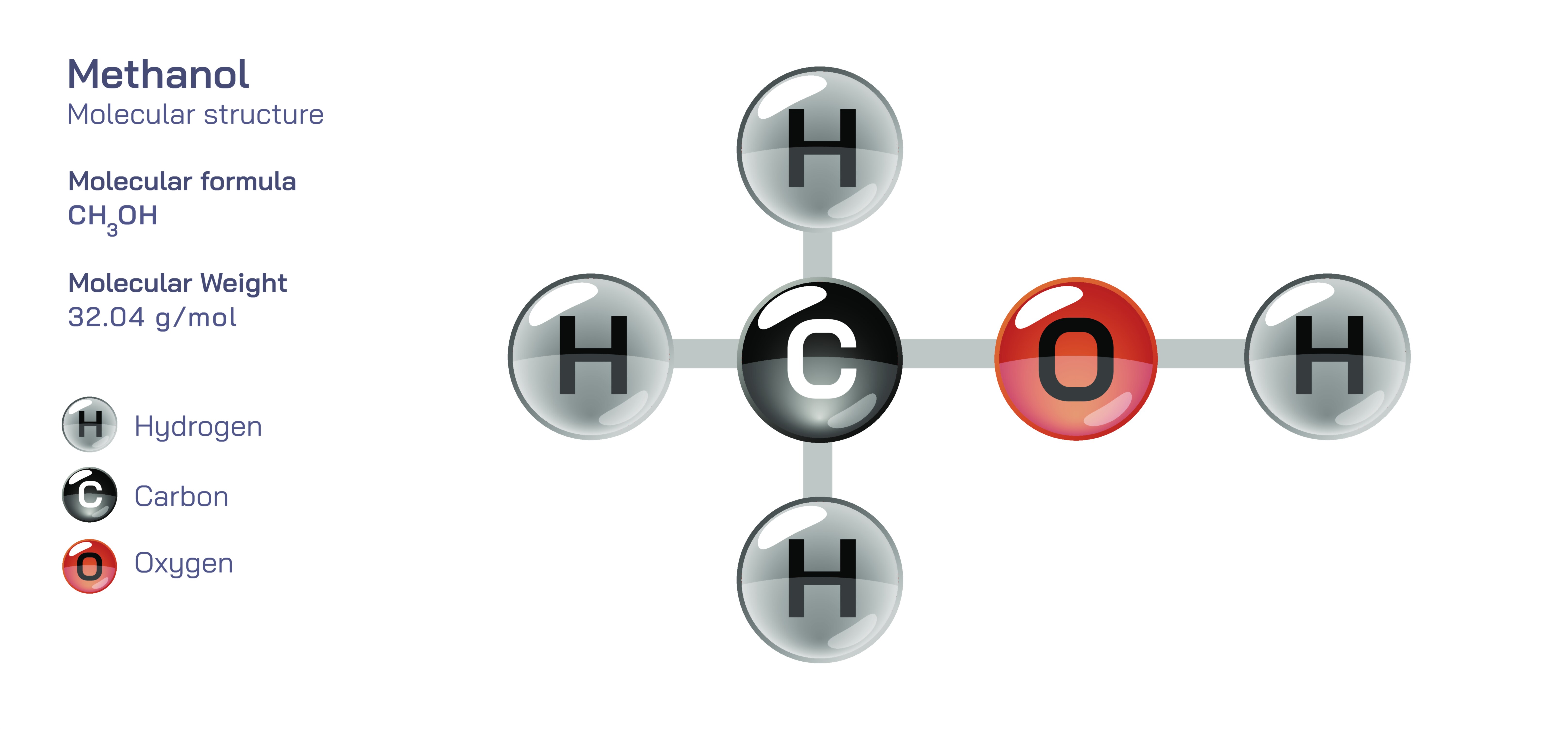
Methanol — Molecular Structure and Composition
Methanol is the simplest molecule in the alcohol family, yet its structural design and chemical behavior have shaped major areas of energy production, industrial chemistry, environmental science, and biological safety. Defined by the formula CH₃OH, methanol contains one carbon atom bonded to three hydrogen atoms and one hydroxyl group, creating a compact molecular architecture that behaves differently from both its hydrocarbon counterpart methane and the larger alcohols that follow it in the chemical series. Within this structure, the carbon atom forms four single covalent bonds, three directed toward hydrogen and one toward an oxygen atom. The oxygen, in turn, binds to a hydrogen atom, forming the hydroxyl group that defines the molecule as an alcohol. Even though this change from four hydrogens in methane to three hydrogens plus hydroxyl in methanol appears small, it completely transforms the physical and chemical identity of the molecule. Methanol becomes polar rather than nonpolar, hydrogen-bonding rather than chemically inert, miscible with water instead of insoluble, and reactive enough to form esters, ethers, aldehydes, and numerous carbon-based industrial derivatives. Its structure creates a distinct combination of volatility, flammability, solvency, and biological reactivity, making methanol a foundational compound in both natural and engineered systems.
