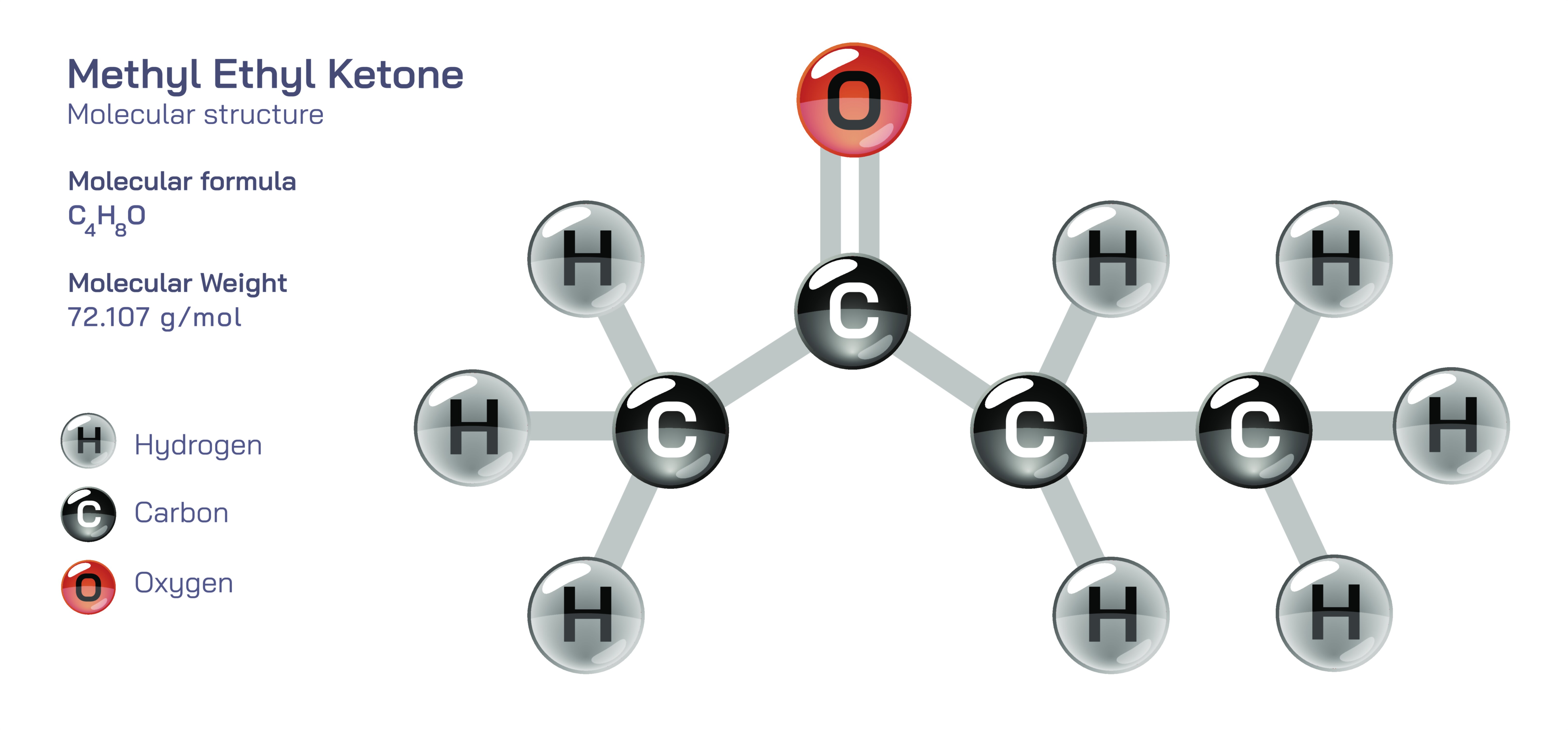
Methyl Ethyl Ketone — Molecular Structure and Composition
Methyl ethyl ketone is an organic compound whose molecular structure places it at the center of modern industrial chemistry, coating technology, polymer science, adhesive formulation, cleaning systems, and synthetic processes. Although often introduced by its common abbreviation MEK, the compound is fully known as methyl ethyl ketone and belongs to the ketone functional group family, a classification that arises because its carbon framework contains a central carbonyl functionality bonded to two separate alkyl chains. This defining structural feature establishes MEK as both highly versatile and comparatively stable under controlled reaction conditions, allowing it to serve as a solvent powerful enough to dissolve difficult polymer systems yet chemically moderate enough to avoid rapid breakdown, premature polymerization, or uncontrolled reactivity during manufacturing. The molecular architecture is compact and linear rather than bulky or symmetrical, and this structural simplicity plays a major role in determining the compound’s physical behavior. It evaporates quickly, diffuses efficiently, and forms predictable interactions with organic coatings, resins, and adhesives, making it one of the most relied-upon industrial solvents of the last century.
