Standard Hydrogen Electrode — Principles, Construction, and Electrochemical Use
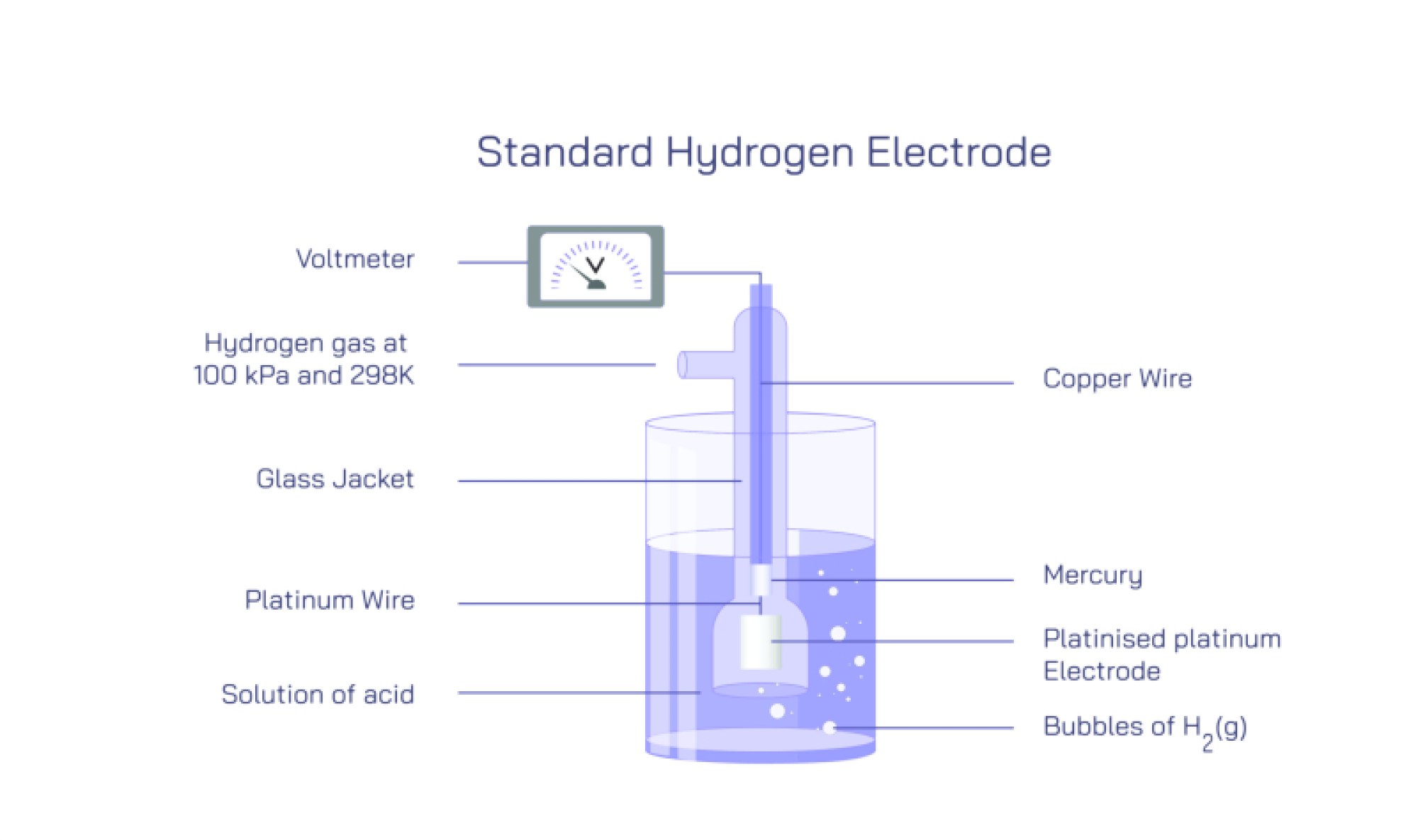
The Standard Hydrogen Electrode (SHE) is a reference electrode widely used in electrochemistry to measure the electrode potential of other electrodes. It is defined to have a potential of 0 volts under standard conditions, providing a benchmark for comparing and calculating the relative potentials of various half-cells in electrochemical cells. The SHE is fundamental in establishing standard electrode potentials and understanding redox reactions.
The construction of the standard hydrogen electrode involves a platinum electrode coated with a fine layer of platinum black to increase surface area and facilitate electron transfer. This electrode is immersed in an acidic solution, typically 1 M H⁺ ions, and hydrogen gas is bubbled over the platinum surface at a pressure of 1 atmosphere. The hydrogen ions in the solution can undergo reduction at the platinum surface, while hydrogen molecules can be oxidized to ions, establishing a dynamic equilibrium. This reversible reaction forms the basis of the SHE’s reference potential.
The working principle of the SHE relies on the half-cell reaction: hydrogen ions gain or lose electrons at the platinum surface, depending on the connected electrode. By coupling the SHE with another half-cell, the electromotive force (EMF) of the cell can be measured, and the potential of the other electrode can be determined relative to the SHE. It is widely used in laboratories and research to calibrate other electrodes and measure standard electrode potentials in a controlled, reproducible manner.
The standard hydrogen electrode is sensitive to conditions such as temperature, hydrogen ion concentration, and hydrogen gas pressure, which must be maintained at standard values for accurate measurements. It plays a critical role in electrochemistry education, research, and industrial applications, including batteries, corrosion studies, and electroplating processes. The SHE provides a reliable and consistent reference, enabling scientists to study electron transfer processes, reaction kinetics, and redox thermodynamics.
In conclusion, the standard hydrogen electrode is a reference half-cell used to measure electrode potentials in electrochemical cells. Its construction with a platinum electrode, hydrogen gas, and acidic solution allows for reversible redox reactions, establishing a zero-volt reference. Understanding SHE principles, construction, and applications is essential for electrochemistry, enabling accurate determination of redox potentials, calibration of electrodes, and analysis of electron transfer reactions in both research and industrial contexts.