The polarity of methanol is one of the most crucial consequences of its composition, because the carbon–oxygen bond and the oxygen–hydrogen bond create strong dipole moments that shape the molecule’s interactions with solvents, surfaces, and biological membranes. The hydroxyl group is capable of donating and accepting hydrogen bonds, allowing methanol to mix uniformly with water across all concentrations. This contrasts sharply with methane, from which methanol is derived via oxidation, and highlights how adding a single oxygen atom to a hydrocarbon backbone changes solubility behavior at a fundamental level. The hydrogen-bonding potential makes methanol an excellent solvent for organic and inorganic substances alike, especially those that are too polar for hydrocarbon solvents but not polar enough for water alone. This dual compatibility is a direct expression of methanol’s hybrid structure, which combines a hydrophobic methyl group and a hydrophilic hydroxyl group within one molecule. Such molecular compatibility is responsible for the valuable role methanol plays in analytical chemistry, resin manufacturing, pharmaceutical synthesis, and coating formulations, where uniform solvation and controlled evaporation are necessary to maintain product integrity.
Methanol’s structure also dictates its energetic behavior. When combusted in oxygen, methanol releases significant heat relative to its molecular size because oxidation of the carbon and hydrogen atoms results in the formation of carbon dioxide and water. This clean and complete combustion produces fewer particulates and nitrogen oxides than gasoline or diesel fuels, giving methanol an environmental advantage in contexts where emission control matters. The presence of the hydroxyl group alters ignition characteristics compared with hydrocarbons of similar molecular weight, making methanol easier to ignite under certain thermodynamic conditions while also allowing for high compression operation in specialized engines. Methanol has become a major alternative fuel option due to this energetic efficiency and relatively clean exhaust, supporting applications ranging from race car fuel to industrial burners and, in some regions, methanol-based transportation energy systems. The ability of methanol to blend with gasoline, to act as a base for biodiesel synthesis via transesterification, and to serve as a feedstock for hydrogen production in reforming systems demonstrates how its molecular composition supports both combustion-based and low-emission energy pathways.
Industrially, methanol is far more than a fuel. It is a fundamental building block for synthesizing higher chemicals. Through carefully controlled oxidation, methanol converts into formaldehyde, a compound used extensively to make plastics, resins, insulation foams, coatings, laminates, adhesives, and construction materials. This oxidation capability arises directly from the molecule’s hydroxyl-bearing structure, which allows oxygen insertion and stepwise dehydrogenation along the carbon backbone. Through catalytic conversion and dehydration, methanol can transform into dimethyl ether, a clean-burning propellant and diesel substitute, and through oligomerization it can serve as a precursor for light olefins that ultimately become polyethylene and polypropylene—two of the most widely used plastics in the world. Methanol-to-gasoline and methanol-to-olefin technologies highlight how the molecule acts not only as an end-use fuel but as a versatile carbon source for synthetic chemistry. Its reactivity stems from the fact that the hydroxyl group weakens the carbon–hydrogen bonds relative to hydrocarbons while still preserving structural stability, making methanol an ideal substrate for reaction pathways that require balanced reactivity rather than uncontrolled decomposition.
