Water Molecule — H₂O Chemical Structure
The water molecule stands as one of the most essential and remarkable structures in nature, both exceedingly simple in composition and profoundly complex in the roles it plays across chemical, biological, geological, and atmospheric systems. Although its familiar representation uses symbols to indicate its atoms, the true significance of water lies not in its shorthand notation but in the unique spatial arrangement, subtle intermolecular forces, and versatile interactions that arise from its structure. Understanding the nature of a water molecule means exploring its geometry, its polar character, its dynamic behaviour, and the ways these features influence the properties and processes that define life and the environment. Without relying on equations or symbolic expressions, this explanation builds a descriptive picture of why water behaves as it does and how its structure shapes almost every aspect of the natural world.
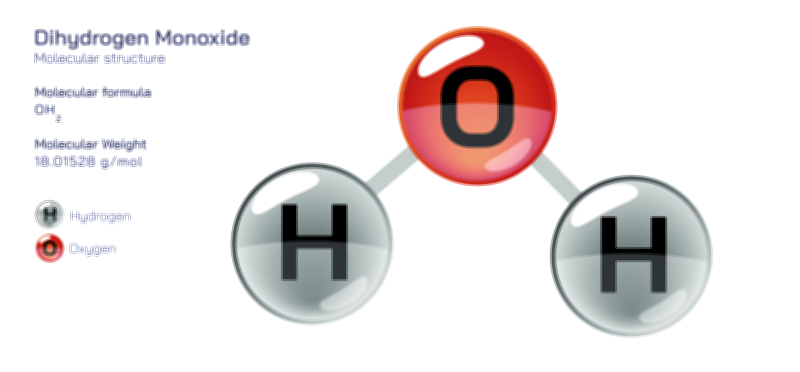
At the heart of water’s distinctiveness is the way its two hydrogen atoms and one oxygen atom are arranged. Rather than positioning its atoms in a straight line, the molecule forms an angular or bent shape. This shape results from the manner in which the oxygen atom’s electrons occupy space, creating regions that influence the positions of the hydrogen atoms. The outcome is a molecule that carries an uneven distribution of electrical charge: one side of the molecule becomes slightly more negative, while the opposite side becomes slightly more positive. This unevenness, known as polarity, gives the water molecule a character unlike many other small molecules. Through this polarity, water becomes capable of forming strong attractions with other water molecules as well as with many different types of substances.
These attractions, often described as hydrogen bonding, are not permanent structural links but rather constantly forming and breaking connections that tie water molecules together in a dynamic network. Each water molecule can attract several neighbours, creating a constantly shifting yet cohesive structure. This characteristic explains why water is both fluid and structurally organized at the same time. Even though the molecules remain mobile, they maintain enough attraction to give water a higher level of internal cohesion compared to many other liquids. This cohesion manifests in familiar ways such as the formation of droplets, the tendency of water to bead on smooth surfaces, and the appearance of surface tension that allows small insects to walk across ponds and enables water to rise through narrow tubes in plants.
Polarity also grants water extraordinary dissolving power. Many substances separate into their components when mixed with water because the charged regions of the water molecules interact with the charged or partially charged particles of the solute. These interactions pull the particles away from their solid structures, dispersing them throughout the liquid. This capacity to dissolve a vast range of substances qualifies water as a universal solvent and makes it indispensable for biological systems. Within living organisms, countless chemical processes depend on water’s ability to transport nutrients, distribute ions, and create environments in which reactions can occur smoothly. Cells rely on water not just as a medium but as an active participant in maintaining shape, regulating temperature, and facilitating molecular interactions essential for life.
The structure of water also influences its unusual thermal behaviour. Many liquids contract steadily when cooled, but water behaves differently due to the network of attractions among its molecules. As water cools toward its freezing point, its molecules arrange themselves into a more open and ordered pattern that occupies more space than the liquid state. This expansion causes ice to become less dense than liquid water, enabling it to float. This single property has profound implications for the Earth’s environment. Floating ice insulates the liquid water beneath it, protecting aquatic life during cold periods. It also influences global climate patterns, sea levels, and the reflective qualities of polar regions. Without this anomaly, the world’s oceans and lakes would behave very differently, altering the conditions necessary for sustaining ecosystems.
