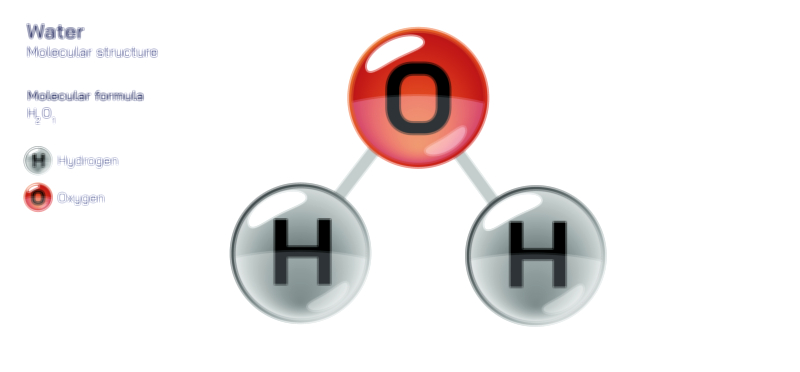
Hydroxide Ion Structure — OH⁻ Chemical Diagram and Properties Explained
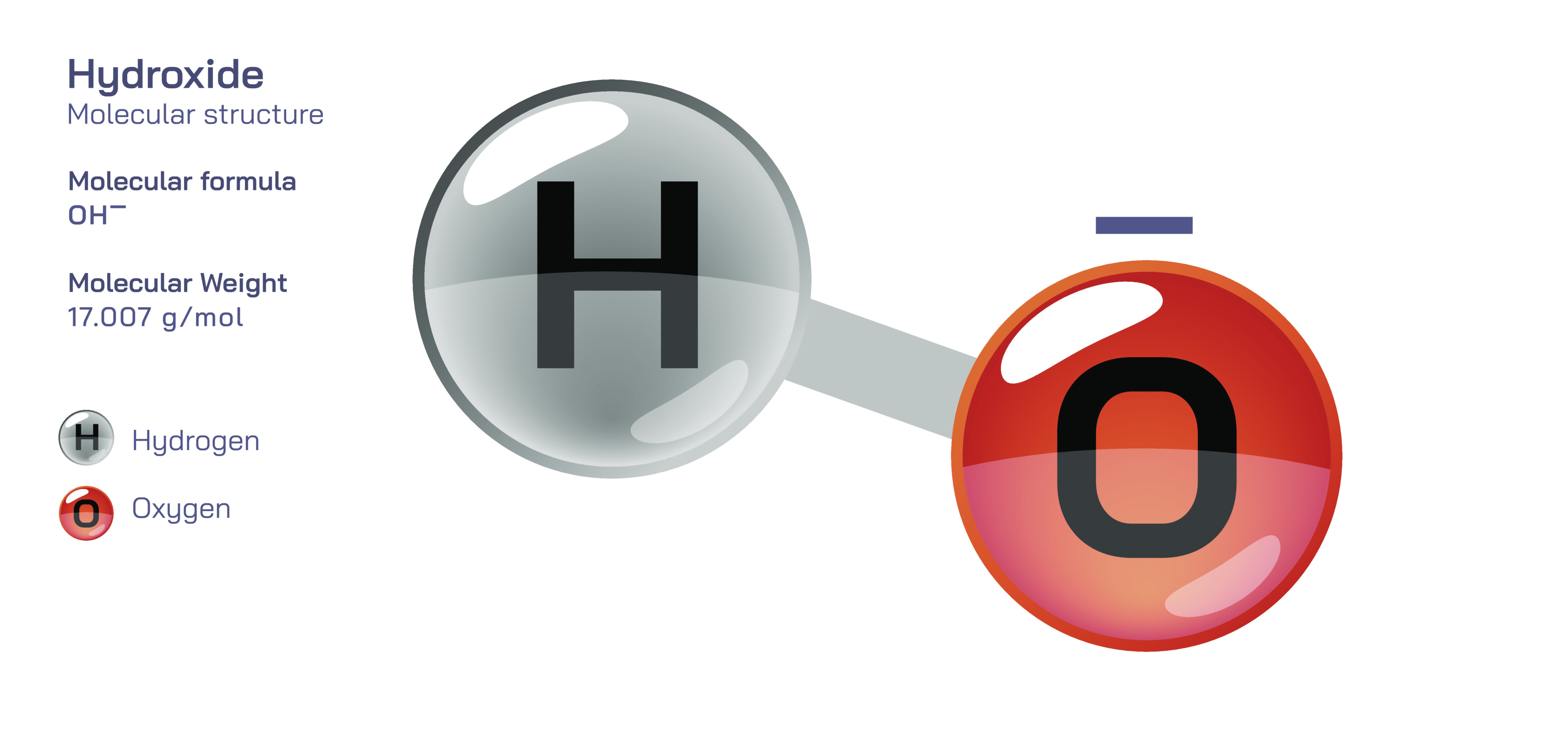
The hydroxide ion, written as OH⁻, is one of the most fundamental species in chemistry, yet its importance extends far beyond the simplicity of its formula. It is central to the concept of alkalinity, acid–base balance, water chemistry, biological systems, and countless industrial processes. Although the hydroxide ion is typically introduced as “water minus a hydrogen ion,” its deeper identity lies in the rearrangement of electrons that occurs when a water molecule loses a proton. The result is a diatomic ion made of one oxygen atom covalently bonded to one hydrogen atom, carrying a net negative charge that dictates its behavior in both chemical reactions and aqueous environments. To understand hydroxide fully, it is essential to examine its structural geometry, electronic distribution, hydrogen-bonding ability, and the consequences of these features on chemical reactivity and equilibrium.
