Thermal Conduction – Understanding Heat Transfer Through Matter, Molecular Interactions, Microscopic Motion, and the Role of Materials in Temperature Flow
Thermal conduction is one of the most fundamental mechanisms of heat transfer in physics, and it describes the process by which thermal energy moves through matter from regions of higher temperature to regions of lower temperature. Although conduction appears simple on the surface—we feel heat pass from a hot object to our hand or see a metal spoon warm up when placed in boiling water—the underlying mechanism is deeply rooted in microscopic particle interactions and the physics of energy distribution within solids, liquids, and gases. Conduction occurs because heat is not a substance but a form of energy stored in the vibration and motion of particles. When part of a material becomes hot, its particles move more rapidly, collide with neighboring particles, and pass on their kinetic energy. This chain of energy transfer continues until the temperature becomes uniform across the material or until heat escapes into the surrounding environment. Understanding thermal conduction provides insight into why some materials feel hot or cold, why metals heat up quickly, why insulating materials protect against burns, and how heat management enables technology ranging from cooking and construction to electronics, refrigeration, and spacecraft engineering.
The Microscopic Origin of Heat Transfer and the Role of Particle Motion
At the heart of thermal conduction lies the behavior of atoms, molecules, and electrons. In solids, particles are arranged in fixed positions within a lattice but continuously vibrate around their equilibrium positions. When part of the solid is heated, the vibrations in that region intensify and cause atoms to knock against their neighbors, triggering a cascade of energy transfer across the lattice. In metals, conduction is especially efficient because in addition to atomic vibrations, free electrons act as highly mobile carriers of energy. These electrons travel through the material with high velocity and rapidly distribute heat, making metals such as copper, aluminum, and silver excellent conductors. In contrast, insulators such as wood, rubber, plastic, and wool lack free electrons and have internal structures that hinder atomic vibration transmission, so heat passes through them very slowly. In liquids and gases, conduction behaves differently because particles are not fixed in place; they collide less frequently, making conduction less effective than in solids. This explains why air and other gases are good thermal insulators and why air gaps are widely used in insulating clothing, building walls, and double glazing.
Temperature Gradient, Direction of Heat Flow, and Dynamic Equilibrium
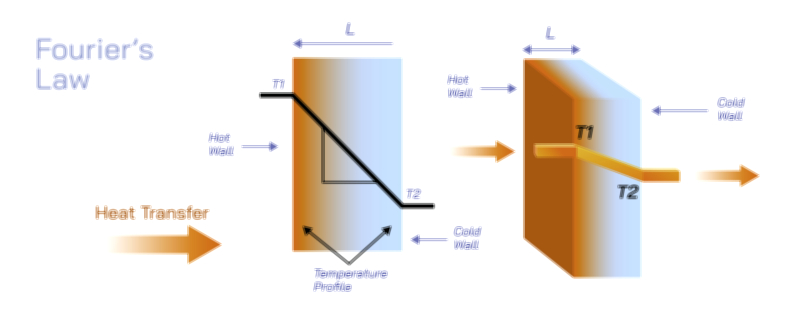
Conduction always takes place from higher temperature regions to lower temperature regions because temperature reflects the average kinetic energy of particles. When two regions are in contact, the hotter one has faster-moving particles and the colder one has slower-moving particles. Energy flows spontaneously in the direction that reduces this difference. This behavior continues until thermal equilibrium is reached, meaning both regions attain the same temperature and no net heat transfer occurs. The rate at which conduction happens depends on the temperature gradient—the difference in temperature across a given distance. A steep gradient, such as touching a hot iron rod at one end, causes rapid heat flow, whereas a gradual gradient results in slower conduction. Engineers use this principle when designing heat sinks, insulation panels, and thermal barriers, shaping materials and interfaces to either accelerate or slow the movement of heat depending on the application.
Thermal Conductivity and Why Materials Behave Differently
Every material has an inherent capacity to conduct heat, determined by a property known as thermal conductivity. Materials with high thermal conductivity transfer heat quickly and include metals such as copper, silver, aluminum, and gold. These substances are used in cookware, heat exchangers, radiator fins, electronic cooling systems, and industrial tools because they can rapidly distribute heat and prevent hotspots from forming. Materials with very low thermal conductivity are thermal insulators. These include wool, foam, fiberglass, wood, rubber, plastics, air, and vacuum-sealed panels. They are used to prevent heat flow and maintain temperature differences, making them ideal for warm clothing, insulating walls, refrigeration, cryogenic storage, and thermal protection systems for space vehicles. The enormous variation in thermal conductivity arises from differences in bonding, electron mobility, atomic spacing, impurities, crystal defects, and internal structure. Real-world materials are often engineered to combine both conductive and insulating layers based on performance requirements.
Conduction in the Natural World and Everyday Experience
Thermal conduction affects countless daily interactions long before we study it formally. The handle of a metal pan gets hot because heat travels through the metal rapidly, but a wooden or silicone handle remains cool because wood and polymers conduct heat poorly. A tile floor feels colder than a carpet even when both are at the same temperature because tile conducts heat away from the skin quicker, giving the sensation of cold. Ice melts faster in contact with metal than with plastic because conduction transfers thermal energy from the surroundings more effectively when the contact material is a conductor. Even the simple act of cooking or maintaining room temperature depends heavily on conduction through materials such as cookware, utensils, walls, insulation layers, and heating components. These everyday examples demonstrate that conduction is not restricted to theoretical physics but plays a constant role in practical comfort, safety, and technology.
The Importance of Thermal Conduction in Technology, Engineering, and Scientific Innovation
One of the highest-stakes applications of thermal conduction is the cooling of electronics. Modern devices generate large amounts of heat in small spaces, and their survival depends on rapid heat removal through conduction to heat sinks, thermal pastes, and cooling plates. Without conduction, processors, LEDs, power electronics, and batteries would overheat and fail. In industrial contexts, conduction governs the design of furnaces, refrigeration systems, heat exchangers, metal forging, and machinery lubrication. In architecture, energy efficiency depends on selecting construction materials that minimize heat loss or heat gain through conduction. In aerospace engineering, conduction plays a crucial role in designing heat shields that prevent spacecraft from overheating when exiting or entering planetary atmospheres. In cryogenics and advanced research, preventing unwanted conduction is essential to maintaining extremely low temperatures for experiments in quantum physics and superconductivity. These examples reveal how deeply mastering conduction enables scientists and engineers to control temperature with precision and prevent thermal failure in high-performance systems.
Why Thermal Conduction Matters in Education and Physics Understanding
Studying thermal conduction teaches more than the mechanics of heat transfer; it demonstrates how microscopic physics explains macroscopic behavior. It reveals how temperature is tied to particle motion and how energy flows not because of intention but because nature distributes energy until uniformity is reached. It shows that materials differ not just in appearance but in the way their internal structure channels or resists heat flow. It connects the invisible world of atoms and electrons with the tangible world of comfort, safety, manufacturing, and technology. By learning how conduction works, students gain an understanding of why metal radiators heat rooms efficiently, why insulation keeps houses warm, why engines require cooling systems, and why a spacecraft needs both conductive and insulating components to survive extreme temperature fluctuations.
Thermal conduction ultimately highlights a central truth in physics: the flow of energy governs the stability of systems, and the way materials manage that flow defines their usefulness. Whether in the warmth of a home, the precision of an electronic device, the performance of a machine, or the survival of an astronaut in space, conduction is a silent but constant process that shapes physical experiences and technological progress. Through this understanding, learners come to appreciate that physics is not only a study of equations but a direct exploration of how the universe balances energy through the structure and behavior of matter.