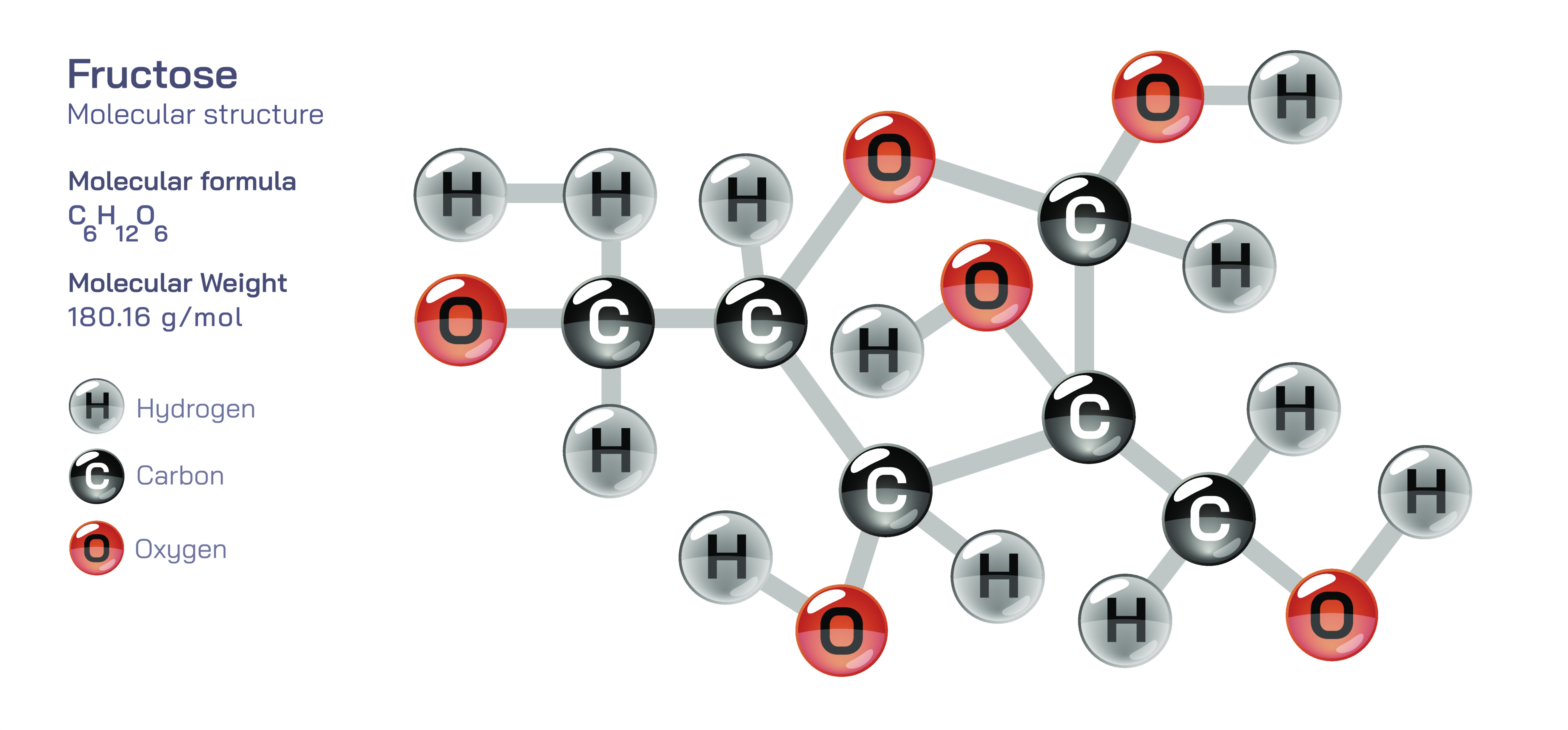
Fructose — Molecular Structure, Chemical Properties, and Natural Sources Explained
Fructose is one of the most important naturally occurring sugars on Earth, and even though its molecular structure is often represented through simple diagrams showing carbon chains, ring formations, or functional groups, its true significance becomes clear only through a deep exploration of its chemical identity, its biochemical behavior, and the way it exists and acts within living organisms and natural environments. Fructose is known primarily as a simple sugar, yet its structure, properties, and sources reveal a far richer story about the fundamental chemistry of carbohydrates, the subtle differences between sugar types, the intricate metabolic pathways in living systems, and the diverse roles that these molecules play in foods, plants, and biological functions. By examining fructose through a detailed conceptual lens, one gains insight not only into what the molecule looks like but also how it behaves, why it tastes the way it does, how it fuels the body, and why it appears broadly across fruits, honey, and many natural sweetening sources.
