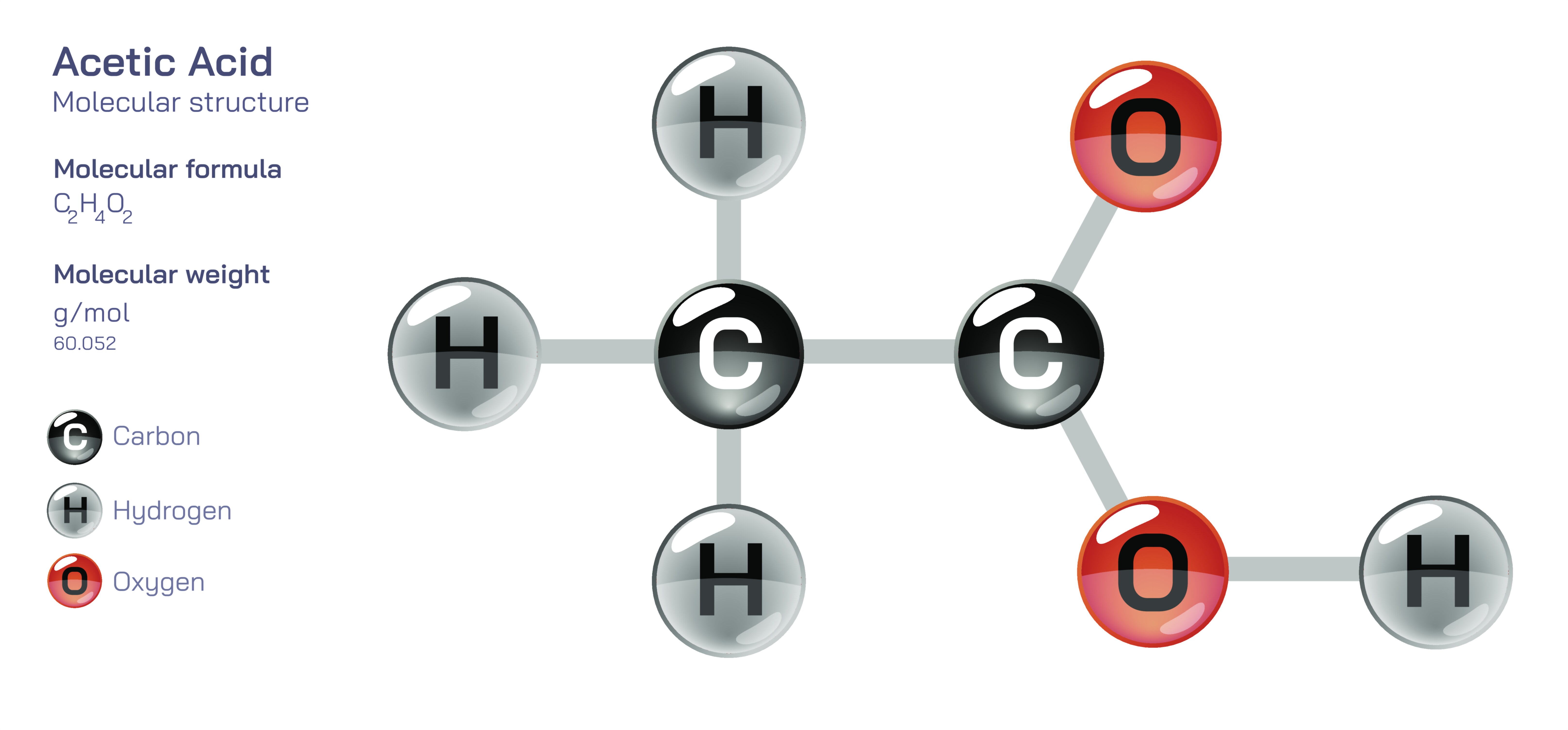
Acetic Acid — Organic Carboxylic Compound
Acetic acid is one of the most fundamental and widely recognized organic carboxylic compounds, forming the chemical and sensory backbone of vinegar while simultaneously serving as a cornerstone of organic chemistry, biochemistry, industry, and environmental processes. Known for its sharp, pungent odor and sour taste, acetic acid belongs to the family of carboxylic acids, characterized by the presence of the carboxyl functional group that imparts acidity and reactivity. Although commonly encountered in dilute form in households, pure acetic acid—often called glacial acetic acid—is a powerful, corrosive substance capable of forming crystals at cool temperatures, a physical property that underscores its structured nature. Its molecular structure, built upon a simple carbon framework, belies the profound influence it has across a wide spectrum of natural and human-made systems. Understanding acetic acid means appreciating not only its role in cooking or fermentation but also its biochemical significance, industrial applications, ecological presence, and the ways in which it contributes to life and technology.
At the molecular level, acetic acid is a small yet highly characteristic organic compound. Its structure consists of two carbon atoms, with one forming part of the functional carboxyl group and the other attached to three hydrogen atoms. This arrangement provides a distinctive combination of polarity and reactivity. The carboxyl group enables acetic acid to donate a proton, giving it acidic properties, while the rest of the molecule offers structural stability and compatibility with a wide variety of chemical environments. The strength of acetic acid as an acid is considered moderate—stronger than carbonic acid but weaker than mineral acids—making it useful in biological systems where harsh acidity would be harmful. In aqueous solutions, acetic acid exists in equilibrium between its undissociated and dissociated forms, allowing it to act as an effective buffer under the right conditions. This equilibrium contributes to its chemical versatility and highlights the sophistication of even the simplest organic acids.
