Combustion Process Illustration: Chemical Reaction Between Fuel and Oxygen Explained
Combustion is one of the most familiar chemical processes in everyday life, yet a deeper look at its mechanism reveals a fascinating sequence of molecular events that transform stored chemical energy into heat, light, and new substances. An illustration of the combustion process typically begins with the presence of a fuel—such as wood, gasoline, natural gas, coal, ethanol, wax, or paper—and a supply of oxygen from the surrounding air. At room temperature these two components can coexist without reacting because the fuel molecules are stable, and the energy barrier required to break their chemical bonds has not been crossed. Once an ignition source provides sufficient energy—whether from a spark, a match, friction, or intense heat—the molecules of the fuel absorb energy and become activated, allowing oxygen to interact with them at the molecular level. This initial step, often represented in diagrams as the input of activation energy, breaks chemical bonds inside the fuel and begins a chain reaction that releases stored energy. The start of the reaction is the moment when heat is high enough to trigger new chemical bonds to form between oxygen and carbon and between oxygen and hydrogen within the fuel, marking the transformation of potential energy into thermal and radiant energy.
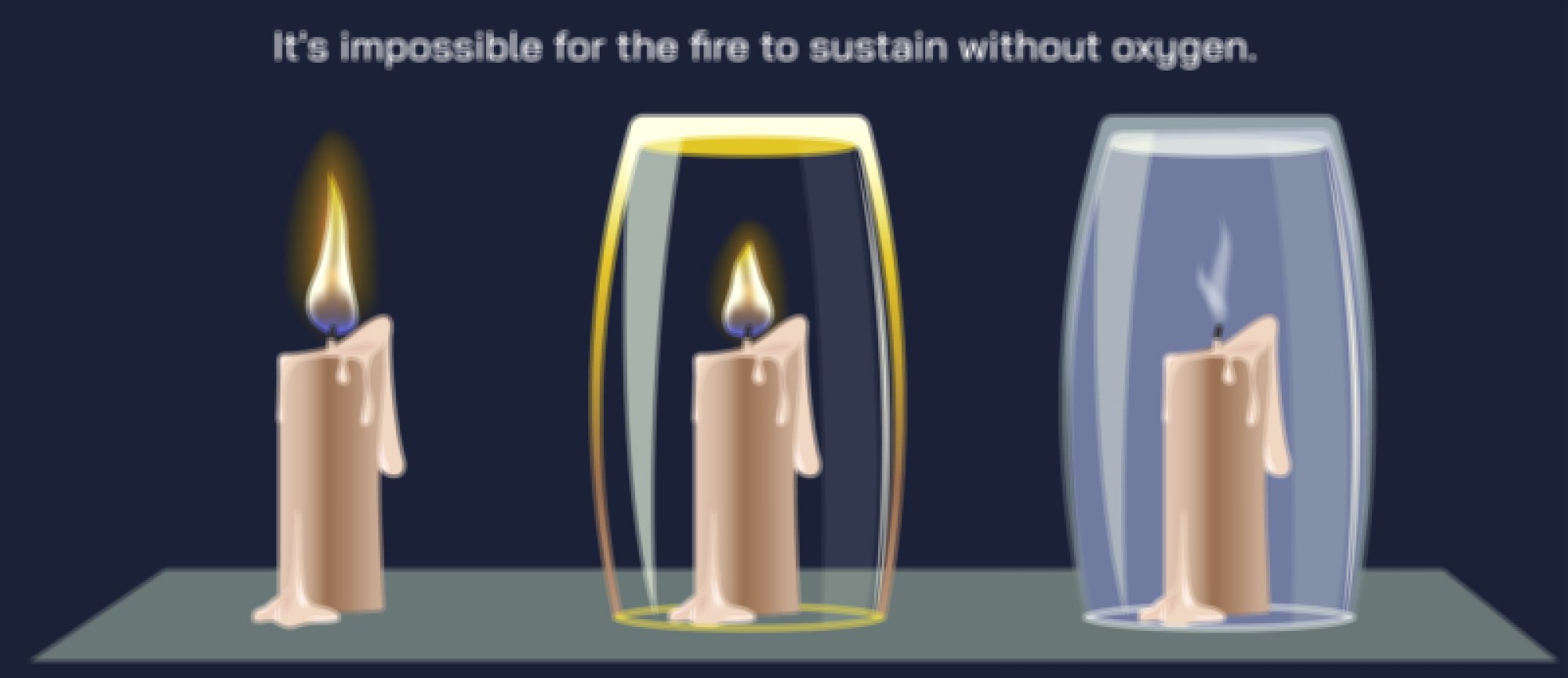
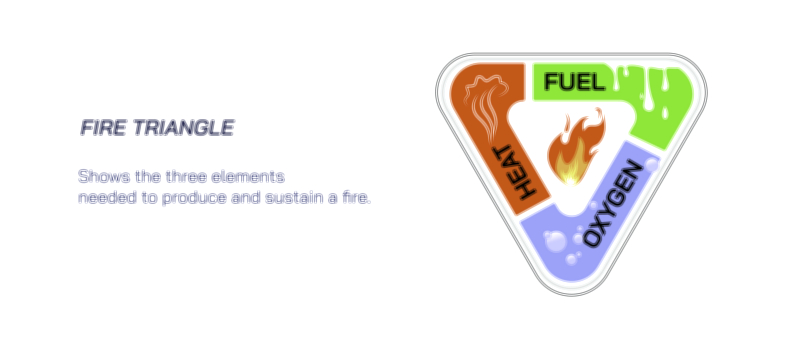
In a more detailed combustion illustration, the reaction is often shown as a sequence of three essential components—fuel, oxygen, and ignition—depicted in the shape of the fire triangle. Each side of this triangle is necessary for the combustion reaction to continue. If oxygen is removed, combustion ends. If