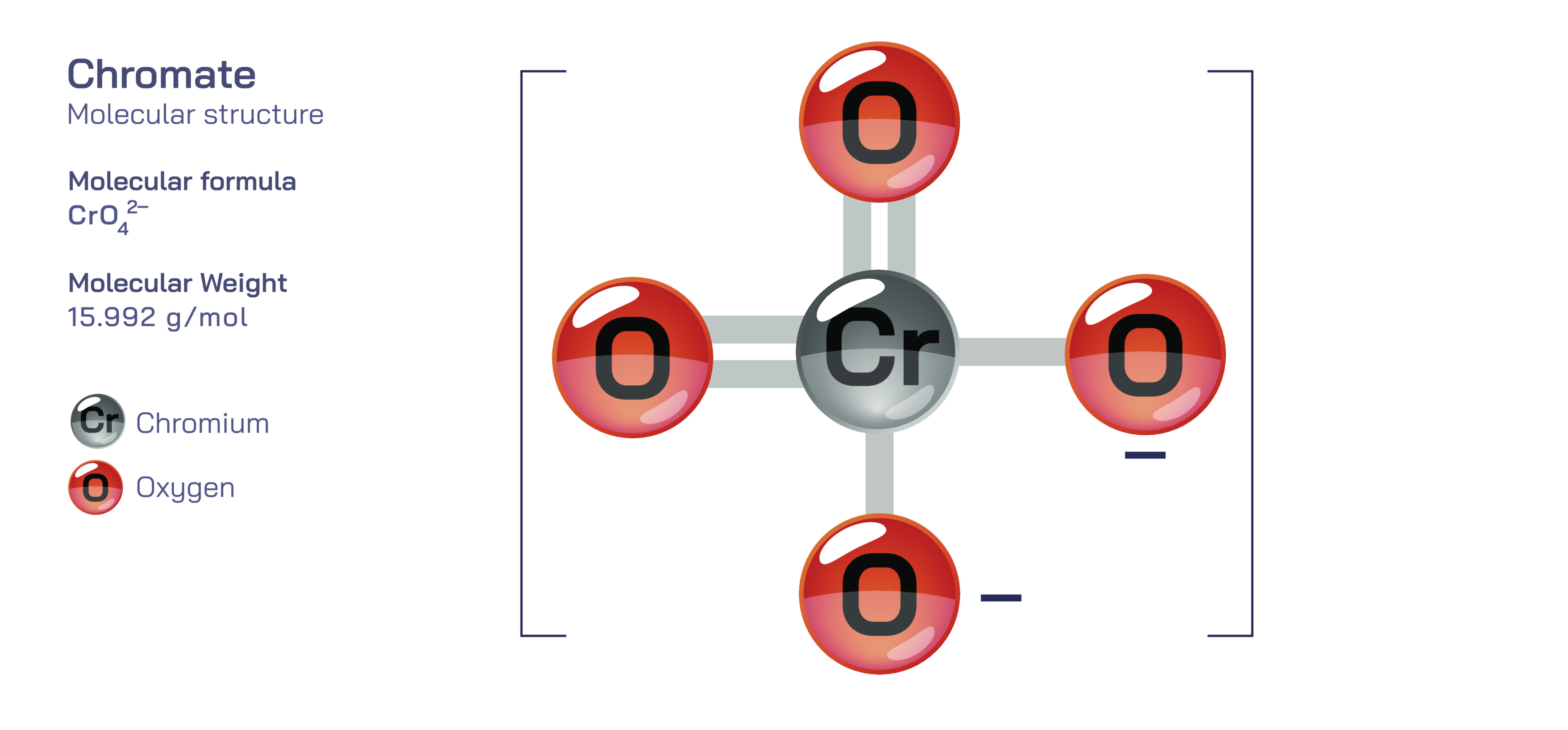
Chromate — Inorganic Oxyanion Compound
Chromate is an inorganic oxyanion compound that occupies a significant place in environmental chemistry, industrial processes, analytical techniques, metallurgy, corrosion science, and material formulations. Consisting of a chromium atom in its hexavalent oxidation state bonded to four oxygen atoms, the chromate ion forms a tetrahedral structure that reflects the high oxidation power intrinsic to chromium(VI) species. This geometry, though deceptively simple in appearance, determines the compound’s reactivity, solubility, toxicity, and capacity to participate in a wide variety of chemical transformations. The chromate ion is typically represented as CrO₄²⁻, often forming brightly colored yellow salts that dissolve readily in water and react strongly with both organic and inorganic substances. Its behavior in aqueous environments, its interactions with reducing agents, its ability to convert into dichromate and other chromium species, and its profound environmental and biological impact make chromate a compound of exceptional scientific importance. To understand chromate is to explore the subtle interplay between oxidation states, structural chemistry, redox reactions, industrial applications, and environmental stewardship, all of which reveal how a single oxyanion can shape technological advancements and ecological challenges.
