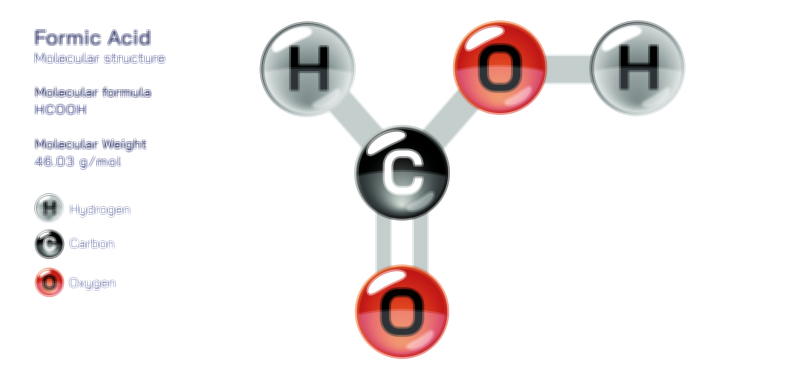
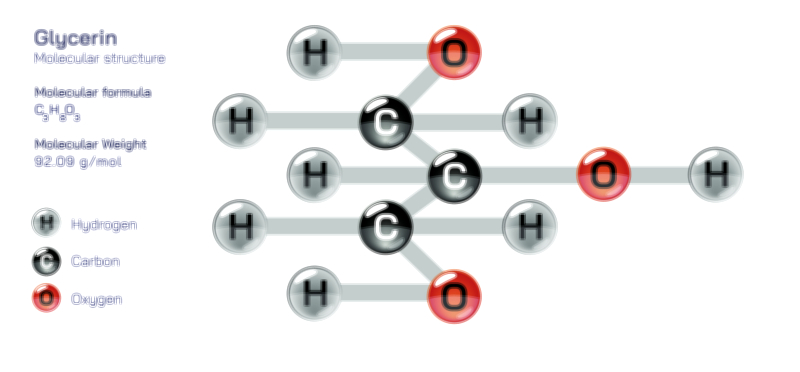
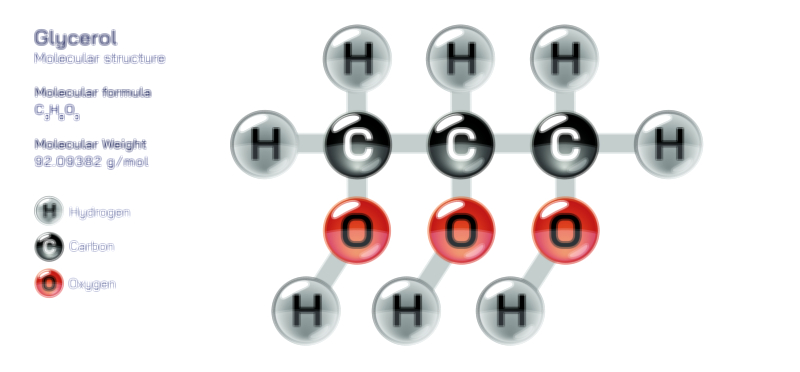
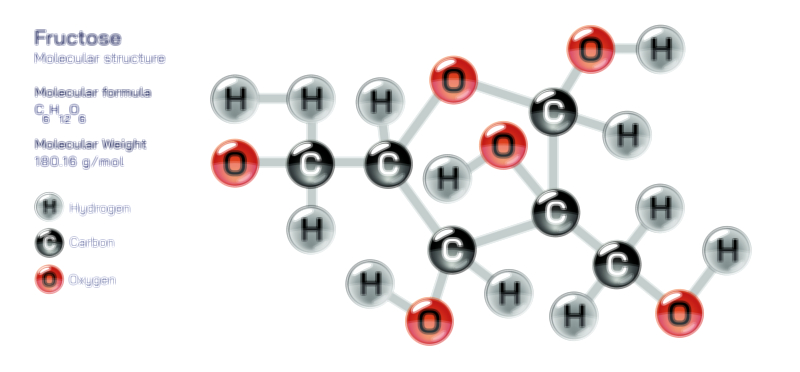
Ethylene Molecular Structure — Organic Compound Diagram for Chemistry and Biology
Ethylene stands as one of the simplest, most influential, and scientifically significant organic molecules, serving as a foundational example for understanding unsaturated hydrocarbons, double-bond chemistry, molecular geometry, and biological signaling. Although its structure is minimal—just two carbon atoms and four hydrogen atoms—the arrangement of those atoms creates a molecule with distinctive properties that reach far beyond its size. Ethylene is central to polymer science, atmospheric chemistry, industrial manufacturing, plant biology, and fundamental organic chemistry. When explored through detailed descriptive explanation, its structural features reveal elegant rules that define how organic molecules form, react, assemble, and transform. A vector illustration or diagram of ethylene generally highlights its carbon–carbon double bond and symmetrical arrangement of hydrogen atoms, but beneath these simple lines lies a deeply informative molecular architecture that influences chemical reactivity, biological processes, and technological applications across many fields.
