Solvent, Solute, and Solution — Chemistry Mixture Concept
In chemistry, the concepts of solvent, solute, and solution form the foundational language through which we understand mixtures at the molecular level, and they reveal the deep interplay between matter, energy, and interaction. Although these terms at first appear simple—one substance dissolves, another does the dissolving, and the final mixture is the result—the processes they represent shape countless natural phenomena, industrial operations, biological systems, and everyday experiences. A solution is far more than a uniform mixture; it is a dynamic molecular environment where particles interact, disperse, stabilize, and respond to physical or chemical changes. Examining the roles of solutes and solvents requires exploring how their structures, polarities, intermolecular forces, and thermodynamic behaviors govern solubility, concentration, equilibrium, and the transformations that occur when substances mix. Solutions illustrate how the microscopic world influences macroscopic properties such as taste, conductivity, color, and reactivity. Through understanding these concepts, one gains insight into chemical processes ranging from digestion and respiration to pharmaceutical delivery, industrial manufacturing, and environmental chemistry.
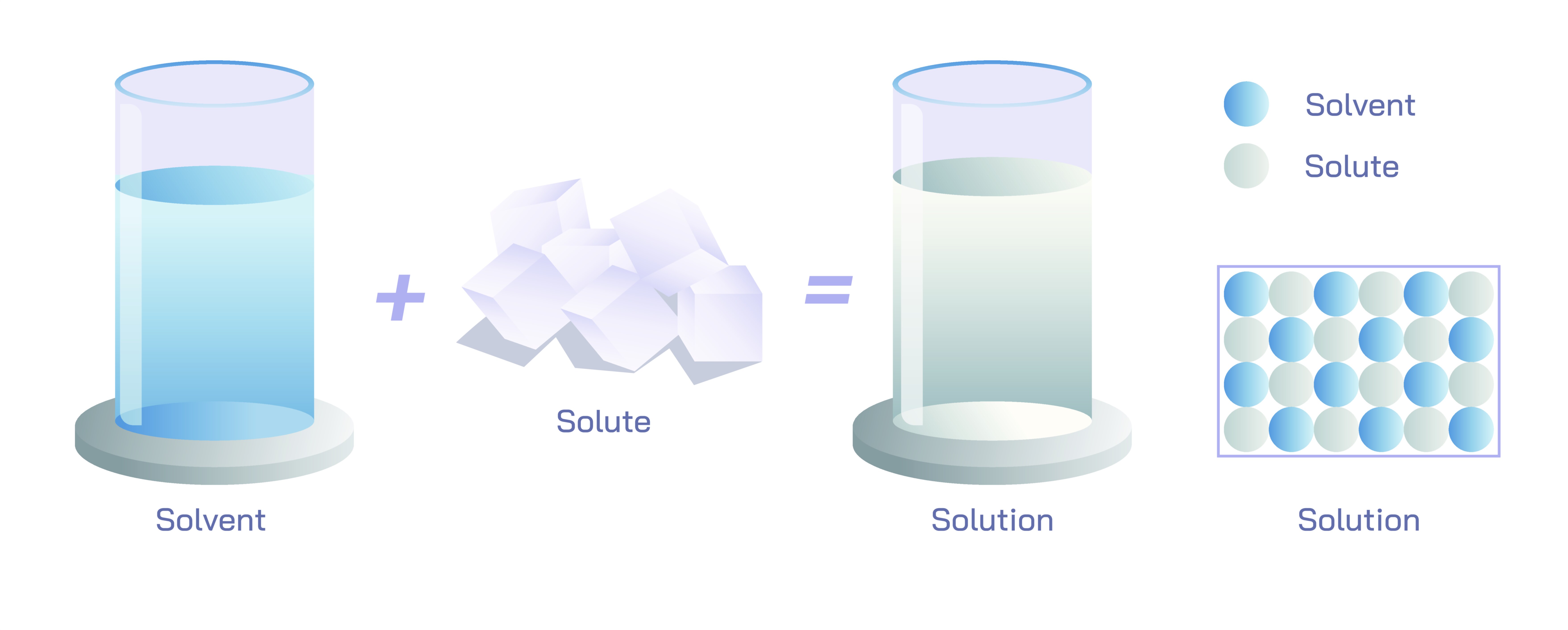
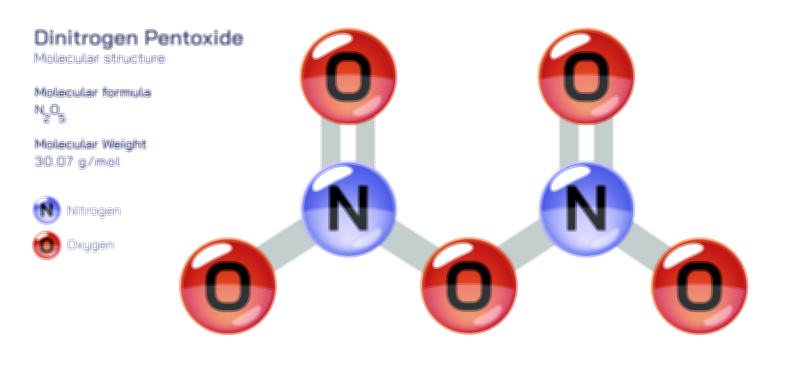
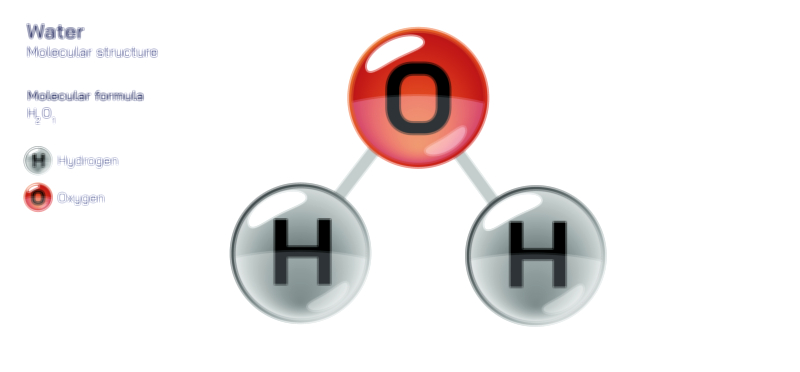
A solvent is the medium in which other substances dissolve, typically present in the largest quantity within a mixture. Its molecular characteristics determine the behavior and stability of the resulting solution. Solvents can be categorized by their polarity, acidity, molecular composition, and ability to stabilize ions or molecules. Water, the most universally used solvent, owes its extraordinary power to its polarity and capacity to form hydrogen bonds. Its bent molecular structure, with partially positive hydrogen atoms and a partially negative oxygen atom, allows it to surround charged or polar solute particles, pulling them apart and dispersing them uniformly. This unique property makes water the “universal solvent,” capable of dissolving enormous varieties of ionic and polar compounds. However, nonpolar solvents such as hexane, benzene, and ether serve equally important roles, especially in organic chemistry where they dissolve oils, fatty acids, hydrocarbons, and other nonpolar compounds that water cannot. The solvent ultimately determines which solutes dissolve, how fast dissolution occurs, what interactions take place within the mixture, and how the solution behaves under changing conditions.
