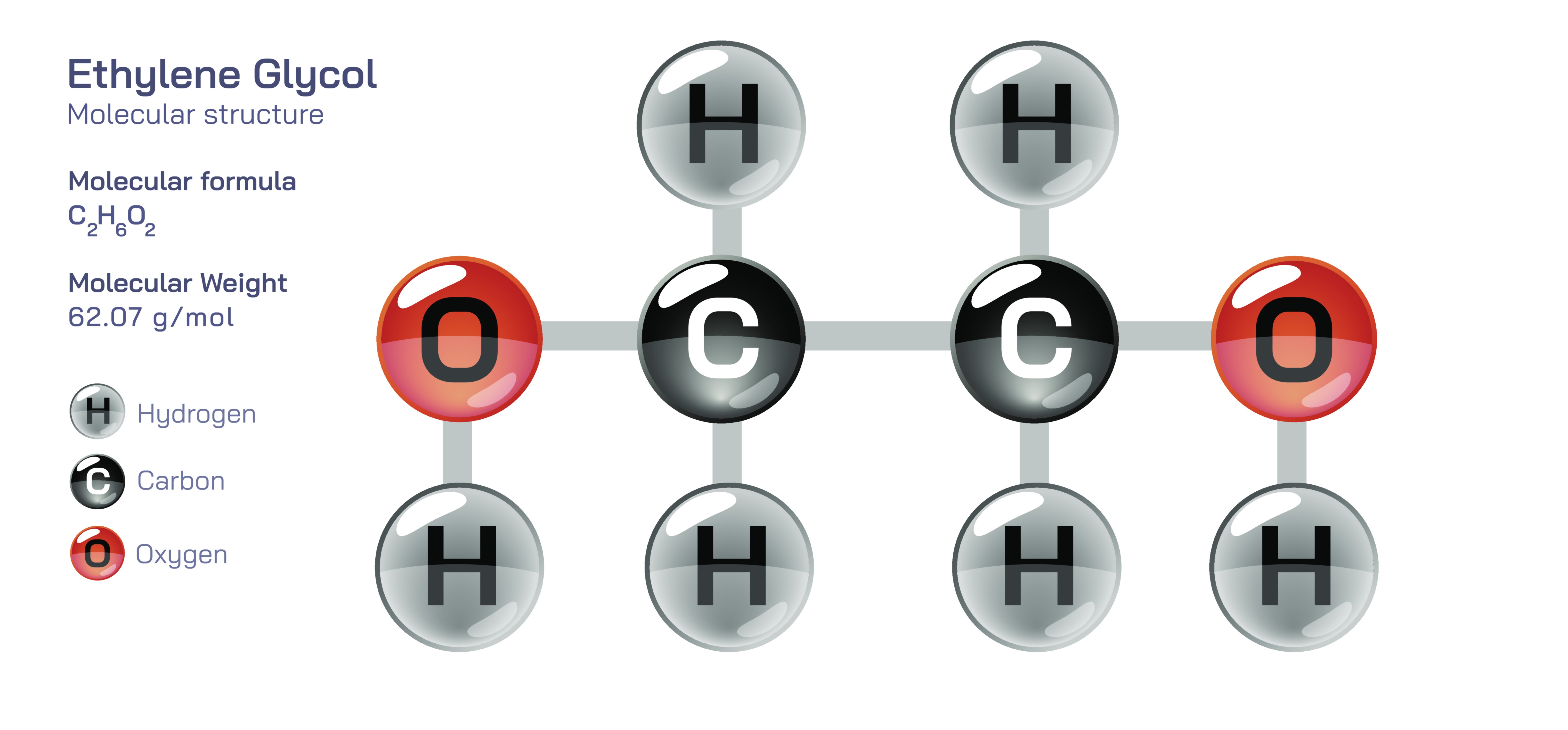
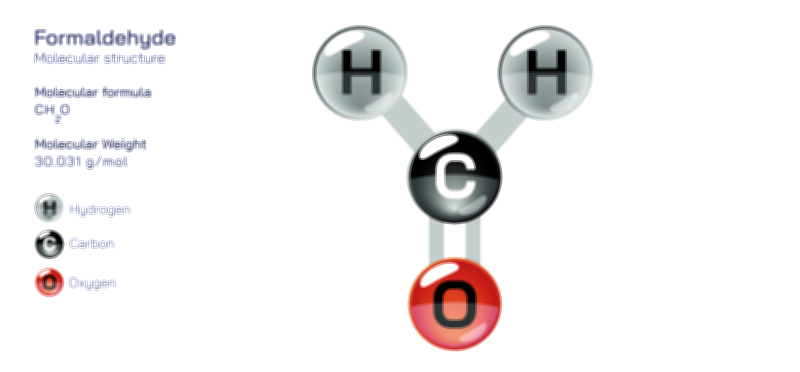
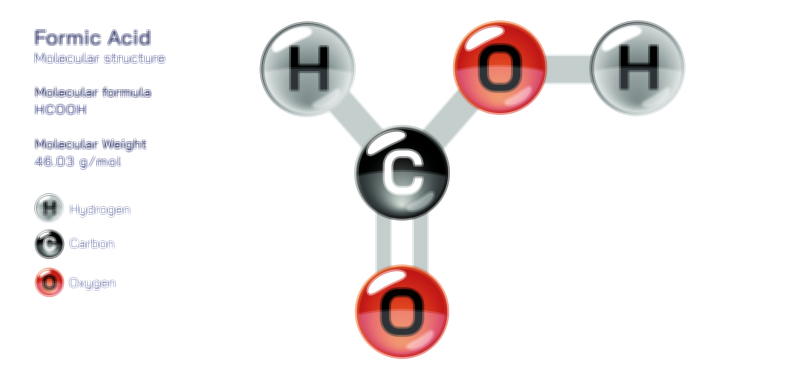
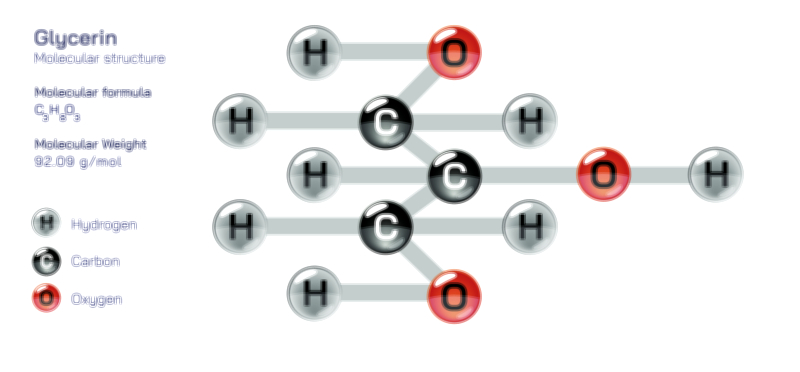
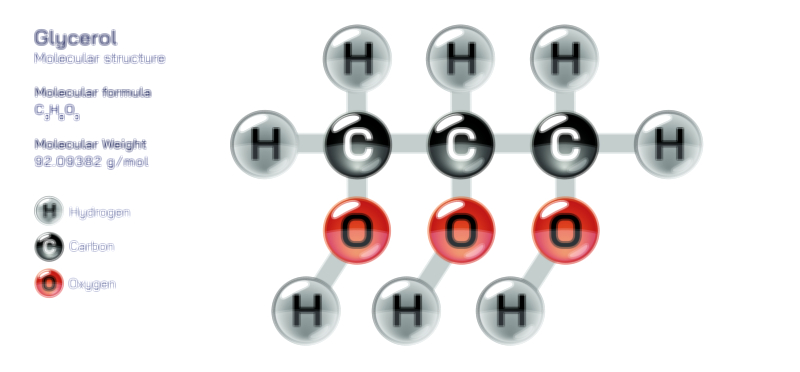
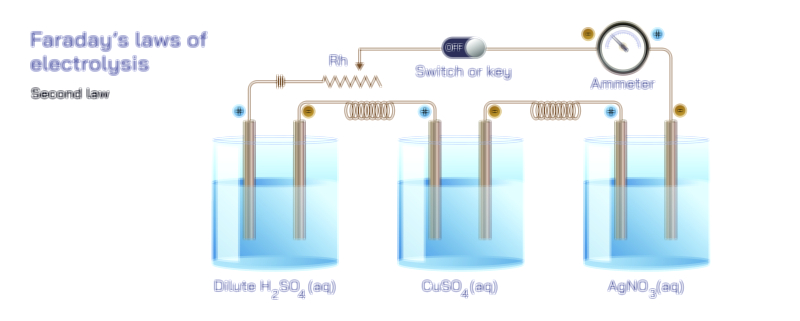
Ethylene Glycol Chemical Structure — Organic Compound Diagram Explained
Ethylene glycol stands as one of the most widely used and chemically intriguing organic compounds, known not only for its practical applications in antifreeze, coolants, polymer production, and industrial chemistry, but also for the structural lessons it provides within the field of organic molecules. Its structure is simple at first glance yet remarkably influential, because it contains two hydroxyl functional groups attached to a two-carbon backbone. This arrangement transforms it from an ordinary small organic molecule into a highly polar, strongly hydrogen-bonding, and chemically versatile diol. Understanding the structure of ethylene glycol in a detailed, descriptive manner reveals how its molecular architecture gives rise to its physical properties, its reactivity, its roles in biological and environmental systems, and its significance in polymer chemistry. A structural diagram or vector illustration typically depicts it with clarity—two carbons connected in sequence, each bearing a hydroxyl group—but beneath this simplicity lies a wealth of molecular insight that can be fully appreciated only through careful explanation.