In environmental and atmospheric science, methanol occupies a distinctive role because its volatility and biological origin contribute to its global presence. Plants, algae, and soil microorganisms produce methanol naturally through the demethylation of pectin and other methylated biomolecules, meaning that methanol is continuously emitted into the air from forests, crops, and decaying vegetation. Oceans also release methanol into the atmosphere through biological cycling. Although far less discussed than greenhouse gases such as carbon dioxide or methane, methanol participates in atmospheric photochemical reactions that contribute to the formation of formaldehyde and ozone in the troposphere. These reactions underscore how even a simple alcohol can influence air quality and climate dynamics when present at sufficient concentrations. At the same time, bacteria known as methylotrophs consume methanol as a carbon and energy source, acting as a biological sink that returns atmospheric methanol to organic systems. The existence of these microorganisms demonstrates that methanol is not simply an industrial solvent but a metabolic substrate that supports ecological energy flow in certain ecosystems. The ability of methanol to serve simultaneously as a natural emission and a natural nutrient is directly linked to its molecular structure: small enough to volatilize, polar enough to dissolve in biological fluids, and energetic enough to support ATP production when metabolized through specialized biochemical pathways.
Despite methanol’s usefulness, its biological effects on humans demonstrate how structure determines toxicity. Because the molecule is small and polar, it is rapidly absorbed through ingestion, inhalation, or prolonged skin exposure. Once in the body, methanol undergoes metabolic oxidation in the liver, first to formaldehyde and then to formic acid. These metabolites—not the methanol itself—are responsible for poisoning symptoms. Formaldehyde damages proteins and cellular structures, while formic acid interferes with mitochondrial energy generation, especially in optic nerves and the central nervous system. This metabolic transformation is a direct consequence of the hydroxyl-bearing carbon structure of methanol, which can be enzymatically oxidized using the same alcohol dehydrogenase pathways that normally break down ethanol. Because formic acid accumulates faster than the body can neutralize it, methanol toxicity can lead to visual impairment, metabolic acidosis, neurological damage, and in severe cases death if not treated promptly. Ironically, ethanol serves as an antidote because the same enzymes preferentially metabolize ethanol over methanol, slowing the conversion of methanol into toxic intermediates and allowing time for its removal. This biochemical truth reinforces how sharply one additional carbon atom in ethanol—compared to methanol—completely changes physiological outcome, again proving that small structural changes lead to dramatic functional differences.
Methanol also plays an increasingly important role in the search for environmentally sustainable technologies. Industrial processes are exploring methanol as a carrier of renewable energy by producing it from captured carbon dioxide and green hydrogen generated through wind, solar, or hydroelectric electrolysis. In such systems, methanol acts as a liquid vector for transporting hydrogen energy safely and efficiently, because handling pure hydrogen in storage and long-distance transport remains technologically challenging. Methanol produced through carbon-neutral pathways can also be used to create synthetic fuels for aviation, shipping, and heavy industry, offering routes to reduce fossil carbon dependence. In this context, methanol is not prized only for its combustion value but also for the molecular convenience of a liquid fuel that can be derived from atmospheric carbon rather than fossil deposits. Once again, the molecule’s compact and reactive structure makes it well suited to industrial, catalytic, and electrochemical conversion cycles that help close carbon loops rather than open them.
Throughout all of these scientific, industrial, and environmental dimensions, the core identity of methanol remains tied to the arrangement of four hydrogen atoms, one carbon atom, and one oxygen atom into the simplest possible alcohol. The addition of a hydroxyl group to a methane backbone transforms the nature of the molecule and unlocks a sweeping array of interactions: it becomes polar enough to bond with water, energetic enough to burn cleanly, reactive enough to form complex derivatives, volatile enough to circulate in the atmosphere, and biologically consequential enough to operate simultaneously as a metabolic resource in nature and a toxic hazard in humans. The chemistry of methanol demonstrates a general truth that applies across molecular science—the composition of a substance describes only what atoms are present, but the structure of a substance determines what those atoms can do. Methanol continues to influence global energy policy, industrial chemistry, atmospheric processes, and biological safety not because of its simplicity but because its precise molecular geometry gives rise to behaviors that reverberate far beyond the scale of individual molecules.