Calorimeter — Understanding Heat Measurement and Energy Transfer in Science
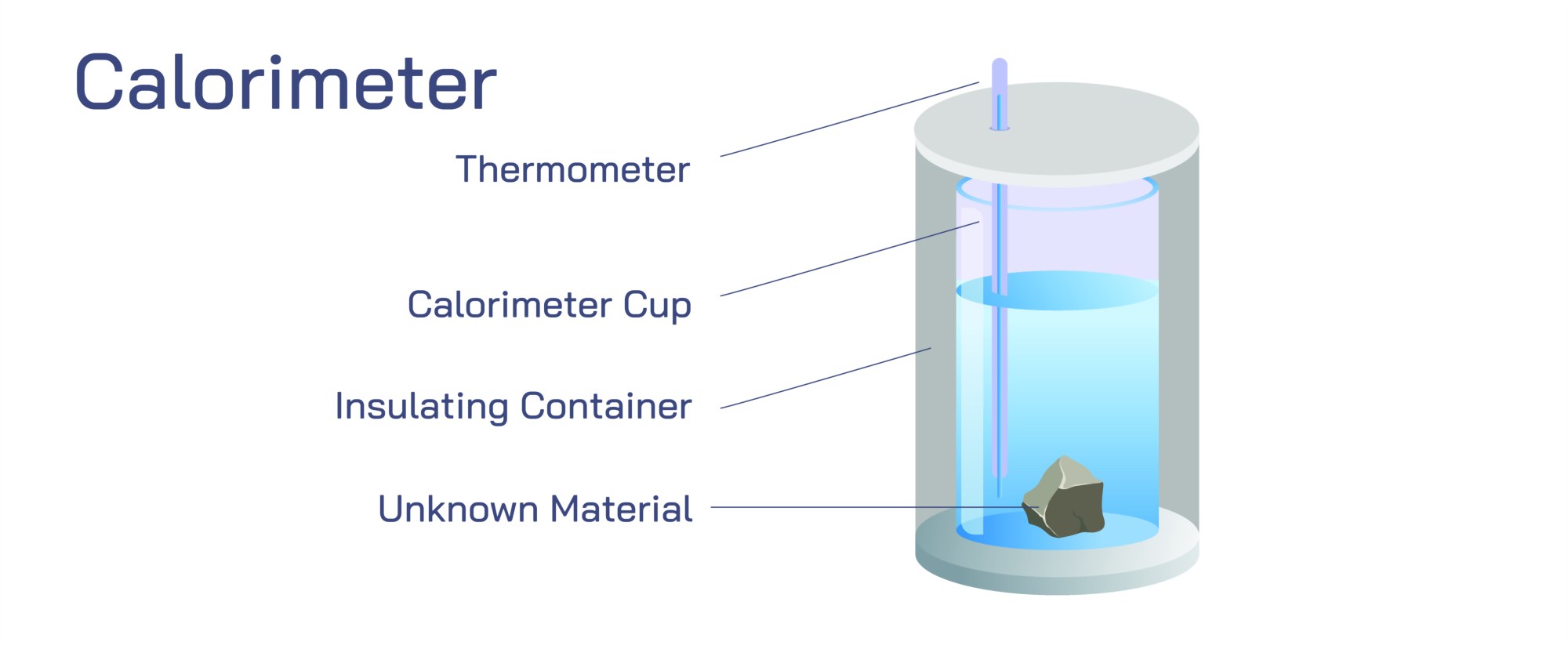
A calorimeter is an instrument used to study how heat moves within a system. It allows scientists and students to observe the change in energy during physical or chemical processes. When a substance undergoes a transformation, the movement of heat can be detected by measuring the change in temperature of the surrounding medium. The calorimeter is designed to limit the exchange of heat with the external environment, making it easier to detect small changes inside the device. By studying these changes, one can understand how materials behave when they absorb or release energy.
The basic idea behind a calorimeter revolves around isolating the process being studied. When a sample is placed inside the container, the device shields it from outside influences as much as possible. A liquid is often used as the surrounding medium because its temperature reacts noticeably to any incoming or outgoing energy. When the sample interacts with the liquid, the temperature within the system shifts. Observing this change helps identify whether energy has entered or left the surrounding medium. This pattern reveals valuable information about the nature of the transformation that has taken place.
Different designs of calorimeters exist depending on the type of study being performed. Some devices are simple and used for basic classroom experiments. These usually involve a container, a liquid, and a temperature-reading instrument. More advanced models are used for detailed studies of materials or reactions that require high accuracy. Some devices are built to withstand strong reactions, while others are created to measure slow and gentle energy changes. All designs share the purpose of capturing heat movement clearly and preventing outside interference.
In many scientific fields, calorimeters play an essential role in analyzing how substances behave. They help explore the energy involved in changes such as melting, dissolving, or reacting with other materials. When a sample warms or cools the surrounding liquid, the shift reflects the amount of energy stored or released within the material. These observations provide insight into the stability of substances, the behavior of mixtures, and the nature of molecular interactions. This makes the calorimeter a valuable tool for understanding energy flow in both natural and controlled environments.
Beyond the laboratory, the idea behind calorimetry also appears in daily experiences. Anytime an object warms or cools another object it contacts, a similar pattern of energy movement occurs. The calorimeter simply captures this movement in a controlled and measurable form. By studying this interaction, students and researchers develop a clearer understanding of how energy behaves in different systems. The device helps demonstrate that every change in matter is accompanied by movement of energy, even when the change appears subtle.
Understanding how a calorimeter works provides a foundation for exploring larger scientific ideas related to heat, energy, and material behavior. The device offers a clear and structured way to observe energy movement, allowing learners to connect theoretical ideas with visible outcomes. Through careful observation inside a calorimeter, one can recognize how materials respond to changing conditions and how energy shapes every transformation in the world around us.