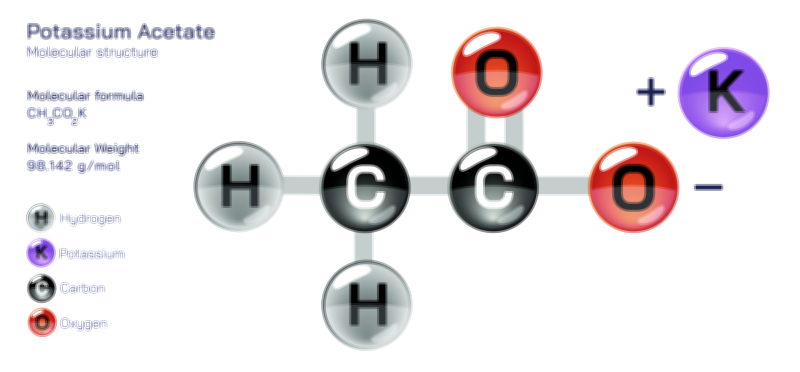
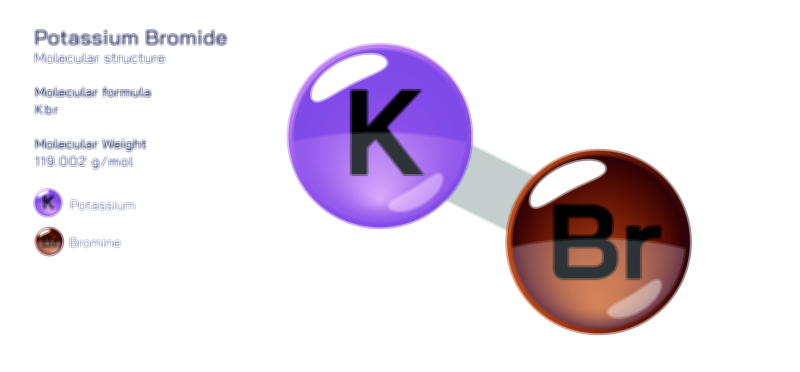
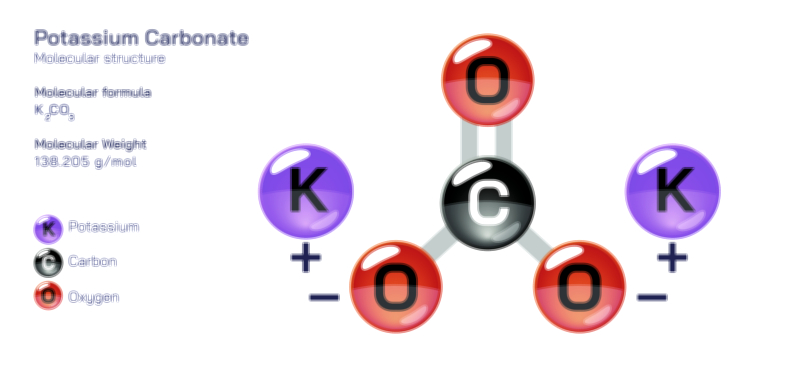
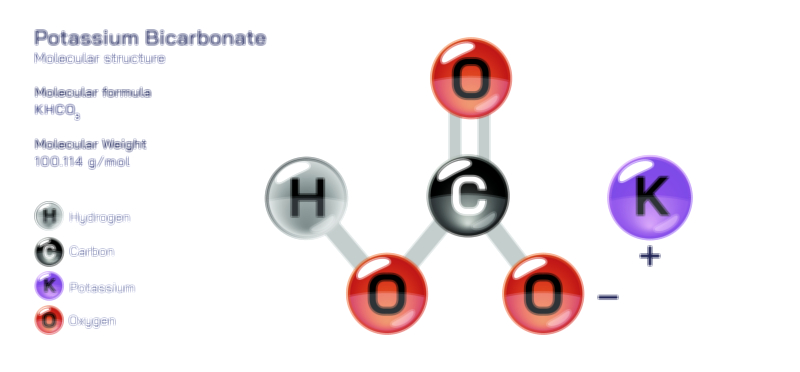
ØÉÅØÉ©ØɽØÉ©ØɼØÉóØÉŁØÉ▓ ØÉéØÉ©ØɦØÉ£ØÉ×ØÉ®ØÉŁ ØÉÜØɦØÉØ ØÉīØÉÜØÉŁØÉ×ØɽØÉóØÉÜØÉź ØÉÅØÉ©ØɽØÉ× ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×
Porosity is a fundamental property that describes the presence, quantity, and arrangement of empty spacesŌĆöcalled poresŌĆöwithin a solid material. Even though many materials appear compact and solid to the naked eye, their internal structure often contains microscopic or macroscopic voids that significantly influence their physical behaviour. These pores can hold air, fluids, or gases, and their distribution determines how a material interacts with its surroundings. The concept of porosity is central in fields such as geology, civil engineering, soil science, materials science, biomedical technology, filtration, environmental engineering, and manufacturing. Whether determining how well a sponge absorbs water, how cement gains strength, how rocks store groundwater, or how catalytic materials accelerate chemical reactions, porosity remains a key factor shaping functionality. Beyond simple emptiness, pores define how matter flows through a structure, how energy transfers within it, and how mechanical stability is maintained under stress.