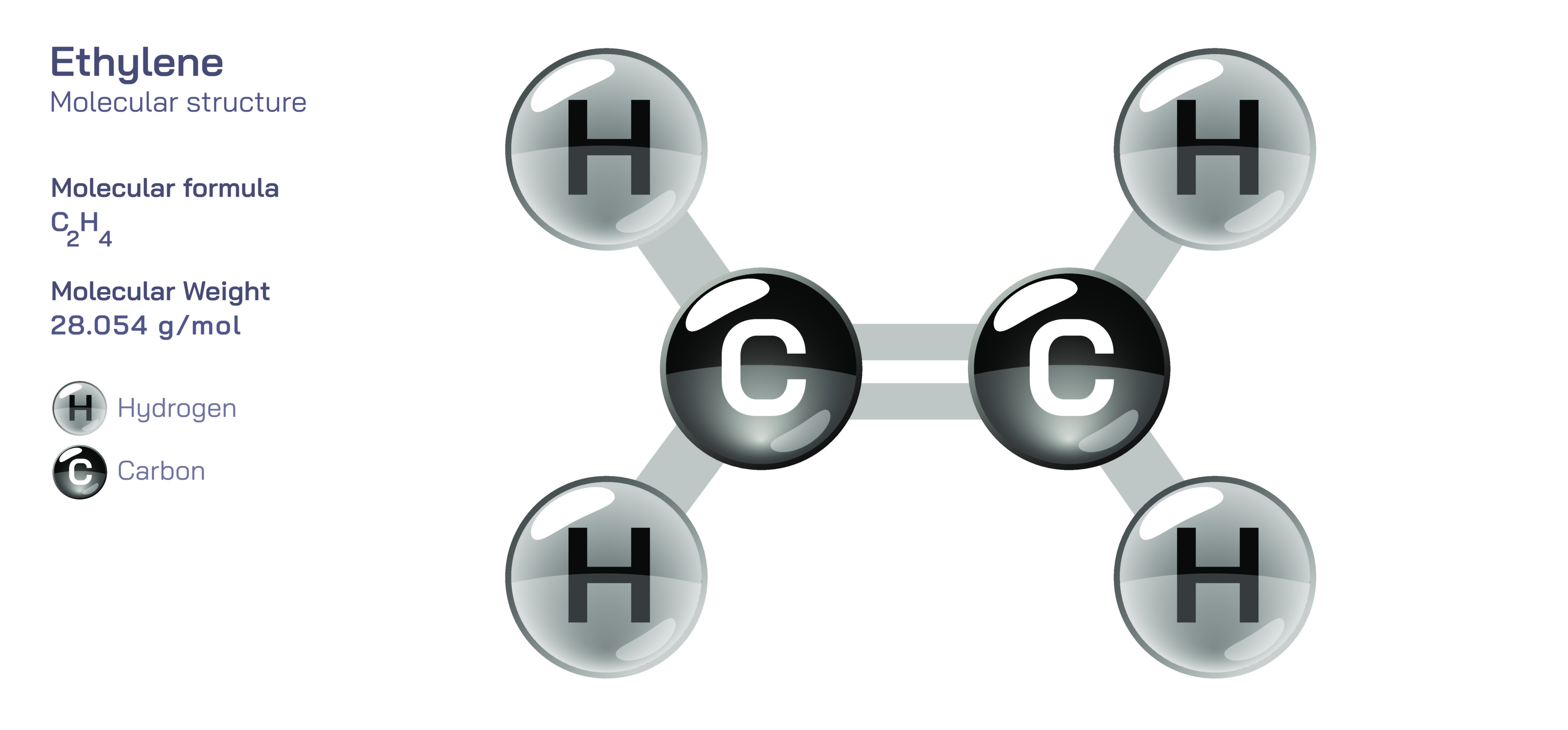
Ethylene has the molecular formula C₂H₄ and is structurally represented as H₂C=CH₂. The defining feature of this molecule is its carbon–carbon double bond, which not only connects the two carbon atoms but also establishes the rigid, planar geometry that shapes its chemical behaviour. In contrast to single bonds, which allow unrestricted rotation, the double bond in ethylene locks the two carbon atoms into a fixed relationship relative to one another. This rigidity arises because a double bond consists of two overlapping bonding interactions: one sigma bond formed by direct orbital overlap along the axis between the carbon atoms, and one additional bonding interaction that emerges from side-by-side overlap of orbitals. The presence of this second interaction prevents free rotation, creating a structural constraint that influences how the molecule participates in chemical reactions and how it assembles into larger structures such as polymers.