Fructose is classified as a monosaccharide, which means it is one of the simplest carbohydrate molecules that cannot be broken down into smaller sugar units through hydrolysis. What makes fructose particularly interesting among monosaccharides is that it belongs to the family of ketoses rather than aldoses. Whereas glucose, a close structural relative, contains an aldehyde functional group, fructose contains a ketone functional group positioned within its carbon chain. This difference may seem small, but it shapes nearly every chemical property of fructose, influencing the way it forms rings, the way it interacts with other biomolecules, the way it participates in chemical reactions, and the way it is metabolized within living organisms. In its linear form, fructose consists of six carbon atoms, making it a hexose, and each carbon is bonded to specific arrangements of hydrogen and oxygen that create a highly polar and highly functional molecule. The presence of the ketone group at the second carbon distinguishes fructose from glucose, whose aldehyde group appears at the first carbon in its linear arrangement.
Although fructose can exist in a straight-chain form, it commonly appears in nature and biological systems in a cyclic configuration because the molecule naturally folds and forms internal bonds that create ring structures. In aqueous environments, the hydroxyl group on one carbon reacts with the carbonyl carbon of the ketone, leading to the formation of a ring. Unlike glucose, which forms a six-membered ring more often, fructose tends to form five-membered rings known as furanose forms, although six-membered pyranose forms can also exist under certain conditions. This ability to shift between forms gives fructose flexibility in biochemical interactions and affects its sweetness, reactivity, and role in various metabolic pathways. The ring structure is stabilized by intramolecular hydrogen bonding, contributing to the molecule’s stability in physiological environments.
One of the defining characteristics of fructose is its exceptional sweetness. Among naturally occurring sugars, fructose is often considered one of the sweetest, significantly sweeter than glucose and sucrose under most conditions. Its sweetness perception arises not solely from its chemical composition but also from the specific arrangement of atoms within its ring forms that interact uniquely with taste receptors on the human tongue. These molecular arrangements trigger sensory responses more intensely than many other sugars, making fructose particularly valued in foods and natural sources. Interestingly, sweetness perception varies depending on whether fructose is in a warm or cool environment, with colder temperatures enhancing its perceived sweetness more than warmer ones. This relationship reflects how structural changes in sugar molecules alter sensory interactions.
Fructose exhibits several unique chemical properties that distinguish it from other sugars. The ketone group allows fructose to participate in a wide range of reactions, including isomerization, dehydration, reduction, and polymerization. Because fructose does not contain an aldehyde group in its open-chain form, it behaves differently in certain reactions compared with glucose. Yet fructose can undergo transformations within physiological systems that convert it into other sugars or intermediates. For example, it can be rearranged into glucose or mannose through enzymatic processes, demonstrating how flexible carbohydrate chemistry is at the molecular level. Its functional groups also allow it to react in Maillard browning reactions, contributing to the flavor and color changes that occur when foods containing proteins and sugars are heated. Although glucose participates in these reactions as well, fructose tends to react more rapidly due to its structural features, influencing the flavor profiles of baked goods, dried fruits, and cooked syrups.
The solubility of fructose is another notable property. It dissolves readily in water because of its numerous hydroxyl groups, all of which form hydrogen bonds with water molecules. This high solubility makes fructose easy for plants and organisms to transport within their tissues. It is also hygroscopic, meaning that it readily attracts and retains moisture, a characteristic that affects food texture, preservation, and stability. Foods high in fructose often stay moist longer and resist crystallization, making fructose-rich sweeteners desirable in products that require softness and long shelf life. For example, honey remains liquid and smooth partly due to its high fructose content, as fructose inhibits crystallization more effectively than glucose.