Ethylene glycol has the molecular formula C₂H₆O₂, and structurally it is written as HO–CH₂–CH₂–OH. This configuration places two hydroxyl groups on adjacent carbon atoms, making it the simplest diol in organic chemistry. Each carbon atom in the molecule adopts a tetrahedral geometry, bonded to two hydrogen atoms, one carbon atom, and one oxygen atom of a hydroxyl group. The tetrahedral structure is important because it determines the three-dimensional orientation of the molecule, influences its intermolecular interactions, and contributes to its distinctive physical properties. While the two-dimensional vector diagrams represent ethylene glycol with straight lines connecting atoms, the true spatial arrangement is three-dimensional, with bond angles and orientations shaped by electron pair repulsions and the hybridization states of the atoms involved.
The presence of two hydroxyl groups dramatically affects the polarity of ethylene glycol. Oxygen atoms carry partial negative charges due to their high electronegativity, while the hydrogen atoms attached to them carry partial positive charges. This uneven distribution of electron density gives the molecule strong dipole moments at two points, making the entire compound highly polar. Because polarity dictates how molecules interact with one another and with surrounding environments, the structural polarity of ethylene glycol is the driving force behind its miscibility with water, its ability to dissolve both polar and some slightly nonpolar substances, and its high affinity for forming intermolecular hydrogen bonds. In fact, the molecule is capable of forming extensive hydrogen-bonding networks due to the presence of two sites where hydrogen bonds can form both as donors and acceptors. These networks create cohesive forces that elevate the boiling point of ethylene glycol far above what one would expect for a molecule of its size, while also preventing it from crystallizing easily, which explains its usefulness in antifreeze formulations.
Ethylene glycol’s structure further illustrates how the number and placement of functional groups influence reactivity. Each hydroxyl group is capable of participating in typical alcohol reactions such as oxidation, esterification, and substitution, yet the proximity of the two groups introduces unique behavior not seen in monohydric alcohols like ethanol. The molecule can undergo intramolecular interactions, form cyclic intermediates in certain chemical reactions, and yield diesters when reacted with acids. When oxidized under controlled conditions, ethylene glycol can produce compounds such as glycoaldehyde, glycolic acid, or oxalic acid, illustrating how the arrangement of atoms influences the pathways by which chemical transformations proceed. This dual functional nature also allows the molecule to serve as a building block for polyesters. When ethylene glycol reacts with dicarboxylic acids such as terephthalic acid, long polymer chains form through repeated ester linkages, resulting in polyethylene terephthalate (PET), one of the most widely used plastics in textiles, packaging, and industrial materials. The ability of ethylene glycol to participate in polymerization reactions arises directly from its structural design.
The two hydrophilic hydroxyl groups also impart hygroscopic behavior to ethylene glycol. This means the molecule readily absorbs moisture from the environment, a property that emerges from the strong attraction between the hydroxyl groups and water molecules. Hygroscopicity plays a significant role in its industrial uses, especially in formulations requiring water control or moisture stabilization. It also helps explain why ethylene glycol must be handled with care—it can pull water into systems where it may not be desired and can influence materials through absorption-related swelling or softening. This affinity for water demonstrates again how the structural features of the molecule dictate its macroscopic behavior.
