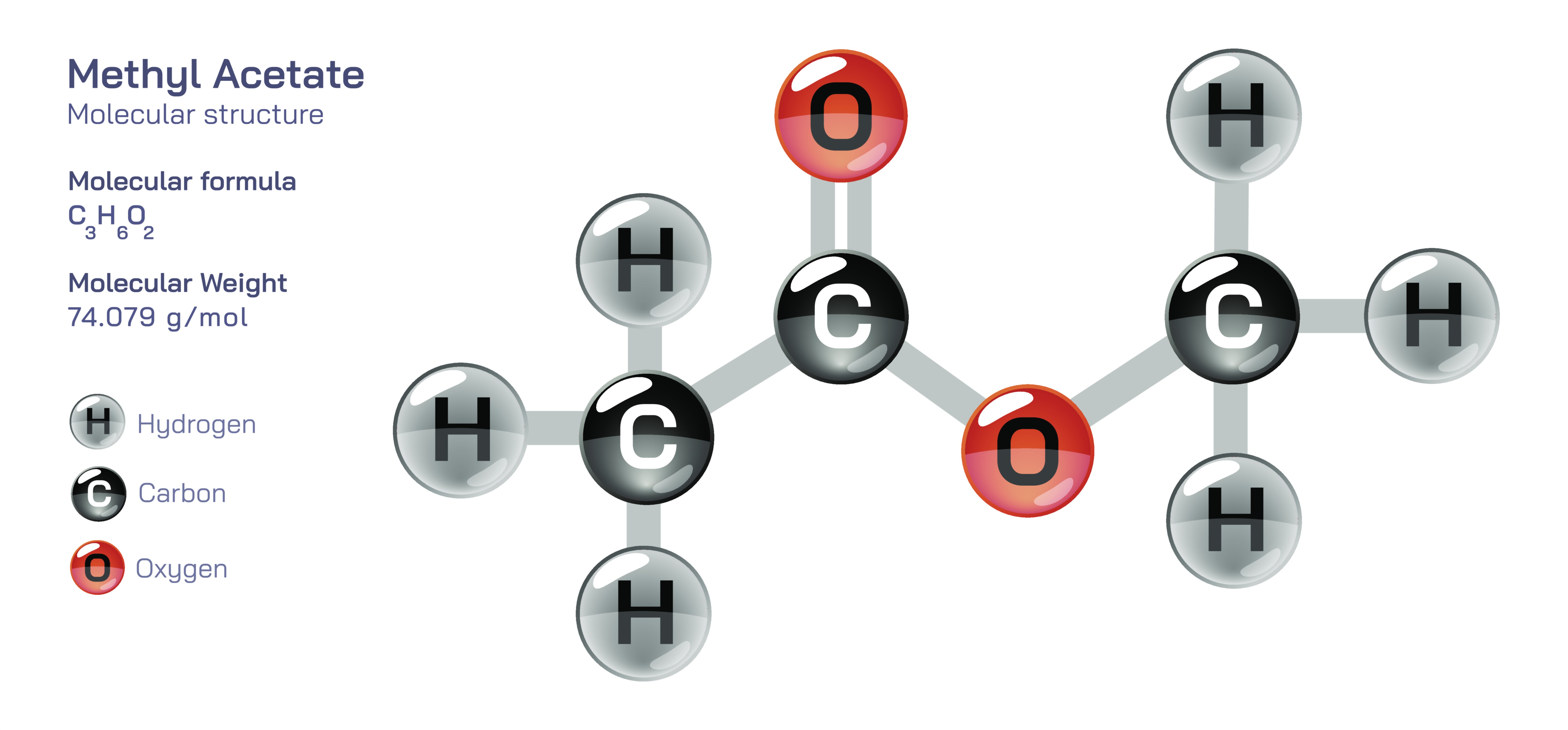
Methyl Acetate — Molecular Structure and Composition
Methyl acetate is an organic compound whose molecular structure illustrates how small, precise arrangements of carbon, hydrogen, and oxygen atoms can create a molecule uniquely suited to high-performance industrial applications, delicate formulation chemistry, and complex environmental interactions. Its chemical formula, C₃H₆O₂, appears simple at first glance, yet the internal architecture of the molecule defines a distinct balance of volatility, solvency, flammability, and biological degradability that has made methyl acetate indispensable across coatings, adhesives, cleaning formulations, inks, polymer processing, pharmaceuticals, and fine-chemical synthesis. Structurally, methyl acetate belongs to the ester family, which means the molecule contains the ester functional group –COO– linking two carbon components. In this case, the parent fragments are an acetyl (CH₃CO–) unit bonded to a methyl (–OCH₃) group. The ester linkage arises when acetic acid and methanol combine through a condensation reaction that removes water, forming a carbonyl carbon double-bonded to oxygen and simultaneously single-bonded to another oxygen atom that connects to the methyl substituent. This arrangement gives methyl acetate a central carbonyl group that is strongly polarized, creating electron-rich and electron-deficient regions throughout the molecule and enabling a wide range of controlled chemical interactions. As a result, methyl acetate becomes neither overly reactive nor functionally inert: it maintains structural stability during storage and use yet can undergo hydrolysis, transesterification, or catalytic modification when exposed to the right environments. The seemingly modest combination of three carbons, six hydrogens, and two oxygen atoms thus produces a molecule that balances volatility, polarity, and chemical reactivity in a way that few solvents can match.
The molecular identity of methyl acetate is inseparable from its physical behavior. Because the oxygen atoms in the ester linkage contribute to a permanent dipole moment, the molecule displays moderate polarity strong enough to solubilize a diverse range of polymers, resins, plasticizers, surface coatings, and organic films, yet weak enough to remain miscible with many nonpolar and weakly polar organic solvents. This