In addition to its thermal anomalies, water exhibits a high capacity to absorb and store heat. This arises from the energy required to disrupt the network of intermolecular attractions. Because so much energy is needed to raise its temperature, water warms and cools slowly relative to many other substances. This feature stabilizes Earth’s climate, moderates seasonal changes, and allows organisms with high water content to maintain stable internal temperatures. Coastal regions benefit from water’s thermal properties through more consistent weather patterns, while global ocean currents distribute heat across the planet, shaping the climate and making many regions habitable.
Water’s structure also plays a central role in shaping geological landscapes. As water seeps into cracks in rocks and subsequently freezes, its expansion exerts force, gradually widening the fractures in a process known as frost weathering. Over large timescales, this contributes to the breakdown of mountains, the formation of soil, and the sculpting of valleys. Water also acts as a transport medium that carries sediments, erodes coastlines, carves river channels, and deposits minerals in new locations. All these geological transformations stem from water’s ability to shift between solid, liquid, and gaseous forms, each influenced by the multi-faceted interactions arising from its molecular structure.
In the atmosphere, water’s behaviour is equally vital. When water evaporates, it absorbs heat from its surroundings, creating a cooling effect. When it condenses, it releases heat, driving atmospheric circulation and powering weather systems. Clouds form because tiny droplets or ice crystals suspended in the air reflect and scatter sunlight, moderating temperature and influencing climate. The formation of rain, snow, hail, and fog all depend on the delicate balance between temperature, pressure, and the structure-driven interactions of water molecules. The global water cycle, which circulates water through evaporation, condensation, precipitation, and movement through land and oceans, is powered entirely by the unique characteristics of the water molecule.
In living organisms, water participates directly in chemical reactions essential for life. Many organic molecules interact with water in selective ways, either binding to it, repelling it, or forming complex structural arrangements influenced by its presence. Proteins, for example, fold into their functional shapes largely because of how their components respond to water, arranging themselves so that water-attracting parts face outward while water-repelling parts turn inward. DNA maintains its stable double-helix structure through interactions with surrounding water molecules. Cellular membranes form because of the opposing ways different molecular groups respond to water. These biological architectures could not exist without the polarity and structural behaviour of water.
Water also serves as a medium for transport inside organisms. Blood, plant sap, digestive fluids, and cellular cytoplasm rely on water to carry nutrients, hormones, waste products, and gases. The movement of water across membranes maintains pressure gradients that support functions such as nutrient absorption, waste removal, and cellular stability. Even the sensation of thirst reflects the body’s dependence on water to sustain these delicate fluid balances. Life emerged in water and continues to depend on the processes shaped by water’s structure at every scale.
Beyond natural systems, human technology takes advantage of water’s structural properties in numerous ways. It is used as a coolant in industrial processes because it can absorb large amounts of heat. It is employed as a cleaning agent because its polarity allows it to dissolve many contaminants. It is essential in food preparation, agriculture, manufacturing, and medicine. The simplicity of its composition belies the immense range of functions it enables in modern society. Every one of these uses, from generating steam in power plants to sustaining crops in fields, traces its effectiveness back to the interactions dictated by the molecule’s structure.
Ultimately, the water molecule represents an elegant interplay of geometry, polarity, and intermolecular behaviour. These features combine to create a liquid that is cohesive yet fluid, structured yet adaptable, simple yet indispensable. Its properties make Earth habitable, life possible, ecosystems stable, and human civilization sustainable. Although its chemical representation is familiar and straightforward, its structural subtleties influence the entire natural world in profound ways. By understanding the water molecule’s unique character in descriptive terms, it becomes clear that its significance reaches far beyond its simplicity, shaping the physical, biological, and environmental systems that define life on our planet.