The core of MEK’s identity is the carbonyl group that gives ketones their defining properties. This functional region creates a polarity gradient across the molecule, meaning that while the compound contains a large nonpolar hydrocarbon region that favors interactions with organic materials, it also possesses a strongly polar region in which electron density is unequally shared. As a consequence, the molecule can dissolve both polar and nonpolar substances to a useful degree, a capability that many specialty solvents are unable to replicate. This duality is why MEK is integral to the formulation of paints, varnishes, printing inks, resins, lacquers, synthetic leathers, acrylic coatings, and high-performance adhesives. When MEK molecules interact with polymer chains, they loosen intermolecular attraction enough to allow coating resins to flow during application; then, due to rapid evaporation, the polymer chains reestablish association to form a smooth, uniform film. The subtle polarity of the carbonyl region is also responsible for the compound’s strong cleaning power on dried resins, uncured adhesives, and grease residues that resist ordinary solvents.
The physical behavior of MEK is inseparable from its structural composition because the size of the hydrocarbon backbone and the position of the carbonyl group determine its volatility and fluidity. It boils at a temperature that is low enough to ensure fast drying but high enough to allow industrial users to control evaporation rate by blending it with slower or faster co-solvents. This controllability is one of the reasons why MEK is a staple in engineered solvent systems where evaporation and application time must be balanced precisely, such as automotive refinishing, aerospace coatings, electrical insulation varnishes, waterproofing membranes, and flooring adhesives. Unlike solvents that leave sticky residues or cease dissolving once a formulation reaches a certain viscosity, MEK maintains its solvency power over the entire evaporation period, a trait that stems from the consistent strength of intermolecular forces across its fluid and vapor states. Even in demanding manufacturing settings, the structural characteristics of MEK ensure that the transition from liquid to vapor does not alter the positioning of polymer chains in unpredictable ways.
Another vital dimension of methyl ethyl ketone is its role in synthetic and chemical manufacturing. Its molecular design allows it to act not only as a solvent but also as a reactant, intermediate, and carrier in complex reaction networks. Chemical producers frequently employ MEK as a starting point in pathways that produce coatings resins, rubber additives, specialty lubricants, industrial catalysts, polymer modifiers, and fine chemical ingredients used in electronics, pharmaceutical formulation, and agricultural formulations. The ability of MEK to undergo selective oxidation, catalytic condensation, and polymer embedding reflects the reactivity that originates from the carbonyl center. This reactive precision, however, is balanced by structural stability, which means MEK does not degrade prematurely during storage or handling, nor does it aggressively attack plastic containers, seals, or gaskets the way some chlorinated solvents do. As chemistry has shifted toward greener and more sustainable practices, MEK’s compatibility with recycling programs, solvent recovery systems, and low-waste production lines has increased its long-term value in industrial supply chains.