Metabolically, fructose follows a unique pathway in the human body. While glucose is absorbed and utilized by nearly every cell and is tightly regulated by insulin, fructose is handled differently. After being absorbed from the digestive tract, most fructose travels directly to the liver, where it undergoes a series of metabolic steps that convert it into intermediates used for energy storage or energy production. Unlike glucose, fructose does not trigger a strong insulin response, and the liver processes it largely independently of insulin control. Once inside liver cells, fructose is quickly phosphorylated and enters pathways that may lead toward glycogen formation, energy production, or the synthesis of lipids under certain conditions. This metabolic uniqueness has prompted scientific discussions about fructose consumption, energy balance, and health effects, but the deeper biochemical truth remains that its behavior is shaped entirely by its molecular architecture and how enzymes recognize, bind, and transform sugar molecules.
In the natural world, fructose is abundant and widespread. Fruits such as apples, grapes, mangoes, pears, and berries contain high levels of free fructose, often accompanied by glucose and sucrose. These sugars contribute to the sweetness, flavor, and energy content of fruit, playing a central role in plant physiology and reproductive strategies. Plants produce fructose primarily through photosynthesis, where complex carbohydrate molecules are synthesized and later broken down into simpler sugars as needed. The presence of fructose helps attract animals that feed on fruit and disperse seeds, aiding plant reproduction. Many vegetables—such as carrots, onions, and sweet corn—also contain fructose, though typically in smaller amounts. Honey is another major natural source, created by bees through the enzymatic transformation of nectar. Its high fructose content gives honey its characteristic sweetness and resistance to crystallization.
Fructose is also present in natural plant syrups such as maple syrup, agave syrup, and certain tree saps. In these fluids, fructose exists either freely or as part of sucrose, which is a disaccharide made of one glucose molecule and one fructose molecule linked together. When sucrose is broken down, fructose is released as a free monosaccharide. This breakdown process occurs both in plant tissues and within the human digestive system, where enzymes cleave sucrose into its component sugars.
Beyond natural sources, fructose appears in various manufactured products that use sweeteners such as high-fructose corn syrup. Although the details of production differ from natural biochemical synthesis, the molecular identity of the fructose produced remains the same. High-fructose corn syrup consists of mixtures of glucose and fructose, created by enzymatically converting a portion of glucose in corn syrup into fructose. The sweetness, solubility, and functional properties of fructose make such sweeteners useful in beverages, baked goods, sauces, processed foods, and confectionery applications.
A deeper understanding of fructose’s chemical properties also reveals why it behaves differently from glucose in food science. Because fructose has a lower tendency to crystallize, it helps create smooth textures in foods such as soft drinks, jams, and frozen desserts. Its ability to retain moisture contributes to extended freshness in baked products and chewy candies. In caramelization and browning reactions, fructose tends to produce deeper color and richer flavor at lower temperatures, influencing culinary outcomes. These behaviors are direct consequences of how the functional groups in fructose interact with heat, other molecules, and environmental conditions.
Fructose’s presence in biological systems goes beyond nutrition and energy. Plants rely on sugars like fructose for osmotic balance, signaling, and metabolic flexibility. Fructose participates in pathways that regulate growth, flowering, and stress responses. In plant tissues, fructose can bond temporarily with other molecules or enter cycles that generate essential metabolic intermediates. Within fruits, fructose accumulates as part of ripening processes driven by enzymatic changes in carbohydrate metabolism. The rising fructose content of ripening fruit contributes to sweetness perception, attracting animals that aid in seed dispersal.
Structurally, fructose’s unique arrangement of atoms gives it an identity that biochemists can recognize instantly. Each carbon is part of a carefully balanced pattern of hydroxyl groups, hydrogen atoms, and carbonyl placement. These groups allow fructose to form glycosidic bonds with other sugars, enabling the construction of more complex carbohydrates. Through these linkages, fructose becomes part of oligosaccharides and polysaccharides found in nature, expanding its role beyond its identity as a single simple sugar.
Ultimately, fructose is far more than a sweet-tasting molecule found in fruits and honey. Its structure reflects the fundamental principles of carbohydrate chemistry, its properties influence food science and sensory perception, its metabolism illustrates the complexity of biochemical pathways, and its natural occurrence reveals the deep connections between plant biology and ecological interactions. Through a detailed conceptual understanding of fructose—its molecular architecture, chemical behavior, and origins in nature—it becomes possible to appreciate the molecule not just as a dietary component but as a fundamental participant in the chemistry of life, energy, and biological diversity.