fuel is consumed or taken away, the flames die out. If heat is lost more quickly than it is produced, the reaction stops because the remaining fuel cannot reach the activation threshold. This triangle representation reinforces a fundamental point: combustion is self-sustaining only when the reaction produces enough heat to keep converting fuel molecules into reactive fragments that continue bonding with oxygen. In diagrams showing this self-sustaining cycle, flames appear as a visual cue for how heat and light are emitted as products of the reaction in addition to the newly formed chemical substances.
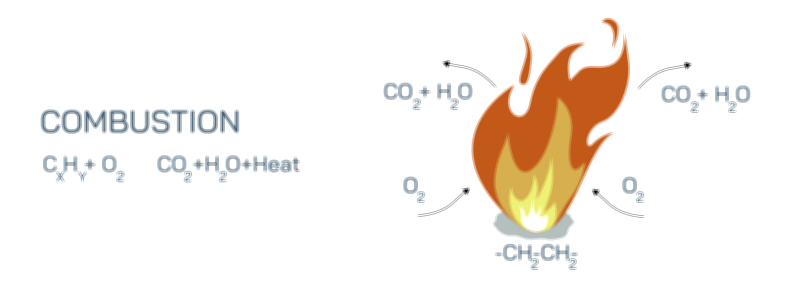
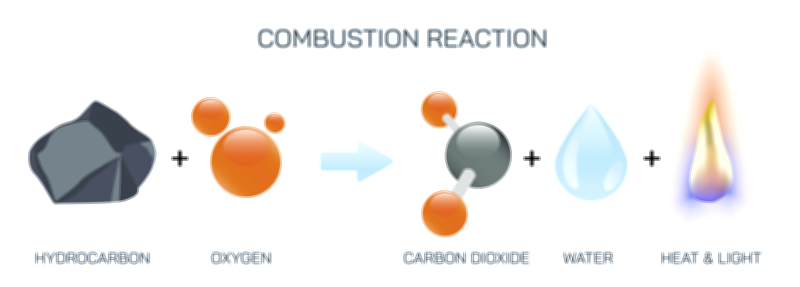
Zooming into the microscopic levels, combustion illustrations typically differentiate between the reactants—fuel and oxygen—and the products—carbon dioxide (CO₂), water vapor (H₂O), and heat. When hydrocarbons burn in the presence of sufficient oxygen, each carbon atom bonds with two oxygen atoms to form CO₂, while each hydrogen atom bonds with one oxygen atom to form water. These newly formed molecules have lower internal energy than the original hydrocarbon molecules, meaning the excess energy is released into the surroundings. That released energy manifests as intense heat and visible light, providing the most recognizable signature of combustion: fire. If an illustration shows arrows depicting energy flow, it emphasizes that combustion converts chemical energy stored in bonded atoms into thermal energy, electromagnetic radiation, sound, and sometimes mechanical work. The chemical rearrangement of atoms—the reorganization of electrons—gives rise to flames, glowing embers, heat waves, and representative color variations in the fire depending on which substances are burning.