In surface-coating science, the unique combination of solvency strength and controlled volatility makes MEK one of the best tools for engineering texture, gloss, leveling, and adhesion strength. Formulators carefully adjust its concentration to influence drying time, film coalescence, flow leveling, viscosity, and resistance to blistering or pinhole formation. MEK interacts with polymers at the molecular scale, softening resin structures just enough to allow reorganization during application. As the solvent evaporates, the matrix solidifies into a strong and durable layer. This evaporative profile is critical in high-speed manufacturing environments such as coil coating, metal finishing, graphic printing, composite layup, and electronics encapsulation. In such operations, productivity depends on maintaining a solvent that dries quickly, dissolves thoroughly, and allows films to reach high mechanical strength without surface defects. MEK fulfills these criteria because its molecular structure supports cohesive evaporation regardless of variations in substrate material, climate, or application method.
MEK’s structure also determines its behavior in environmental and occupational safety contexts. Its volatility ensures that it disperses rapidly in air rather than accumulating on surfaces, and its biodegradability means microbial activity can break it down under aerobic conditions, preventing long-term persistence in soil and water. Compared with many industrial solvents historically used in coatings and cleaning, MEK does not accumulate in biological tissue. These positive attributes, however, do not eliminate the need for caution. Because the molecule evaporates quickly, exposure can occur through inhalation in closed environments, and its organic composition makes it flammable with a broad vapor ignition range. The same solvation strength that makes MEK effective in industrial cleaning means prolonged skin contact can remove natural oils. Safe handling therefore depends less on intrinsic toxicity and more on responsible ventilation, storage, and personal protective practices. These practical safety considerations originate directly from the same molecular structure that offers high volatility, strong solvation, and reliable industrial performance.
In adhesive and sealant science, MEK influences bond structure at the microscopic scale. By partially dissolving polymer surfaces, MEK allows polymer chains across two joining faces to migrate and entangle during solvent evaporation. This merging of polymer networks is the chemical basis of high-strength solvent welding used in synthetics such as vinyl, ABS, acrylic, nitrile rubber, neoprene, polyurethane, and chloroprene rubber. The strength and durability of such bonds depend on deep molecular penetration of the solvent before evaporation closes the polymer structure again. MEK’s compact molecular size and predictable diffusion speed make it ideal for creating these interpenetrating networks that generate structural adhesion, waterproofing performance, and long-term resilience against stress, vibration, and thermal cycling.
In broader industrial chemistry, MEK demonstrates how structure determines functional reach. A molecule small enough to evaporate rapidly, polar enough to dissolve complex coatings, stable enough to remain predictable over time, and reactive enough to participate in synthesis becomes an indispensable tool across multiple industries. MEK performs in automotive paint lines, aviation maintenance hangars, printing facilities, flooring factories, adhesive laboratories, and industrial cleaning systems not because it is merely a solvent but because its molecular architecture provides the exact balance of strength, speed, volatility, and compatibility upon which these manufacturing processes depend. The physical world of coatings, polymers, adhesion technology, and synthetic chemistry reflects the invisible geometry of MEK’s carbonyl-bearing structure.
Ultimately, methyl ethyl ketone stands as a clear illustration of how the molecular structure of an organic compound dictates its role in technology, industry, and environmental systems. Its subtle polarity, carbonyl functionality, and compact hydrocarbon framework allow it to dissolve substances that weaker solvents cannot, evaporate rapidly enough to support high-speed production, and integrate into chemical synthesis without destabilizing other functional groups. The advantages that MEK provides across manufacturing and engineering are rooted not in its name or industrial history but in the detailed relationship between its atoms and the forces that govern their arrangement. The composition of the molecule specifies what elements are present, but the structure of the molecule determines what those elements can accomplish. Through this precision of atomic design, methyl ethyl ketone has become one of the most influential and reliable solvents in modern manufacturing—and a reminder that the world of materials is ultimately constructed from the geometry of molecules working invisibly behind every engineered surface.