Acetic acid is produced naturally through biological processes that rely on fermentation, oxidation, and metabolic pathways. One of the most familiar sources is the fermentation of ethanol by acetic acid bacteria, particularly species belonging to the genera Acetobacter and Gluconobacter. These microorganisms thrive in oxygen-rich environments, converting alcohol into acetic acid as part of their metabolic activity. This process is the basis of vinegar production, a tradition that spans thousands of years and cultures worldwide. Vinegar itself typically contains around four to eight percent acetic acid, complemented by trace compounds that provide flavor and complexity. The biological origins of acetic acid extend beyond food production; it appears in plant metabolism, microbial ecosystems, and even the human body. For organisms that metabolize fats and carbohydrates, acetic acid or its derivatives act as intermediates in essential pathways such as the Krebs cycle, demonstrating that this seemingly simple molecule plays a role in sustaining life at the cellular level.
In human metabolism, acetic acid appears in the form of acetyl groups, which are essential intermediates in energy production and biosynthetic processes. Acetyl coenzyme A, often referred to as acetyl-CoA, is one of the most important molecules in biochemistry, and its core structure is essentially an activated form of acetic acid. Through acetyl-CoA, acetic acid becomes integrated into pathways that generate ATP, synthesize fatty acids, and regulate cellular functioning. The fact that such a foundational molecule is rooted in the chemistry of a simple acid highlights the elegance of biological design. Every breath taken, every movement made, and every metabolic function performed depends, in part, on the chemical logic anchored in the acetic acid framework. This deep connection underscores how organic chemistry forms the backbone of biological complexity.
Beyond its biochemical significance, acetic acid is vital in numerous industrial processes. Its ability to interact with alcohols, metals, bases, and various organic molecules makes it a flexible starting material for synthesizing a wide range of compounds. One major industrial product derived from acetic acid is vinyl acetate monomer, used in the creation of adhesives, paints, packaging materials, and textiles. Acetic acid is also used in producing solvents, plasticizers, cellulose acetate, and acetic anhydride—an important reagent in chemical manufacturing. In the textile industry, acetic acid plays a role in dyeing processes, helping set dyes and adjust fabric pH. In the food industry, its antimicrobial properties make it a natural preservative, helping inhibit the growth of spoilage bacteria and molds. Because it is relatively mild compared to strong mineral acids, acetic acid is often used for cleaning, descaling, and sanitizing purposes in both commercial and household settings.
The environmental role of acetic acid also deserves attention. It appears naturally in soils, waters, and atmospheric systems due to its formation from decomposition, fermentation, and oxidation processes. Microorganisms readily consume acetic acid, making it a key intermediate in the breakdown of organic matter and the recycling of carbon through ecosystems. In wetland environments, acetic acid serves as a substrate for methanogenic microorganisms that convert it into methane—a gas with both ecological and climatic significance. Understanding how acetic acid flows through natural cycles helps scientists study ecological interactions, carbon flux, and the effects of human activity on natural systems.
Acetic acid has long had cultural and historical importance, especially through its association with vinegar. Ancient civilizations used vinegar not only as a seasoning and preservative but also as a medicinal remedy, cleaning agent, and ritual substance. Its ability to dissolve mineral deposits, inhibit bacterial growth, and modify flavors made it valuable throughout human history. In traditional medicine, vinegar solutions were sometimes used to clean wounds, treat infections, or soothe skin conditions. Although modern medicine uses more advanced techniques, the historical reliance on vinegar reflects early recognition of acetic acid’s antimicrobial and chemical properties. Many traditional culinary practices also highlight the versatility of acetic acid, from pickling vegetables to marinating meats and balancing the flavors of sauces and dressings.
In modern science and medicine, acetic acid continues to play significant roles. Dilute acetic acid solutions are used in medical examinations, where they help highlight abnormal tissues during certain diagnostic procedures. In laboratory settings, acetic acid helps fix biological specimens, preserve tissues, and maintain stable pH environments for analysis. Its predictable behavior and compatibility with a wide array of reagents make it a standard component in research and experimentation. Its presence in pharmaceuticals, laboratory formulations, and biotechnological processes reflects its continued relevance in advancing scientific knowledge and practical innovation.
However, concentrated acetic acid must be handled with caution. Glacial acetic acid is corrosive, capable of causing burns on skin contact and releasing strong vapors that irritate the eyes and respiratory system. Safe handling requires appropriate protective equipment and ventilation. Despite these hazards, acetic acid remains safer and more environmentally compatible than many industrial chemicals, as it biodegrades readily and has relatively low toxicity at dilute concentrations. This balance between utility and safety contributes to its wide acceptance in science, industry, and daily life.
Acetic acid also holds significance in the chemistry classroom, where it serves as a gateway to understanding functional groups, acid–base behavior, molecular polarity, and organic synthesis. Students encounter acetic acid early in their education because it illustrates fundamental principles in an accessible way. Through its study, they learn how structure influences function, how organic molecules react, and why carboxylic acids are central to both living processes and chemical manufacturing. The familiarity of vinegar gives acetic acid a relatable presence, transforming abstract principles into tangible concepts grounded in everyday experience.
Ultimately, acetic acid exemplifies how a modest organic molecule can hold extraordinary importance across disciplines. It bridges chemistry and biochemistry, tradition and technology, food and industry, biology and ecology. Its structure is simple, yet its influence is vast. Whether participating in the metabolic pathways of living cells, flavoring a meal, supporting industrial synthesis, preserving food, enabling scientific research, or flowing through natural ecosystems, acetic acid is an essential and ubiquitous component of the world. Understanding it reveals not only the interconnectedness of chemical processes but also the elegance with which simple molecular frameworks contribute to the complexity of life and human innovation.