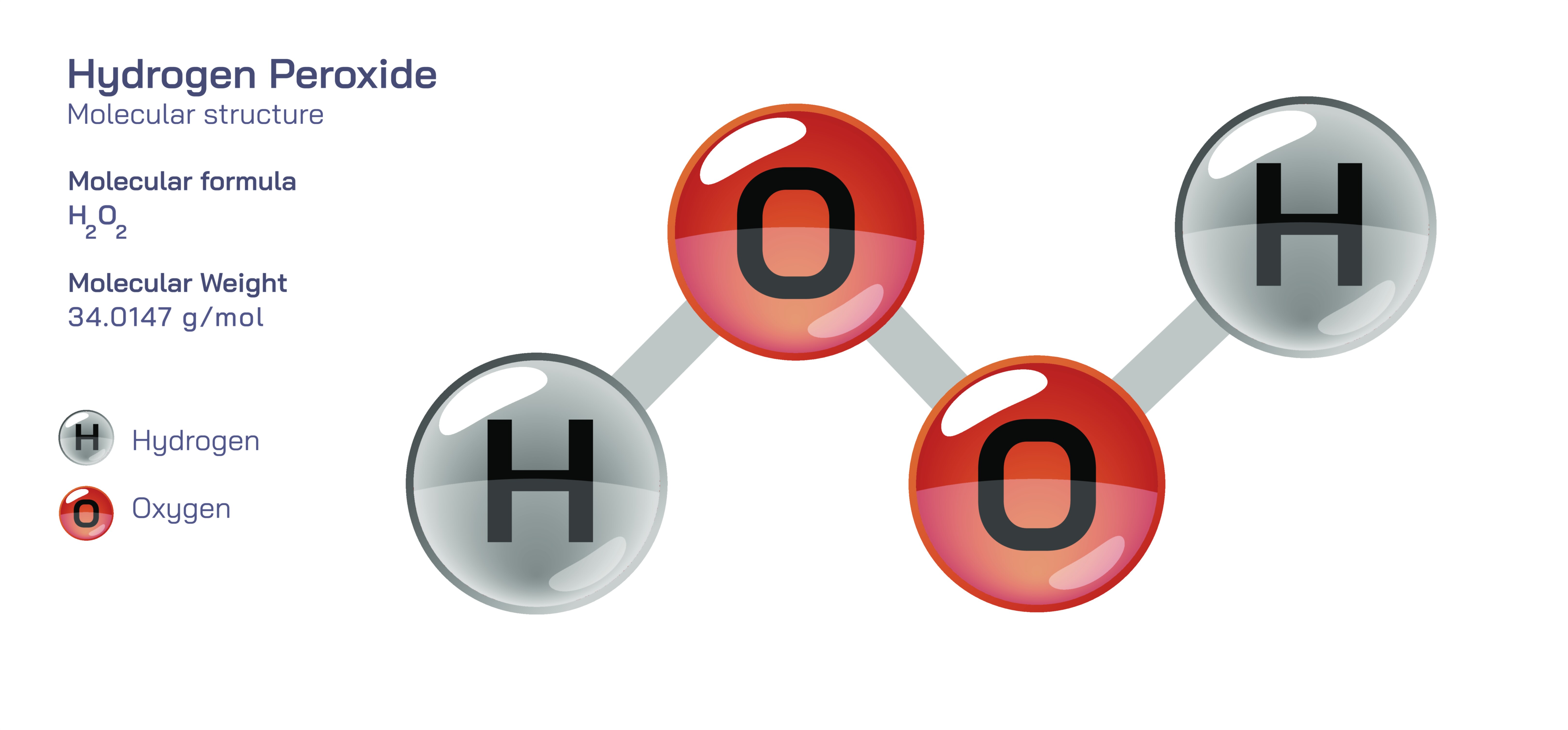
Hydrogen Peroxide Chemical Structure Explained
Hydrogen peroxide is a deceptively simple molecule whose properties and behavior cannot be understood solely by looking at the atoms that compose it, but rather by examining the subtle angles, bonding characteristics, and reactive tendencies that emerge from its structural arrangement. At first glance, the formula of hydrogen peroxide, H₂O₂, appears to suggest nothing more complex than a water molecule with an extra oxygen atom. However, its molecular geometry reveals far more than that. Instead of forming a linear or symmetrical shape, hydrogen peroxide adopts a nonplanar, twisted configuration in which the two oxygen atoms are connected through a single O–O bond and each oxygen is additionally bonded to a hydrogen atom. The bond between the two oxygen atoms is known as a peroxide bond, and this internal link is the key to almost all the chemical behavior that makes hydrogen peroxide unique. While the water molecule possesses an extraordinarily stable oxygen–hydrogen structure, hydrogen peroxide’s oxygen–oxygen bond is relatively weak and stores latent reactivity that becomes available under the right conditions, enabling the molecule to behave as either an oxidizer or a reducer depending on the surrounding chemical environment. This dual behavior, rooted in its structural instability, allows hydrogen peroxide to serve as a disinfectant, bleaching agent, industrial oxidant, biological signaling molecule, and, in high concentrations, a dangerously reactive chemical with the ability to decompose explosively.