duality is essential in industrial coatings and ink systems, where formulations require solvents that leave microstructures consistent and uniform when evaporated. Methyl acetate evaporates quickly relative to many traditional solvents because its molecular interactions with neighboring molecules are not dominated by hydrogen bonding but by dipole–dipole attraction and London dispersion forces. This volatility allows the molecule to perform fast-drying functions in surface finishing, cleaning systems, and aerosol-based coatings. While its boiling point is lower than that of many esters, its vapor pressure and evaporation rate are controlled enough that films formed from methyl acetate-containing formulations do not suffer from undesirable surface bubbling or uneven texture provided that the blend is properly engineered. This physical behavior stems directly from the geometric distribution of electrons around the carbonyl group and the methyl substituent, which together generate a molecule that prefers mobility over strong intermolecular bonding.
Chemically, the ester linkage endows methyl acetate with reactivity that can be controlled and exploited in manufacturing. The carbonyl carbon is susceptible to nucleophilic attack under acidic or basic conditions, making methyl acetate a substrate for reactions such as saponification, transesterification, and alcoholysis. When exposed to water and a catalyst, the ester can hydrolyze and revert to acetic acid and methanol, a reversible reaction that has been used extensively to study equilibrium thermodynamics in ester chemistry. In industrial transformation pathways, methyl acetate is frequently used as a precursor or intermediate in processes that require selective ester cleavage or exchange without disrupting other functional groups. This selective reactivity arises from predictable electron distribution around the carbon–oxygen double bond and the electronegativity difference between oxygen and carbon. The ability to undergo substitution at the ester bond while resisting uncontrollable decomposition is a major reason why methyl acetate serves as a feedstock in polymer synthesis, cellulose processing, and the manufacture of fine chemicals. The molecule operates as an active participant in synthesis when needed and as a stable solvent when not, highlighting how structural identity dictates chemical versatility.
