Absorption and Adsorption — Understanding Internal Uptake and Surface-Level Interaction
Absorption and adsorption are two important concepts used to describe how one substance interacts with another. Although the words may sound similar, they refer to very different processes that occur in distinct ways. Understanding the difference helps learners grasp ideas related to chemistry, material science, daily life processes, and industrial applications. These ideas appear in natural systems, biological functions, purification methods, and environmental science, making them essential for both academic study and practical observation.
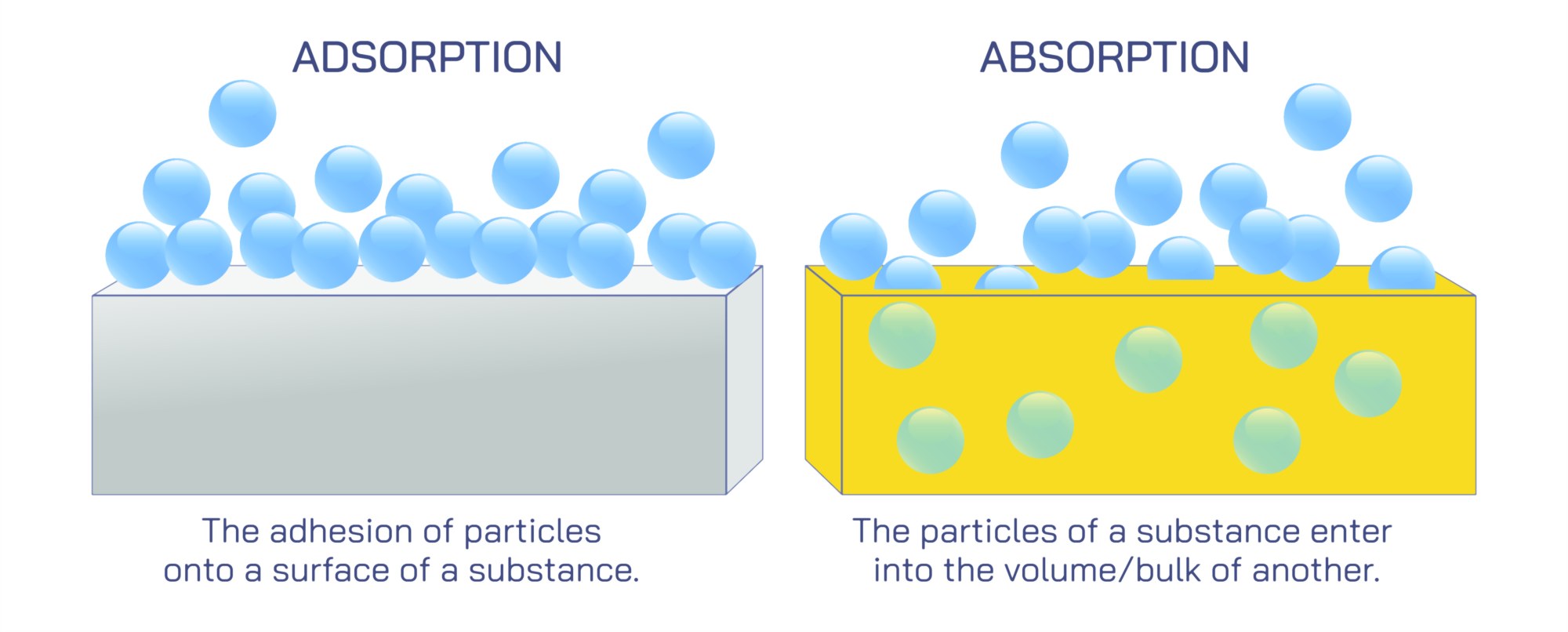
Absorption refers to a process in which one substance enters the internal volume of another. In this form of interaction, particles spread throughout the entire body of the absorbing material. The idea is similar to how a sponge takes in water, allowing the liquid to move inside its structure. The internal regions of the absorbing medium provide ample space for the incoming substance, allowing uniform distribution. This internal movement typically depends on factors such as the nature of the material, temperature, physical structure, and the overall environment in which the interaction occurs. Absorption is commonly encountered in natural settings, industrial tasks, and even inside living systems where various components travel through membranes and tissues.
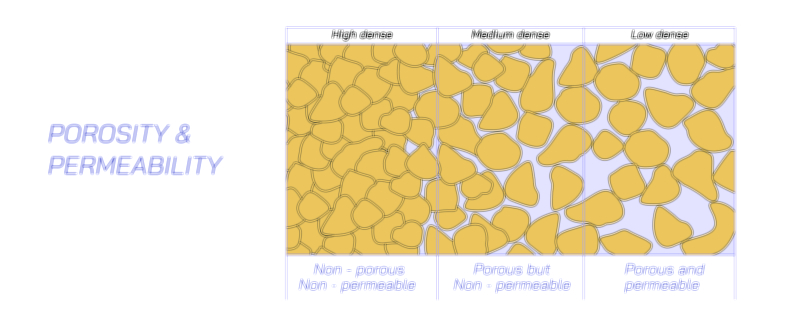
Adsorption, on the other hand, is a surface-based phenomenon. Instead of entering the internal region of a material, the incoming particles attach only to the outer layer. This forms a thin clustered pattern on the material’s exterior. The interaction often occurs due to forces acting at the surface of solids or specific contact points. A familiar example can be seen when certain solids hold molecules from surrounding fluids along their surfaces. This form of interaction is important in fields like filtration, purification, and environmental cleaning, where harmful particles need to be trapped without being absorbed internally. The surface of the material plays the main role here, as the effectiveness of adsorption depends strongly on available area and the nature of the solid.
A major difference between absorption and adsorption is how the added particles distribute themselves. In absorption, they move inside and spread throughout the entire medium. In adsorption, they remain on the outer boundary, forming a surface layer. Because of this difference, the rate of interaction, the amount of material involved, and the conditions required for both processes show noticeable variation. Absorption generally involves deeper penetration and may occur over a longer time. Adsorption often happens quickly because it is limited to the exposed external region.
These processes also appear in various scientific and technological systems. Plants rely on absorption when roots take in water from soil. Certain cleaning systems and protective masks depend on adsorption to trap unwanted components from air or liquid. Industrial procedures use both mechanisms depending on whether inner uptake or surface holding is needed. Even environmental processes follow these principles when substances travel between soil, air, and water, interacting with mineral surfaces or porous natural materials.
Understanding both concepts helps learners compare internal and surface interactions in clear and simple terms. This distinction also aids in studying how materials behave when exposed to different regions of contact. Whether observed in daily activities, natural cycles, or laboratory experiments, the difference between absorption and adsorption remains essential for interpreting how matter interacts within a system.