Vector illustrations of ethylene glycol often highlight the linearity of its carbon chain, but this can lead to the assumption that the molecule is rigid. In reality, the central carbon–carbon bond is a sigma bond that allows free rotation, giving the molecule the flexibility to adopt numerous conformations. These conformations influence how the molecule interacts in various environments. For example, in aqueous solutions, ethylene glycol can rotate to maximize hydrogen bonding interactions with water molecules, creating highly stable solvation structures that contribute to its exceptional miscibility. This rotational freedom also affects the molecule’s behavior in polymerization processes and in the formation of crystalline or amorphous structures when incorporated into larger chemical networks. The conformational variability illustrates how flexible and dynamic even small molecules can be, and how these motions play a role in chemical and physical properties.
When considering the behavior of ethylene glycol in solution, its structure again proves crucial. Because its two hydroxyl groups can each form hydrogen bonds, the molecule strongly disrupts the ability of water to form its usual crystalline lattice when cooled. Instead of forming an ordered solid structure, the water molecules are held in disorganized arrangements, lowering the freezing point. This freezing-point depression is the foundational principle behind antifreeze, de-icing solutions, and heat-transfer fluids used in automotive and industrial systems. Ethylene glycol’s structure is therefore directly responsible for its function in preventing ice formation and ensuring fluid stability in cold environments. At the same time, the molecule raises the boiling point of water when mixed with it, enabling it to serve as a heat-transfer medium that withstands high temperatures without vaporizing easily. This dual effect—lowering freezing point while raising boiling point—comes entirely from the interactions dictated by the molecule’s structural features.
The structure of ethylene glycol also explains its density, viscosity, and physical texture. Hydrogen bonding creates strong intermolecular attractions that impede flow, giving ethylene glycol a noticeably thick, syrup-like consistency. Its density is higher than many hydrocarbons of similar size because of the presence of oxygen atoms, which increase molecular mass while maintaining a compact structure. The molecule therefore demonstrates how functional group composition influences macroscopic behavior in ways that can be directly felt or measured.
In biological and environmental contexts, ethylene glycol’s structure determines its interactions with living systems. Its polarity allows it to enter cells and tissues easily, and its small size permits rapid absorption. However, the molecule’s hydroxyl groups make it susceptible to specific metabolic pathways that convert it into acidic and harmful byproducts, which is why ingestion of ethylene glycol is toxic. The same structural characteristics that make it chemically versatile also enable it to participate in biological reactions that generate metabolites such as glycolic acid and oxalic acid, which can cause organ damage. Understanding the structure, therefore, becomes essential not only for appreciating its chemical utility but also for recognizing the risks associated with exposure.
In industrial chemistry and materials science, ethylene glycol’s structure serves as a foundation for numerous processes. Its dual hydroxyl groups make it a valuable reagent in polymer synthesis, acting as a monomer that provides flexibility and hydrophilicity to polymer chains. In textile production, its polymeric forms create fibers that balance strength, durability, and moisture control. In resins and coatings, the molecule’s structure enables cross-linking and contributes to the flexibility and clarity of final products. Each of these applications arises from the chemical behavior dictated by the arrangement of atoms within the ethylene glycol molecule.
Even though ethylene glycol is represented simply in vector illustrations, these diagrams conceal the underlying complexity of its molecular identity. Every bond angle, every electron distribution, and every functional group contributes to a web of behavior that defines its role in chemistry, industry, biology, and the environment. The molecule is an example of how structural simplicity can lead to functional richness, embodying principles of polarity, hydrogen bonding, molecular flexibility, and reactive versatility. Through these principles, ethylene glycol demonstrates how the architecture of atoms and bonds gives rise to extensive real-world applications and profound chemical significance.
Ultimately, the structure of ethylene glycol serves as a powerful illustration of the connection between molecular design and chemical behavior. It shows how functional groups shape interactions, how geometry influences physical properties, and how small organic compounds can hold tremendous technological and scientific value. Whether depicted through a clean vector illustration or explored through three-dimensional molecular modeling, ethylene glycol remains a fundamental and revealing example of how structural chemistry forms the basis for countless practical uses and deeper scientific understanding.