The structural arrangement of hydrogen peroxide is best understood by considering the orientation of its atoms in three-dimensional space. Each oxygen atom holds two hydrogen atoms and two lone pairs of electrons, and the molecular electron repulsion forces create a shape in which the H–O–O–H framework twists rather than staying linear. This twist is not a trivial detail, because the torsion angle influences the molecule’s ability to participate in chemical reactions. It determines the ease with which the peroxide bond can break and governs the pathways through which hydrogen peroxide decomposes into water and oxygen. This decomposition does not demand violent conditions. Even mild heating, light exposure, metal contamination, or contact with certain surfaces can destabilize the peroxide bond. That instability provides the chemical foundation for hydrogen peroxide’s usefulness in oxidation reactions — a process in which the molecule accepts electrons during its conversion to water, thereby oxidizing other substances around it. This is the exact mechanism by which hydrogen peroxide eliminates bacteria, viruses, fungi, and organic stains, because it donates reactive oxygen species capable of attacking cell walls, genetic material, and protein structures. The molecule therefore becomes more than a structural entity; it becomes a biological and industrial tool shaped by the underlying tension stored in its oxygen–oxygen bond.
Although hydrogen peroxide can act as a strong oxidizer, its structure also allows for the opposite effect in certain reactions, enabling it to behave as a reducing agent. This dual ability makes hydrogen peroxide a versatile molecule, particularly in biochemical systems where redox balance determines cellular conditions. In the human body, hydrogen peroxide is not merely an external disinfectant but a naturally produced signaling compound used by immune cells to attack pathogens. At extremely controlled cellular levels, H₂O₂ serves as a communication molecule helping regulate inflammation and stress responses. However, when its concentration rises above the narrow margin tolerated by biological tissue, hydrogen peroxide becomes destructive by releasing radicals that damage DNA, proteins, and membranes. The same oxidative potential that makes it valuable in immunity also makes it potentially harmful if allowed to accumulate beyond the control of cellular antioxidant systems. The structural root of this behavior lies again in the weakness of the peroxide bond, which allows hydrogen peroxide to split with the release of highly reactive oxygen species.
Industrial and household formulations of hydrogen peroxide reflect this intrinsic reactivity in scaled forms. The 3% solutions commonly sold in pharmacies for wound cleansing and disinfecting represent a controlled version of the molecule’s oxidative potential, diluted to reduce tissue damage while still capable of destroying pathogens. Higher concentrations, such as those used for bleaching hair, whitening textiles, or treating industrial wastewater, become increasingly aggressive and must be handled carefully because of their ability to rapidly degrade organic matter. Very high concentrations, like those used in rocket propellants and chemical processing, highlight the full force of the molecule’s structural energy — hydrogen peroxide in these forms can decompose with explosive violence, releasing oxygen so rapidly that it produces massive heat and pressure. This dramatic escalation in power results from nothing more than the structural instability of the O–O bond amplified by concentration and catalytic activation. A seemingly simple molecule becomes a formidable oxidizing force when uncontrolled, demonstrating that the true story of hydrogen peroxide is written in the architecture of its bonds.
Storage and safety considerations for hydrogen peroxide exist because its structure predisposes it to reactivity. The molecule cannot be stored casually like water because even the smallest contamination — metal particles, dust, heat, or ultraviolet light — can accelerate decomposition. For this reason, hydrogen peroxide is sold in opaque containers that block light, and stabilizers are often added to slow spontaneous breakdown. These precautions ensure that the oxygen–oxygen bond remains intact until the molecule is intentionally used. In concentrated industrial settings, hydrogen peroxide must be kept in corrosion-resistant vessels and monitored continuously to prevent runaway reactions. Ironically, once hydrogen peroxide decomposes completely into water and oxygen, it becomes harmless. It is the transition — not the final products — that holds danger, because the decomposition process releases energy in a surge that can cause tissue damage or mechanical stress if uncontrolled.
The behavior of hydrogen peroxide therefore represents a profound lesson in chemistry: the function of a molecule is inseparable from its structure. A single oxygen–oxygen bond, weaker than most other covalent bonds and loaded with potential energy, defines the physical, chemical, and biological identity of H₂O₂. This bond is the engine behind its disinfecting power, bleaching capacity, industrial oxidation role, immune signaling involvement, decomposition risk, and even explosive potential. Hydrogen peroxide is neither inherently safe nor inherently dangerous; it is a molecule that magnifies the importance of control, environment, and concentration. In small, stabilized quantities, it is a valuable antiseptic and household cleaner. In industrial quantities, it becomes a precision tool for chemical engineering and material treatment. In uncontrolled or extreme concentrations, it becomes a hazard tied directly to the same structural weakness that gives it usefulness.
Understanding hydrogen peroxide means looking beyond its formula to the deeper blueprint encoded in its molecular structure. Its twisted configuration, its oxygen–oxygen bond, and its interplay between stability and reactivity make it one of the most striking examples in chemistry of how a simple arrangement of atoms can exhibit astonishing versatility. Hydrogen peroxide is a disinfectant, an oxidizer, a metabolic signal, an industrial reagent, and a potential explosive — all because of the way its atoms are connected. Structure becomes function, function becomes application, and application becomes either benefit or danger depending on how humans choose to harness the molecular energy held within H₂O₂.