Absorption and Adsorption — Surface Action, Bulk Uptake, and Material Interaction Explained
Absorption and adsorption are two important processes that describe how substances interact with different materials. Although the words sound similar, they refer to very different mechanisms. Understanding these differences is essential across chemistry, physics, environmental studies, material science, and many industrial applications. Both processes involve the capture of particles, but the way this capture happens defines the distinction between internal uptake and surface attachment.
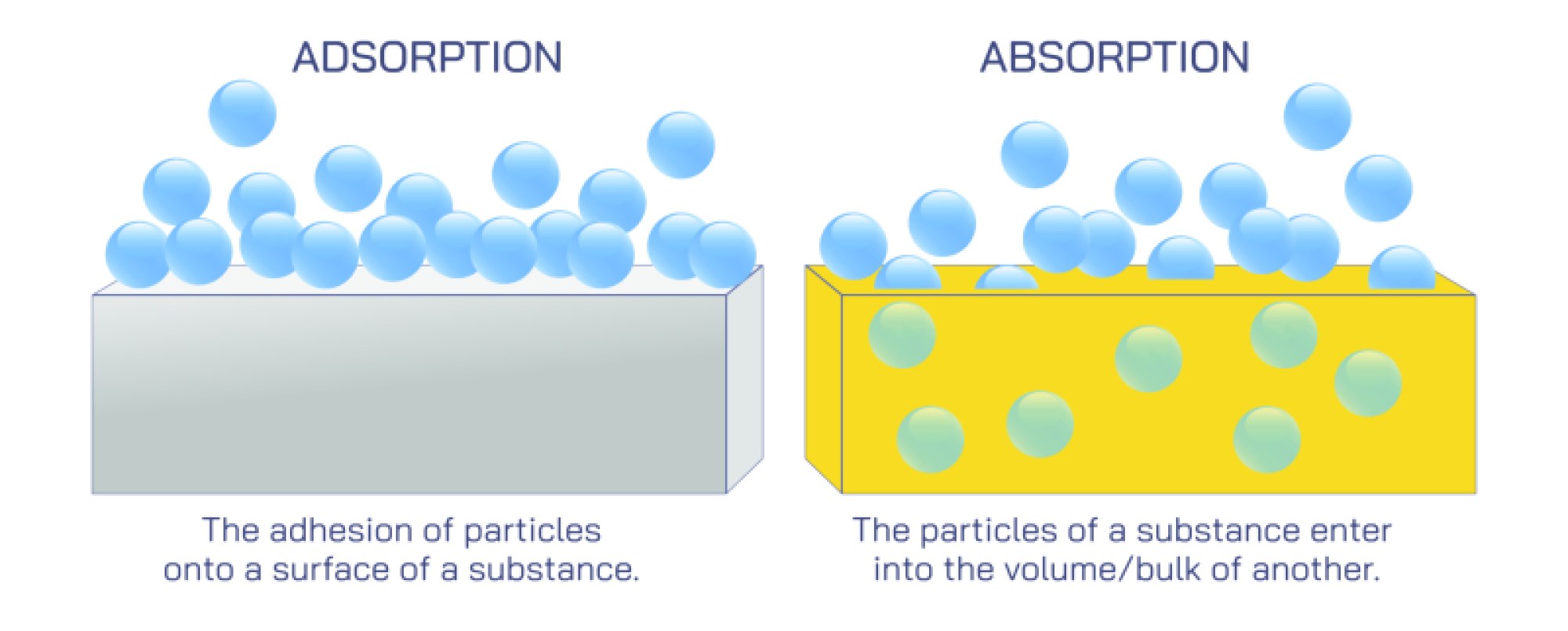
Absorption is a process where one substance enters completely into another, distributing through the entire volume. In this mechanism, the absorbed substance moves from the outer region into the inner regions of the absorbing medium. This bulk penetration often occurs through diffusion, where particles move from an area of higher concentration to lower concentration. Examples include water soaking into a sponge, gases dissolving into liquids, or moisture entering plant tissues. Absorption involves the full internal body of the material, meaning the interacting substance becomes part of the overall volume of the medium.
Adsorption, in contrast, takes place only on the surface of a material. Instead of entering the interior, particles stay attached to the outer surface through physical or chemical interactions. These interactions may include weak forces, stronger bonding, or electrostatic attraction depending on the system. Because adsorption is surface-based, materials with large surface areas—such as charcoal, silica gel, and porous solids—are particularly effective. This process plays a major role in purification, filtration, and catalyst systems where surface action is important for capturing unwanted particles.
One of the primary differences between the two processes lies in the depth of interaction. Absorption affects the entire internal region, while adsorption is limited to the surface. This difference influences how fast the processes occur, the type of materials involved, and the energy changes required. In absorption, the captured substance spreads within the medium, often involving molecular movement into deeper layers. In adsorption, particles stay on the outer layer, forming a film or layer on the surface.
Energy behavior also differentiates the two. Absorption often involves uniform internal distribution and may depend on solubility or compatibility between materials. Adsorption may occur through physical forces or chemical reactions, which can lead to energy release or a more complex interaction depending on how strongly the particles attach to the surface. This distinction is especially important in industrial processes where efficiency and resource use depend on how substances interact with surfaces or internal regions.
Real-world examples help highlight the differences. When a liquid is soaked by paper, the process is absorption because the liquid enters the entire thickness of the paper. When air pollutants stick to the surface of activated charcoal, the process is adsorption because the particles remain on the surface. Similarly, when gas dissolves into water inside a sealed container, absorption occurs, while the binding of dye onto cloth fibers often involves adsorption on the surface before deeper interactions take place.
Both processes play crucial roles in systems involving purification, separation, and chemical reactions. Absorption is widely used in refrigeration systems, chemical reactions in solutions, and biological uptake within plants and animals. Adsorption is essential in water purification, gas masks, catalysts, and various industrial applications requiring surface-based interactions.
Overall, absorption and adsorption describe two different yet important ways substances interact with materials. Absorption involves full internal penetration into the medium, while adsorption focuses on surface attachment. Understanding these concepts provides clarity in scientific analysis and enhances comprehension of natural processes and technological systems.