Methyl acetate’s composition also determines how it behaves in biological and environmental systems. Because the ester bond is naturally hydrolyzed by water and biological enzyme systems, methyl acetate is biodegradable and breaks down into methanol and acetic acid, which are further metabolized by microbial communities. This hydrolytic susceptibility contrasts with the environmental persistence of chlorinated and aromatic solvents and is one reason methyl acetate is often chosen for eco-conscious surface coatings and cleaning products intended to minimize environmental footprint. Nevertheless, the molecule’s rapid evaporation can lead to temporary accumulation in air in poorly ventilated spaces, and occupational safety practices take into account its potential to cause irritation or central nervous system effects at high vapor concentration. The biological safety profile is therefore not determined only by biodegradability but also by volatility and kinetic interactions with exposed tissues. The same molecular architecture that yields effective solvency also enables rapid inhalation exposure, demonstrating how structural properties directly govern both industrial usefulness and safety considerations.
In formulation science, methyl acetate has acquired a reputation as a high-performance solvent capable of balancing solvency strength and evaporation characteristics without damaging delicate substrates. It dissolves nitrocellulose, acrylic polymers, vinyl resins, polyester resins, polyurethane precursors, and numerous synthetic coatings that require both fast drying and clean film formation. Its moderate polarity allows it to bridge resin-rich and pigment-rich components in ink systems, promoting uniform dispersion and smooth deposition. In adhesive chemistry, methyl acetate supports pressure-sensitive and hot-melt adhesive technologies by dissolving polymeric tackifiers while evaporating quickly enough to allow rapid bonding. Its compatibility with a wide range of co-solvents enables engineered solvent blends that adjust viscosity, flash point, flow level, and curing rate to meet diverse manufacturing requirements. Every one of these functions is ultimately rooted in the molecular geometry of the ester group and the way electron distribution shapes intermolecular forces across liquid and vapor phases.
Because of its efficient combustion behavior and clean decomposition pattern, methyl acetate has also emerged as a modern alternative to more hazardous solvents in sectors that prioritize environmental performance. When burned, methyl acetate produces carbon dioxide and water without forming halogenated by-products or heavy soot. While its flammability demands responsible handling, this same characteristic allows coatings and industrial systems to benefit from rapid curing under heat-assisted drying without excessive pollutant formation. Compared with aromatic solvents such as toluene and xylene, methyl acetate provides fast-drying capability with lower odor intensity and greater biodegradability, making it valuable in consumer-facing products where user comfort and environmental impact matter. The shift toward “green chemistry” in the coatings and adhesives industries reflects a growing recognition that molecular structure—not just application performance—should align with environmental responsibility.
From a broader perspective, methyl acetate illustrates how the addition of a single functional group to a hydrocarbon chain can dramatically alter the physical world. The transformation of methanol and acetic acid into methyl acetate introduces an ester bond that changes nearly every property—from solubility and polarity to volatility, reactivity, and environmental fate. These changes are not abstract; they determine whether a substance becomes an industrial solvent, a synthetic intermediate, a fuel, a pollutant, or a biodegradable chemical. In methyl acetate, the molecular structure serves as a blueprint for a compound that is simultaneously stable, reactive, volatile, biodegradable, and industrially efficient. Its influence spans coatings labs, automotive refinish shops, printing plants, electronics cleanrooms, polymer factories, environmental engineering, and chemical synthesis. The molecule demonstrates how composition defines what atoms are present, while structure dictates what those atoms can accomplish.
Whether enabling the rapid drying of high-performance coatings, dissolving precision-engineered polymers, participating in controlled chemical conversion pathways, breaking down safely in the environment, or supporting advances in sustainable industrial chemistry, methyl acetate continues to prove that even small molecules have broad scientific and technological impact. The reason lies not in size but in structure: when carbon, hydrogen, and oxygen are arranged into an ester configuration, they produce a molecule with a unique balance of volatility, polarity, and reactivity that industry and chemistry continue to rely on. Through this blend of molecular simplicity and functional sophistication, methyl acetate stands as a clear example of how the invisible architecture of atoms quietly shapes modern manufacturing, environmental sustainability, and scientific progress.