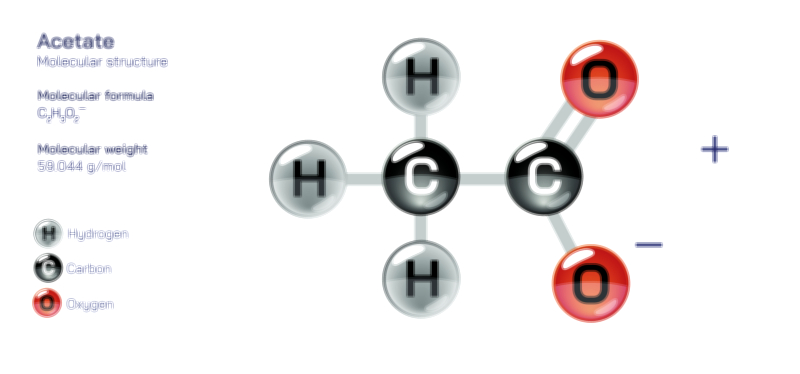
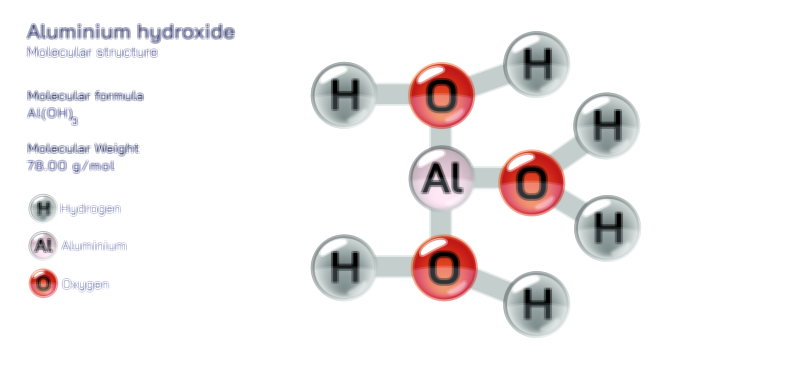
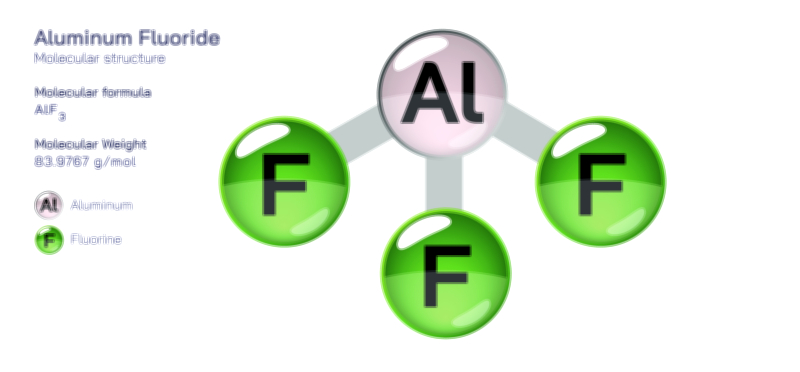
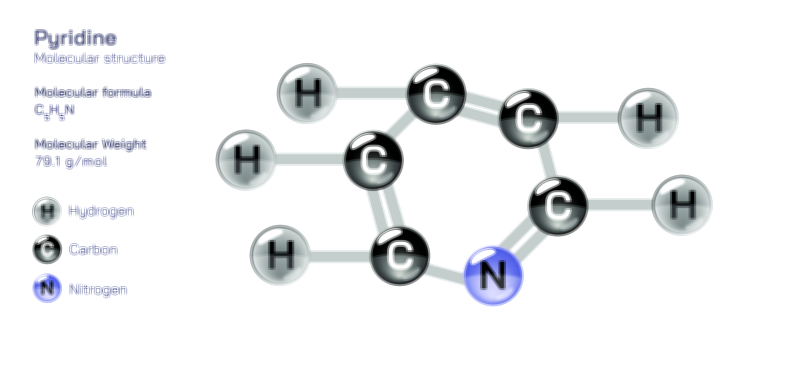
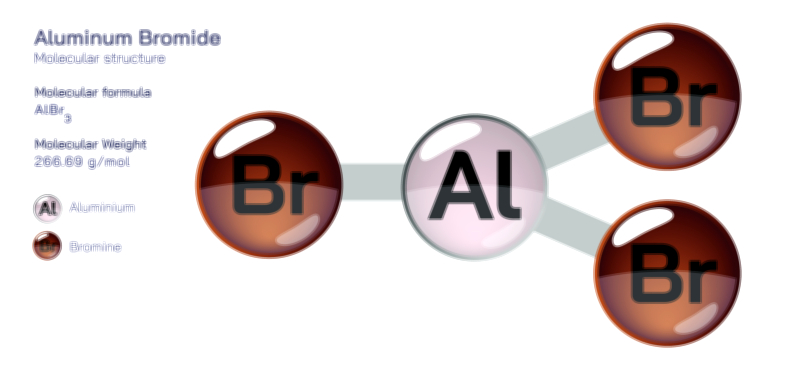
Acetate Ion — Chemical Structure and Molecular Composition Illustration
Structural Identity and Fundamental Nature of the Acetate Ion
The acetate ion, represented by the formula CH₃COO⁻, is one of the most important anions in organic and biological chemistry. It originates from acetic acid when the acidic proton is removed, leaving behind a negatively charged ion capable of participating in a wide range of chemical interactions. Its structure consists of a methyl group attached to a carboxylate group, forming a molecular arrangement that is both simple in appearance and remarkably rich in reactivity. The defining feature of the acetate ion is the carboxylate functional group, where the carbon atom is double-bonded to one oxygen and single-bonded to another oxygen. In the ionized form, however, the two carbon–oxygen bonds do not behave like classical single and double bonds. Instead, resonance delocalization equalizes the bond lengths, distributing the negative charge between both oxygen atoms. This resonance stability is a central concept in understanding the acetate ion’s chemical behavior, providing the structural foundation that makes it such a widespread and essential species across chemical, environmental, and biological systems.