At its core, porosity refers to the fraction of a material's volume that is occupied by voids rather than solid matter. These voids vary greatly in size, shape, and connectivity, and this variation governs how a material behaves. Some materials contain large, open channels that allow liquids and gases to flow freelyŌĆöas in highly permeable soils, foams, and filters. Others may contain tiny, isolated cavities that remain sealed off and contribute to properties like lightweight structure, buoyancy, and thermal insulation rather than fluid movement. Porosity does not only concern how many pores exist; it also concerns how they are interconnected. A material with high porosity but poor connectivity may store fluid without allowing it to pass through, while a material with moderate porosity but well-connected channels may become an excellent conductor of flow. Therefore, porosity and permeability go hand-in-hand, but they are not identical concepts. Porosity describes the total volume of pores, whereas permeability describes the ease with which fluids move through those pores.
The structure of pores within a material can be classified broadly into open pores and closed pores. Open pores form channels that connect to each other and to the materialŌĆÖs exterior surface. These pores allow filtration, fluid transport, capillary action, and diffusion of gases, making them important in applications like water purification membranes, soil moisture movement, breathable textiles, catalytic supports, and biomedical implants. Closed pores, on the other hand, remain sealed within the material without openings to the outside. These pores trap air or gas and contribute to lightweight structure, insulation, shock absorption, acoustic dampening, and buoyancy. Materials such as expanded polystyrene foam, lightweight concrete, and volcanic pumice contain mostly closed pores, which explains their insulation properties and low density. Many engineering materials contain a combination of both pore types, and manipulating this balance allows experts to optimize a material for specific performance needs.
Pores also vary dramatically in size, and this variation gives rise to different categories of porous materials. Macroporous structures have large pores visible under optical microscopy and allow rapid flow of liquids and gases, as seen in natural sponges, sandstone, and filter foams. Mesoporous materials contain intermediate-sized pores that support controlled flow and adsorption, making them useful in catalysis, drug delivery, and gas storage. Microporous structures contain extremely small pores approaching molecular dimensions, making them valuable for selective separation, molecular sieving, and chemical capture. Activated carbon, zeolites, and metalŌĆōorganic frameworks rely on microporosity to isolate specific molecules from mixtures by using precisely sized pore networks. Even within the same material type, pore sizes can vary across scales, resulting in hierarchical pore structures that maximize both storage capacity and flow pathways. This complexity reveals that porosity is not merely a static characteristic but an engineered feature that governs both physical and chemical behaviour.
In natural environments, porosity plays a major role in controlling how resources and energy move. In geology, porous rock formations such as sandstone, limestone, and shale determine how groundwater, petroleum, and natural gas accumulate and migrate. Soil porosity regulates how plants receive water, air, and nutrients, directly affecting agricultural productivity. Porous snow and ice structures influence the stability of glaciers and determine how water flows in cold regions. In biomedicine, the porosity of bones allows blood vessels to penetrate the skeletal structure and support tissue nourishment, healing, and growth. Each of these examples shows how porosity controls biological, geological, and environmental functionality on a massive scale.
In industrial and engineering applications, porosity becomes a critical performance tool. In filtration systems, porous membranes allow selective passage of particles and contaminants. In construction materials such as concrete, brick, and stone, porosity affects strength, freezeŌĆōthaw resistance, water absorption, and durability. High porosity can weaken structures if not properly managed, especially when water enters and expands during freezing conditions, causing cracking. In energy systems, porous electrodes in batteries and fuel cells enable efficient ion transport and chemical reactions, driving high performance in modern energy storage technology. Thermal insulation materials such as polyurethane foam, fiberglass, and mineral wool use closed pores to trap air and reduce heat transfer. Porosity also enhances catalytic materials by increasing surface area, enabling faster chemical reactions and higher efficiency in manufacturing processes.
The creation and control of pore structure is one of the most advanced areas of materials engineering today. Techniques such as 3D printing, freeze casting, foaming reactions, laser structuring, and solvent extraction allow precise adjustment of pore arrangement for customized performance. Porous scaffolds used in tissue engineering are designed with interconnected pores that support cell growth, nutrient flow, and bone regeneration. Aerospace and automotive industries use lightweight porous metals and ceramics to balance strength with reduced mass and improved thermal properties. Even emerging fields like hydrogen fuel storage, carbon capture, and smart textiles depend on engineered porosity.
Porosity reminds us that the internal world of materialsŌĆöoften hidden out of sightŌĆödetermines how they behave under real conditions. The presence and pattern of tiny spaces inside a solid can make the difference between a brittle rock and a water reservoir, between a light foam and a rigid panel, between a breathable membrane and an airtight barrier. As scientists and engineers continue to shape the internal architecture of materials with increasing precision, porosity becomes more than a passive characteristic; it becomes an active design parameter that influences strength, transport, durability, efficiency, and sustainability. By understanding pore structure, we gain deeper insight into how the smallest voids can define the performance of the largest systems in nature, technology, and everyday life.