Molecular Identity and Structural Foundation of Acetaldehyde
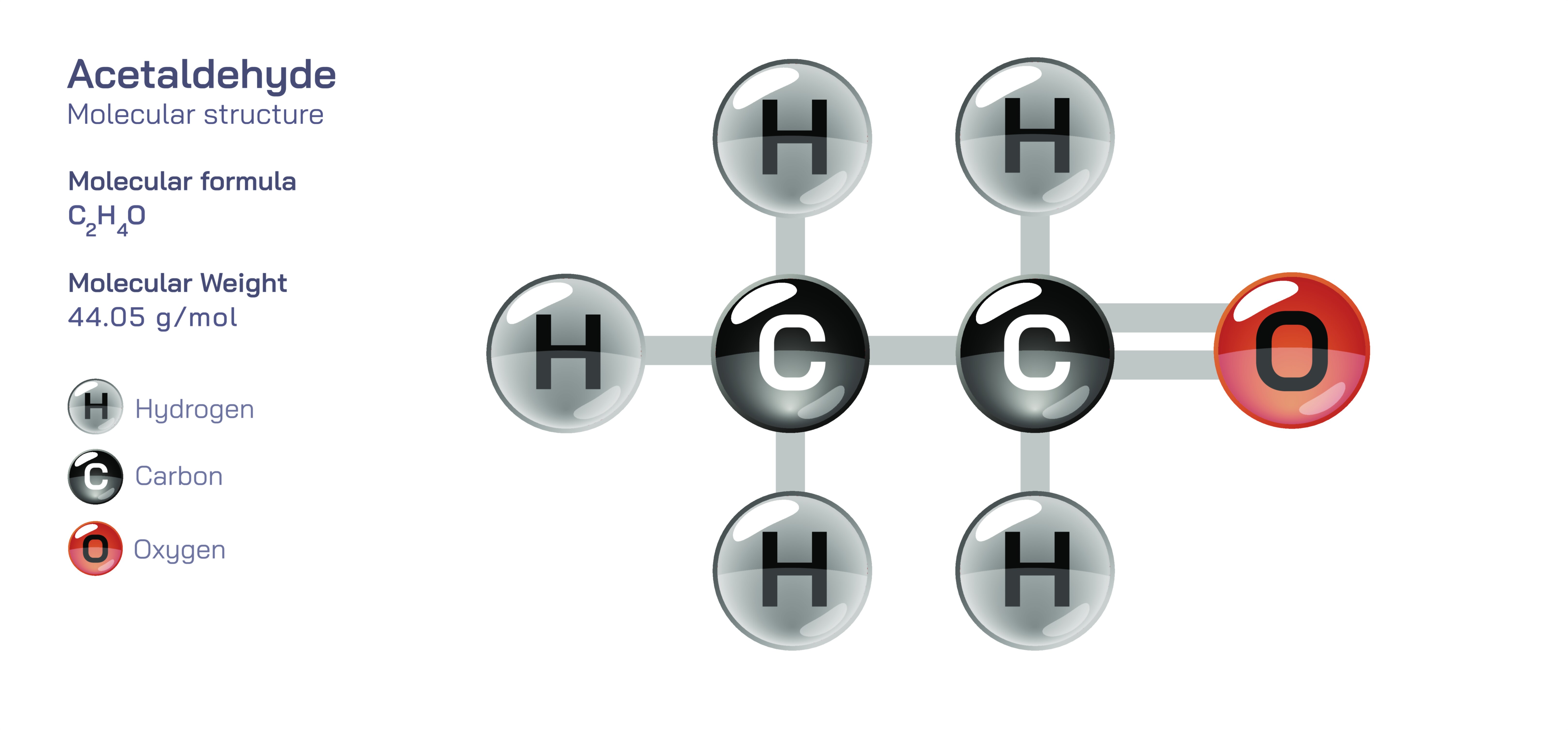
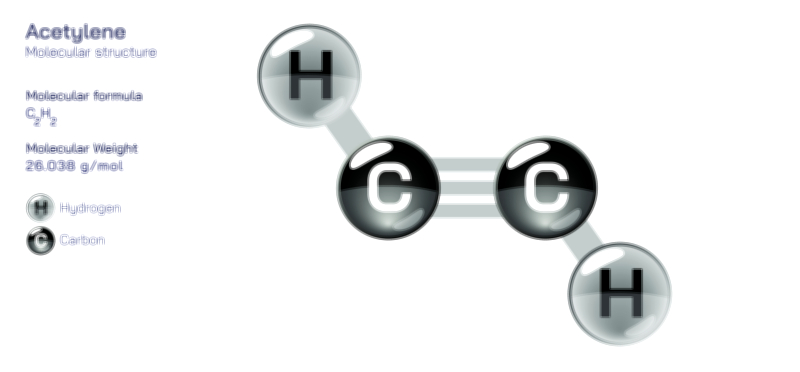
Acetaldehyde is one of the simplest yet most chemically influential aldehydes, represented by the formula CH₃CHO. Its compact structure conceals its significant role across biological, industrial, and environmental systems. The molecule consists of a carbonyl group attached to a methyl group, forming a structure that is both highly reactive and incredibly versatile. The defining feature of acetaldehyde is its aldehydic carbonyl, where a carbon atom is double-bonded to oxygen and single-bonded to hydrogen, creating a reactive functional group central to the molecule’s behavior. This configuration generates a unique combination of polarity, electrophilicity, and chemical instability, making acetaldehyde an essential intermediate in countless metabolic and synthetic pathways. Even though it is small in size, its influence extends across atmospheric chemistry, industrial synthesis of solvents and plastics, and biological oxidation processes involving alcohol metabolism. Understanding its molecular structure provides a window into numerous reactions where acetaldehyde serves as a transitional yet essential chemical entity.