Not all combustion is the same, and illustrations frequently distinguish complete combustion from incomplete combustion. In complete combustion, oxygen is abundant and fuel burns efficiently, producing carbon dioxide and water as the primary byproducts. A visual depiction shows clean blue flames—like those from a gas stove—representing the high-temperature, highly oxygenated reaction environment. In incomplete combustion, however, oxygen availability is limited. Under these conditions carbon atoms cannot fully bond with oxygen and instead form carbon monoxide (CO) or even soot (C), which appears as black particulate matter. An illustration showing incomplete combustion might depict yellow or orange flames with smoke rising from the fuel. The color originates from glowing soot particles radiating light rather than exclusively from gas-phase reactions. This side-by-side comparison of complete versus incomplete combustion visually explains why engines require tuned airflow systems and why indoor fires need ventilation: insufficient oxygen leads to lower efficiency, lower temperature, increased smoke, and production of harmful gases.
A particularly valuable combustion illustration reveals the reaction zone, which includes the fuel-rich region, the oxygen-rich region, and the flame front where combustion occurs. In a burning candle, for example, the outermost part of the flame burns cleanly because it contains an abundance of oxygen, while the inner zone closer to the wick burns incompletely and produces soot. Educational diagrams often show the flame divided into three sections—dark inner core where vaporizing fuel resides, bright yellow luminous region where partial combustion and glowing soot occur, and nearly invisible pale-blue outer region where complete combustion takes place. These layered depictions help viewers understand why the flame color changes with temperature and oxygen supply. They also explain why adjusting airflow changes the appearance of flames in burners, stoves, car engines, and furnaces.
Beyond heat and flame formation, ignition and propagation mechanisms play vital roles in combustion illustrations. The ignition phase, represented visually by the spark or initial heat surge, increases the kinetic energy of fuel molecules enough to initiate the reaction. Once molecules are activated, a chain reaction takes over: each successful fuel-oxygen reaction releases heat that activates more fuel molecules. In diagrams this is sometimes shown as a branching sequence, where each molecular reaction contributes to the next. The speed of this propagation determines the reaction’s nature. A slow, controlled combustion—as in a candle—progresses steadily, releasing heat gradually. A rapid and explosive combustion—as in gunpowder or gasoline vapor—occurs when heat propagation accelerates faster than heat can dissipate, resulting in powerful pressure waves and rapid gas expansion. Illustrations comparing these two show still-burning flames on one side and shock waves on the other, emphasizing how fuel concentration, oxygen ratio, and confinement define the reaction type.
The role of temperature is another core theme in combustion diagrams. Combustion cannot start without reaching the ignition temperature, the point at which fuel molecules can react. But once combustion is underway, the reaction continues only if the flame temperature is maintained above the ignition threshold. This is often shown with feedback loops in illustrations—heat sustains combustion, combustion produces heat. If something disrupts this cycle—water cooling, reduction of oxygen flow, or removal of fuel—the flame goes out. These diagrams explain why water extinguishes fire by absorbing heat and lowering flame temperature, why carbon dioxide extinguishers work by displacing oxygen, and why fire blankets smother flames by cutting off airflow. Each extinguishing mechanism works by interrupting a different corner of the fire triangle.
Combustion illustrations also extend beyond ordinary flames to include specialized reactions like internal combustion engines, jet propulsion, and industrial furnaces, which apply combustion to mechanical work. In an engine illustration, fuel and air enter the combustion chamber, the spark causes ignition, gases expand rapidly due to the release of heat, and the resulting pressure pushes the piston, converting chemical energy into mechanical energy. In jet engines, combusted air expands through turbines and nozzles, creating thrust. In power plants, combustion heats water to generate steam that rotates turbines. These industrial diagrams show that combustion is not just a source of heat but a driver of technology, transportation, and power generation.
On a global scale, illustrations of combustion increasingly incorporate environmental consequences. The molecular reaction that produces carbon dioxide contributes to greenhouse gas emissions, while the formation of soot contributes to air pollution and respiratory health concerns. Combustion of biofuels, hydrogen, or renewable synthetic fuels is sometimes compared visually to combustion of fossil fuels to show differences in carbon output. In these comparisons, sustainable combustion cycles illustrate how CO₂ absorbed during plant growth can offset emissions, whereas fossil fuel combustion introduces carbon stored underground for millions of years into the atmosphere. These images connect the chemistry of fire to larger ecological and climate systems.
A well-designed combustion illustration ultimately communicates more than the transformation of fuel into heat; it captures a process that sits at the intersection of chemistry, physics, engineering, energy, safety, and environmental science. It visualizes how the arrangement of atoms determines energy storage, how oxygen unlocks that energy, how heat drives reaction cycles, and how control of combustion shapes technology and daily life. By showing the transformation from reactants to products, from stored potential energy to dynamic motion and radiant heat, a combustion illustration transforms an ordinary flame into a scientific narrative—one that explains how burning begins, how it sustains itself, how it can be controlled, and how understanding it can benefit society from the level of a candle wick to the engines that power global movement.