The planar geometry of ethylene results from the hybridization state of the carbon atoms. Each carbon atom is bonded to two hydrogen atoms and the other carbon through three bonding regions, causing it to adopt a trigonal planar shape. While diagrams usually present this structure flat on the page, the actual molecule is genuinely planar, with all six atoms lying in the same plane. The hydrogen atoms around each carbon arrange themselves roughly one hundred and twenty degrees apart, forming a balanced and symmetrical arrangement that minimizes electron repulsion. This geometry is essential for understanding ethylene’s behaviour, because the orientation of orbitals around the double bond directly influences how the molecule interacts with reagents, catalysts, and biological targets.
The double bond not only determines the geometry but also defines the molecule’s reactivity. Ethylene’s double bond is considered an unsaturated region, meaning it contains fewer hydrogen atoms than would be present in a fully saturated molecule of similar size, such as ethane. This unsaturated feature creates an area of concentrated electron density that makes the molecule particularly reactive toward species that can accept electrons or attach to the carbon atoms. In this sense, the diagram of ethylene, with its clearly marked double bond, is more than an illustration—it is a representation of a chemically active site. Many reactions involving ethylene occur at this double bond, where molecules such as halogens, acids, or catalysts can add across the bond, converting the unsaturated system into a saturated one. This reactivity forms the basis for numerous industrial processes, including polymerization, oxidation, hydration, and chlorination.
Ethylene’s role in polymer chemistry is especially significant. Its structure makes it the primary monomer used to create polyethylene, one of the most widely manufactured plastics in the world. When ethylene undergoes polymerization, the double bond opens, allowing carbon atoms from one ethylene molecule to link with carbon atoms from others. This reaction forms long chains or networks of carbon atoms, generating materials that vary in flexibility, strength, and transparency depending on how the chains form and how they pack together. The fact that such an immense range of commercial materials arises from a molecule as simple as ethylene illustrates how profoundly structure dictates function. The double bond’s ability to convert into single bonds during polymer formation exemplifies the chemical utility embedded within this small unsaturated hydrocarbon.
Beyond industrial significance, ethylene plays a surprisingly important role in biological systems. In plants, ethylene acts as a hormone that regulates numerous developmental processes, including fruit ripening, leaf abscission, flower opening, and stress responses. Its small size allows it to diffuse easily through plant tissues, while its structure enables it to interact with specific receptors that initiate biochemical pathways. The same features that give ethylene high reactivity in chemical environments—its electron-rich double bond and planar geometry—also allow it to participate in biological signaling. Understanding ethylene’s structure therefore helps explain how such a simple molecule can produce profound physiological effects in crops, ecosystems, and agricultural systems. Even the aroma of certain ripening fruits arises from ethylene release and response, showing how chemistry and biology intersect through molecular structure.
Because of its unsaturated nature, ethylene participates in atmospheric reactions as well. Its structure allows it to interact with reactive oxygen species and sunlight-driven radicals in the atmosphere, influencing the formation of secondary pollutants and contributing to chemical cycles that affect air quality. While naturally occurring ethylene from plant emissions plays a role in environmental processes, anthropogenic sources, such as industrial refining, vehicle exhaust, and biomass burning, increase its presence in the atmosphere. Its structure enables rapid reactions that ultimately feed into broader atmospheric chemistry, demonstrating once again that molecular architecture determines not only microscopic behaviour but large-scale environmental outcomes.