The hydroxide ion originates from the water molecule, H₂O, which has a bent geometry due to the lone pairs on oxygen. When one proton (H⁺) is removed, what remains is OH⁻ — a structure consisting of the oxygen atom still bonded to a single hydrogen but now possessing three lone pairs instead of two. This added lone pair increases the electron density on the oxygen atom, making the negative charge strongly localized. As a result, the hydroxide ion is not neutral or symmetrical like water; it is highly polar and strongly nucleophilic. The negative charge concentrates primarily on the oxygen because oxygen is far more electronegative than hydrogen, meaning it attracts electrons much more strongly. This uneven distribution of electron density is the reason the hydroxide ion is chemically aggressive in accepting protons and reacting with electrophilic species. It is eager to share its excess electron density, allowing it to form new covalent bonds or restore neutrality by capturing a proton and reforming water.
The structural simplicity of OH⁻ belies its immense influence in acid–base chemistry. In aqueous solutions, the hydroxide ion is the complementary partner to the hydronium ion (H₃O⁺), forming the basis of the pH scale. The presence of hydroxide ions makes a solution alkaline, and their concentration determines how strongly basic the solution is. When an acid is added to water, the acid donates protons, which react with hydroxide ions to form water. Conversely, when a base dissolves in water and increases the concentration of OH⁻ ions, the solution becomes more alkaline and can neutralize acids. The dynamic balance between H₃O⁺ and OH⁻ is what gives water its buffering ability — even pure water contains extremely small but equal amounts of both ions due to the natural autoionization of water molecules. This equilibrium ensures that water remains electrically neutral over a wide range of temperatures and concentrations.
Chemically, the hydroxide ion acts as a powerful nucleophile and a strong base. As a nucleophile, it readily attacks positively polarized atoms, especially carbon centers in organic molecules, leading to substitution and elimination reactions that form the basis of many synthesis processes. In biological systems, hydroxide plays a role in metabolic pathways, enzyme activity, and the structural integrity of biomolecules, although living organisms tightly regulate its concentration because excess OH⁻ can denature proteins, disrupt hydrogen bonding, and destabilize cellular membranes. As a base, hydroxide reacts intensely with acids. The reaction is energetically favorable because it leads to water formation — a highly stable state. This is why adding OH⁻ to an acidic solution immediately raises the pH, and why strong bases such as sodium hydroxide dissolve to release hydroxide ions directly into solution.
In real aqueous environments, hydroxide ions do not float independently for long; they participate in hydrogen-bond networks. Water molecules surround the ion, forming what is called a hydration shell. The negative charge on oxygen attracts the partial positive charges on water’s hydrogen atoms, creating a dynamic cluster that shields and stabilizes the ion. This feature is essential in understanding how hydroxide migrates through solution: instead of drifting as a single molecule, it moves by rapidly exchanging hydrogen bonds through a mechanism that transfers the extra negative charge along a chain of water molecules. This charge-hopping process — similar to the proton-hopping process that governs hydronium transport — is responsible for the unusually high mobility of OH⁻ in water. The hydroxide ion’s hydrogen-bonding ability also influences solubility; ionic substances that produce OH⁻ dissolve readily in water because the hydration process makes the dissolution energetically favorable.
The hydroxide ion plays critical roles across industrial fields as well. It is used for neutralizing acids, manufacturing soaps through saponification, purifying metals in processing plants, adjusting pH in wastewater treatment, cleaning surfaces, and breaking down organic matter in chemical reactions. Its high reactivity makes it both powerful and potentially dangerous. Concentrated hydroxide solutions are corrosive to skin, metals, and organic materials because the OH⁻ ion disrupts molecular structures by breaking chemical bonds and drawing protons away from biological tissues. This property reflects the same chemical motive that governs its role in simple acid–base reactions: hydroxide tends to remove protons wherever possible to restore its neutral form as water. Because biological systems rely on precise hydrogen-bonding and proton-controlled environments, unregulated exposure to hydroxide is harmful and can quickly degrade tissues.
Despite its reactive nature, the hydroxide ion is not inherently destructive — it is essential to life, technology, and environmental balance. In the human body, natural buffering systems regulate OH⁻ concentrations to maintain physiological pH, ensuring that cells, proteins, and enzymes operate in stable chemical conditions. In environmental systems, hydroxide contributes to mineral weathering and soil composition and plays a role in natural water alkalinity. In technology, it enables some of the most important manufacturing processes that sustain modern society. The same reactivity that allows hydroxide to break chemical bonds also allows chemists to build new ones in controlled environments.
Ultimately, the hydroxide ion illustrates how a molecule with a simple composition can become foundational to both chemical theory and real-world function. Its structure — a hydrogen atom bonded to an oxygen carrying excess electron density — is the source of every property it exhibits, whether acting as a proton acceptor, a nucleophile, a ligand in coordination chemistry, or a component of the pH scale. The OH⁻ ion shows that the fundamental behaviors of chemistry emerge not from complexity but from the precise arrangement of atoms and electrons in even the smallest species. Through its unique structure and properties, the hydroxide ion stands as a perfect example of how microscopic molecular identity governs macroscopic chemical behavior across nature, biology, and industry.