves at the molecular level. The reactivity that protects drinking water from pathogens also demands careful storage, proper dilution, and thoughtful use.
Despite its risks, hypochlorous acid remains one of the most valuable and irreplaceable molecules in public health, environmental sanitation, and biology. It protects drinking water globally, supports surface disinfection in disease control, maintains hygiene in medical facilities, and plays a biological role in innate immunity. The extraordinary reach of HOCl across such diverse fields originates from one feature — the polarity and electron distribution of the H–O–Cl structure. The molecule’s identity emerges from the electronegativity gradient between hydrogen, oxygen, and chlorine, and from the chlorine center’s ability to accept electrons from organic and biological substrates.
In its deepest interpretation, hypochlorous acid is a vivid reminder that molecules shape the world not through size or complexity, but through structure and electron behavior. HOCl demonstrates that a molecule of only three atoms can influence medicine, sanitation, environmental safety, microbiology, industrial chemistry, and immune function. Its power lies not in brute strength but in precision: the exact placement of hydrogen, oxygen, and chlorine in a single chain defines how the molecule behaves, how it reacts, and how it interacts with life. Understanding hypochlorous acid means appreciating that the smallest structural variations within a molecule can determine whether it becomes a life-preserving disinfectant, a weapon of the immune system, a tool of industry, or a hazard requiring respect and control. Through this perspective, HOCl stands not simply as a disinfectant compound, but as one of the most chemically consequential and biologically integrated molecules in human experience.