Ethylene’s structure also provides a gateway to understanding stereochemistry and geometric isomerism. Although ethylene itself cannot show cis–trans isomerism because it is symmetrical, its double-bond framework introduces the concept that substituents on double-bonded carbons can adopt fixed positions. The planar and rigid nature of the ethylene double bond becomes the foundation upon which stereochemistry builds, explaining why isomers of larger alkenes differ in physical properties, reactivity, and biological activity. Ethylene illustrates the simplest form of this bonding reinforcement, preparing learners to recognize how unsaturated molecules adopt specific shapes that cannot freely interchange without breaking bonds. In this way, the structural diagram of ethylene becomes not just a specific representation but a model for interpreting broader chemical principles.
The symmetrical nature of ethylene also influences its physical properties. The molecule is nonpolar overall, even though the double bond contains localized electron density. Its symmetry ensures that the dipoles cancel out, making the entire molecule relatively unreactive toward polar solvents and highly soluble in nonpolar environments. Its low molecular weight results in a gas at room temperature, and its relatively weak intermolecular forces explain its low boiling point. These characteristics stem directly from its structural simplicity: the combination of symmetry, lack of dipole moment, and absence of highly electronegative functional groups keeps the molecule light, nonpolar, and mobile. A vector illustration with evenly placed hydrogens around the double bond subtly communicates this symmetry, allowing viewers to infer the molecule’s physical behaviour even from a simplified drawing.
Despite its simplicity, ethylene also plays a role in energy science. Its double bond stores a moderate amount of chemical energy that can be released through combustion or transformed through catalytic reactions. In industrial cracking processes, ethylene is produced from larger hydrocarbons through controlled heat input, exploiting its stable yet reactive structure. Once produced, it becomes a versatile building block for creating polymers, solvents, chemical intermediates, and various organic materials. Its structure is therefore not merely a static arrangement of atoms but a gateway to countless synthetic pathways that define modern chemical manufacturing.
In toxicological and safety contexts, ethylene’s structure influences how it behaves in the environment. Because it is a simple hydrocarbon, its reactivity and biological interactions differ from those of alcohols, aldehydes, or acids. Ethylene is not strongly toxic but can contribute to oxygen displacement in enclosed spaces due to its gaseous form. Its chemical inertness toward many solvents and its low solubility in water also result from its symmetrical structure. Understanding these behavioural principles begins with examining the arrangement of atoms in the molecule and recognizing how structure dictates interaction.
From an educational perspective, ethylene’s structure offers a perfect illustration of unsaturated hydrocarbons, sigma–pi bonding, planar geometry, hybridization, and the reactivity of alkenes. Students encountering alkene chemistry for the first time learn through ethylene that double bonds act as reactive centers, do not rotate, and participate in specific reactions that differ from those of alkanes. Ethylene diagrams often break down the bonding visually, showing how the double bond forms and how orbitals overlap. Even without mathematical treatment, these diagrams express concepts about electron density, molecular constraints, and structural rigidity that become foundational for understanding more advanced organic chemistry topics.
Ethylene is also widely used as a reference molecule in spectroscopy. Because of its symmetrical structure, it displays characteristic infrared absorption patterns and shows predictable behaviours under nuclear magnetic resonance analysis. These features help spectroscopists interpret signals from more complex molecules, using ethylene as a benchmark. Its vibrational modes, electron distribution, and bond characteristics provide a model system for studying organic molecules at fundamental levels.
Ultimately, the structure of ethylene—simple, symmetric, planar, and centered around a carbon–carbon double bond—captures the essence of how molecular architecture shapes the behaviour of organic compounds across chemistry, biology, industry, and the environment. Whether represented through a clean vector illustration or examined through three-dimensional modeling, the ethylene molecule serves as a foundational example of how even the smallest structural features lead to extensive functional consequences. Its role as a building block, a biological messenger, a reactive intermediate, and an industrial powerhouse demonstrates the profound impact of molecular structure. Through careful examination of ethylene’s architecture, one gains insight not only into this single molecule but into the broader principles that govern the entire world of organic chemistry.