The solute represents the substance that is dissolved into the solvent. Solutes can be gases, liquids, or solids, each interacting differently with the solvent’s molecules. In the case of solid solutes, dissolution typically involves breaking the lattice structure of the solid and dispersing individual ions or molecules throughout the solvent. For ionic compounds such as sodium chloride, water separates the ions and stabilizes them by forming hydration shells. For molecular solids like sugar, water disrupts the intermolecular attraction between molecules and encourages them to disperse. Gaseous solutes dissolve by interacting with the surface of the solvent, diffusing into it until equilibrium is reached; their solubility is influenced by temperature and pressure. Liquid solutes mix more readily with liquid solvents, often forming homogeneous mixtures when their polarities or intermolecular forces are compatible. Thus, the behavior of solutes depends on particle size, charge, molecular structure, and the nature of the solvent. Solutes influence properties such as vapor pressure, boiling point, freezing point, electrical conductivity, and color of the solution, demonstrating how even small quantities can significantly alter the macroscopic behavior of the mixture.
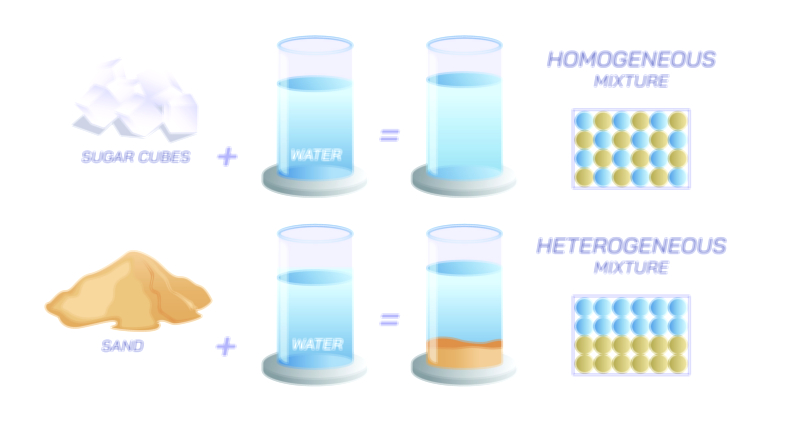
When a solute dissolves in a solvent to form a solution, the outcome is a homogeneous mixture where the particles are uniformly distributed at the microscopic level. This uniformity distinguishes solutions from heterogeneous mixtures, in which components remain visibly separate. A solution displays consistent properties throughout, meaning that any portion taken from the mixture contains solute particles in the same proportion as any other. The creation of a solution is governed by thermodynamic principles, particularly the balance between enthalpy and entropy. Dissolution may absorb or release heat depending on the strength of interactions formed or broken. Entropy, the measure of disorder, often favors the dispersal of solute particles, promoting solubility. When the energy required to separate solute particles and the energy needed to make space within the solvent are sufficiently compensated by the formation of new solute–solvent interactions, dissolution proceeds spontaneously. These subtle energy changes shape solubility limits, temperature effects, and concentration behavior.
One of the central ideas of solution chemistry is the relationship between polarity and solubility, often summarized through the principle “like dissolves like.” Polar solvents dissolve polar solutes because the solvent’s molecules can interact through dipole interactions or hydrogen bonding. Nonpolar solvents dissolve nonpolar solutes because their molecules interact through dispersion forces without competing polar interactions. This guiding principle informs countless applications: oils dissolve in hexane, alcohol dissolves in water, ionic salts dissolve in polar solvents, and many pharmaceuticals must be paired with appropriate solvents for effective delivery. The microscopic interactions between solute and solvent dictate whether dissolution occurs and how stable the resulting solution will be.
Another essential concept in solutions is concentration, which measures how much solute is present relative to the solvent. Solutions can range from extremely dilute, with very little solute, to saturated, where no additional solute can dissolve under the given conditions. The saturation point reflects equilibrium between dissolution and crystallization processes. If more solute is added beyond this point, it remains undissolved. Concentration affects virtually every property of a solution: higher concentrations can inhibit reactivity, alter freezing and boiling behavior through colligative properties, change osmotic balance, and influence rates of biological or chemical reactions. In medicine, precise concentrations ensure correct dosage, while in environmental chemistry, concentrations determine pollutant toxicity. Concentration units such as molarity, molality, mass percentage, mole fraction, and parts per million reflect the diverse contexts in which solutions are studied and applied.