Chemical Structure and Electronic Characteristics
The chemical structure of acetaldehyde creates an electron distribution that strongly influences its reactivity. The carbonyl group, being highly polar, places a significant partial positive charge on the carbonyl carbon and a partial negative charge on the oxygen atom. This polarized bond becomes the focal point for nucleophilic attack in many chemical reactions. The methyl group attached to the carbonyl carbon also contributes slightly to the molecule’s stability, but not enough to diminish its overall reactivity. The double-bond character between carbon and oxygen forms the basis of aldehyde functionality, giving acetaldehyde the ability to participate in oxidation, reduction, addition, condensation, and polymerization reactions. Unlike larger aldehydes that are more resistant to oxidation or spontaneous reactions, acetaldehyde remains extremely susceptible to transformation. This susceptibility comes from the ease with which its carbonyl can undergo nucleophilic additions or be converted to other compounds such as acetic acid, ethanol, or larger organic molecules. Its structure can also lead to rapid polymerization under certain conditions, forming paraldehyde or metaldehyde, which further highlights its structural responsiveness.
Physical Properties and Their Chemical Implications
The physical properties of acetaldehyde reflect its small molecular size and high reactivity. It exists at room temperature as a volatile, colorless liquid with a sharp, pungent odor often associated with cut fruit, fermentation processes, or certain industrial environments. Its low boiling point allows it to evaporate quickly, enabling it to disperse in air and participate in atmospheric reactions that influence pollution chemistry. Its solubility in water arises from its polar carbonyl group, which can interact with water molecules through hydrogen bonding and dipole interactions. However, its solubility is not as high as simpler alcohols or acids, reflecting the balance between its hydrophobic methyl group and hydrophilic carbonyl. Acetaldehyde's physical properties also make it highly flammable and capable of forming explosive mixtures with air. These characteristics underscore the importance of handling it with caution in industrial settings. In biological contexts, its volatility and reactivity are crucial because acetaldehyde is produced inside the human body during the metabolism of alcohol, contributing to unpleasant physiological effects such as headaches, nausea, and rapid heartbeat.
Reactivity and Transformation Pathways
Acetaldehyde’s chemical properties allow it to participate in numerous reactions that shape its importance across various sciences. In oxidation reactions, it readily converts into acetic acid, a transformation that mirrors metabolic pathways in living organisms. When reduced, it converts to ethanol, demonstrating reversible interconversion between key organic functional groups. This versatility is the cornerstone of its role as a metabolic intermediate. In addition to redox reactions, acetaldehyde participates in nucleophilic addition reactions due to the electrophilic nature of its carbonyl carbon. Alcohols, amines, and hydrogen cyanide can all add to acetaldehyde, forming hemiacetals, imines, or cyanohydrins respectively. The molecule is also prone to aldol condensation, where two acetaldehyde molecules combine to produce larger compounds such as crotonaldehyde. This process forms the basis for synthetic pathways in organic chemistry. Another important behavior of acetaldehyde involves polymerization, where under acidic conditions it can transform into cyclic trimers like paraldehyde or more complex structures like metaldehyde. These products have historical pharmaceutical and industrial uses, demonstrating how acetaldehyde’s reactivity generates compounds with broader applications.
Biological and Environmental Significance
In biological contexts, acetaldehyde plays a critical role in the metabolism of ethanol. Once alcohol enters the human body, enzymes rapidly convert it to acetaldehyde, which is substantially more toxic than ethanol itself. Elevated levels of acetaldehyde are responsible for many acute symptoms associated with alcohol consumption, including facial flushing, headaches, and overall discomfort commonly known as the hangover effect. The body then converts acetaldehyde into acetic acid, but this step can be inefficient in individuals with genetic variations affecting enzymes such as aldehyde dehydrogenase. Beyond human metabolism, acetaldehyde also forms naturally in fruits, plants, and fermenting substances, contributing to distinctive aromas and flavors. In environmental chemistry, acetaldehyde participates in atmospheric reactions that lead to smog formation, making it an important compound in discussions involving air quality and pollutant formation. Industrially, its role as a precursor to acetic acid, solvents, perfumes, and synthetic materials highlights how such a small molecule can influence large-scale chemical manufacturing.
Illustrating Acetaldehyde’s Structure and Behavior
Scientific illustrations of acetaldehyde commonly emphasize its carbonyl group, showing the oxygen with lone electron pairs and the carbon atom as a site of electrophilic activity. These illustrations help visualize how the molecule interacts with nucleophiles, participates in redox transformations, or undergoes polymerization. By depicting how its atoms align, how charge distribution flows across the molecule, and how chemical bonds shift during reactions, such illustrations clarify the underlying principles that govern acetaldehyde’s chemical behavior. They make it easier to understand how a small and simple molecule can be so reactive, so biologically consequential, and so important in environmental and industrial chemistry.