Resonance, Charge Distribution, and Molecular Geometry
The electronic structure of the acetate ion reveals a balance created through resonance, where electrons move within the carboxylate group to create two equivalent resonance forms. These forms depict the negative charge alternating between the two oxygen atoms, and together they describe the true, averaged state of the molecule. As a result of this resonance, both C–O bonds in the acetate ion possess partial double-bond character, causing them to be identical in length and contributing to the overall stability of the ion. The geometry around the carbonyl carbon is trigonal planar, with bond angles close to 120 degrees, reflecting the sp² hybridization of the carbon atom. This planar arrangement also facilitates interactions with other ions, solvents, and biological molecules. The delocalization of the negative charge makes acetate less reactive than a typical alkoxide ion but enables it to participate effectively in nucleophilic reactions, coordination chemistry, and acid–base exchange processes. The methyl group attached to the carboxylate moiety provides hydrophobic character, but the strongly polar carboxylate group gives acetate the ability to dissolve readily in water and interact with a variety of polar solvents.
Physical and Chemical Properties Shaped by Molecular Structure
The acetate ion exhibits a combination of physical and chemical properties governed by its unique structural features. Its solubility in water is exceptionally high due to the capacity of the carboxylate group to form strong ion–dipole interactions with water molecules. This solubility explains why acetate salts, such as sodium acetate and potassium acetate, dissolve easily and play crucial roles in laboratory chemistry, medicine, and industry. The resonance-stabilized structure reduces the reactivity of the ion in comparison to other oxygen-based anions, creating a balance that allows it to act as a mild nucleophile and a relatively weak base. Its basicity is stronger than that of molecules like water but weaker than more highly charged nucleophiles, providing controlled reactivity in organic and biochemical systems. The acetate ion also displays buffering properties when paired with acetic acid, forming the well-known acetate buffer system widely used in biochemical experiments to maintain stable pH levels. This buffering ability emerges from the equilibrium between the protonated and deprotonated forms of the molecule, allowing it to moderate fluctuations in hydrogen ion concentration.
Biological Significance and Metabolic Integration
In biological systems, the acetate ion holds immense importance, serving as a key intermediate in metabolic pathways. It participates in the formation of acetyl-CoA, one of the most essential molecules in cellular respiration, fatty acid synthesis, and energy production. The simplicity and stability of the ion allow it to move easily through enzymatic pathways, providing carbon units for the Krebs cycle and supporting cellular function across a vast array of organisms. The acetate ion is also produced naturally during fermentation processes, especially in anaerobic bacteria that convert sugars into organic acids. In humans, acetate forms during the breakdown of alcohol and plays roles in hepatic metabolism. Because of its small size and balanced polarity, it diffuses efficiently through biological fluids and integrates seamlessly into biochemical networks. This versatility underscores why the acetate ion is foundational in both microbiological systems and complex organisms.
Industrial and Environmental Relevance of Acetate Compounds
Beyond biological contexts, the acetate ion is vital in industrial chemistry where acetate salts and esters are used in manufacturing polymers, photographic chemicals, adhesives, textiles, and food preservatives. Sodium acetate is employed in heating pads that rely on crystallization-based heat release, while calcium magnesium acetate is used as an environmentally friendly de-icing agent that reduces corrosion compared to traditional salts. In the realm of environmental chemistry, acetate is a readily biodegradable species that acts as a carbon source for microorganisms involved in wastewater treatment, soil nutrient cycles, and bioremediation processes. Because it is non-toxic at typical environmental concentrations, acetate becomes an ideal chemical for processes that depend on microbial metabolism and natural ecological interactions. Its participation in these systems emphasizes its role as a bridge between organic chemistry and environmental sustainability, showing how a simple anion can influence large-scale biological and ecological networks.
Illustrating the Acetate Ion and Its Chemical Behavior
Scientific illustrations of the acetate ion typically emphasize its resonance structures, showing the flow of electron density between the two oxygen atoms. These depictions reveal why both carbon–oxygen bonds appear equivalent and how the negative charge is distributed across the ion. Diagrams also highlight the planar geometry and the orientation of the methyl group, helping visualize how the ion interacts with surrounding molecules. Such illustrations often include representations of hydrogen bonding, ion–dipole interactions, or coordination complexes where acetate binds to metal centers, providing insights into its behavior in different environments. By understanding these structural and electronic features through visual and conceptual models, one gains a deeper appreciation for how the acetate ion operates as a fundamental building block in chemistry, biology, and industry, despite its apparent simplicity.