At the molecular level, chromate’s tetrahedral structure forms the conceptual foundation for its chemical personality. The chromium atom sits at the center of the molecule in its +6 oxidation state—one of the highest and most reactive oxidation states accessible to transition metals. This heightened oxidation state imbues chromate with strong electron-accepting ability, making it a powerful oxidizing agent capable of driving energetic redox reactions. Because chromium(VI) is such an electron-deficient species, chromate ions readily participate in reduction processes that convert them into chromium(III), a more stable and biologically compatible oxidation state. This redox transformation lies at the heart of chromate’s environmental behavior, toxicological effects, and industrial utility. The tetrahedral symmetry also affects its spectroscopic properties, giving chromate its characteristic yellow coloration and making its electronic transitions easy to detect in analytical chemistry.
Chromate commonly exists in equilibrium with dichromate (Cr₂O₇²⁻), another chromium(VI) oxyanion. The relationship between chromate and dichromate is governed by pH: under alkaline conditions, chromate predominates, while under acidic conditions, dichromate forms as the dominant species. This interconvertibility demonstrates how the same chemical system shifts depending on proton availability, an important feature that influences chromate’s behavior in natural waters, industrial waste streams, soil environments, and biological systems. Because of this equilibrium, chromate and dichromate often appear together in real-world contexts, and their chemical and environmental profiles are closely intertwined.
In industrial applications, chromate has been widely used for its oxidizing, anticorrosive, and colorant properties. Chromate salts play a key role in metal passivation processes, especially in protecting aluminum, zinc, and magnesium against corrosion. Chromate conversion coatings form thin protective layers by reacting with metal surfaces, producing films that adhere strongly and resist environmental degradation. These coatings have been historically prized in aerospace, automotive, construction, and electronics industries because they offer durable protection while also enhancing paint adhesion. However, due to environmental and health concerns, many sectors have shifted toward chromium-free alternatives, though chromate-based coatings remain in use in specialized contexts where long-term stability is essential. In addition to corrosion control, chromate compounds have served as pigments, giving rise to vibrant yellow, orange, and red colors historically used in paints, ceramics, and pottery glazes. While visually striking, these pigments have declined in popularity due to concerns about chromium(VI) toxicity, prompting the development of safer substitutes.
Chromate also plays a critical role in analytical chemistry, where its strong oxidizing power facilitates quantitative titrations and classical qualitative tests. In redox titrations, chromate serves as an indicator or reactant in procedures that determine chloride content, oxidizable organic matter, and various metal ions. Its distinctive color changes allow scientists to detect endpoints visually, providing a simple yet effective method for analysis. Chromate’s spectroscopic properties make it useful in UV-visible studies, where its absorption peaks provide insight into electronic transitions and allow precise measurement of chromium(VI) concentrations in environmental samples. These analytical applications reflect how chromate bridges theoretical chemistry and practical measurement, offering tools that have supported generations of researchers and technicians.
In environmental science, chromate represents both a challenge and an opportunity for understanding contaminant behavior. Chromium(VI) species such as chromate are highly mobile in groundwater due to their negative charge and solubility. They can travel long distances through soil and aquifers, posing risks to ecosystems and human health. Once introduced—whether through industrial effluents, improper waste disposal, mining activities, or natural geological sources—chromate can persist unless actively transformed. The primary pathway for environmental remediation involves reducing chromate to chromium(III), which precipitates as insoluble hydroxides or forms stable complexes that are far less toxic and mobile. Techniques such as chemical reduction, bioremediation using specialized microorganisms, and adsorption onto iron-bearing minerals all exploit this natural redox shift to manage contamination. The persistence of chromate in water systems has led to extensive research on its environmental chemistry, including its interactions with organic matter, clay minerals, surface oxides, and natural reducing agents.
The toxicity of chromate derives from its strong oxidizing power and ability to penetrate biological systems. Chromate ions can enter cells through sulfate transport pathways due to their structural similarity to sulfate ions. Once inside, their oxidizing activity triggers the formation of reactive intermediates that damage proteins, DNA, and cellular structures. This property underlies chromate’s classification as a carcinogenic and mutagenic substance, prompting strict regulations on its handling, disposal, and permissible concentration in drinking water. Occupational exposure to chromate-containing dusts, mists, or fumes historically affected workers in construction, manufacturing, and plating industries, leading to respiratory issues, skin irritation, and long-term health complications. Modern safety protocols, protective equipment, and regulatory standards now minimize exposure in industrial environments, reflecting scientific understanding of chromate’s hazards and commitment to protecting human health.
Despite its toxicity, chromate also participates in beneficial natural and engineered processes that leverage its redox behavior. Microorganisms capable of reducing chromium(VI) to chromium(III) demonstrate an ecological mechanism for detoxification, contributing to natural attenuation in contaminated sites. These biological reduction pathways form the basis of bioremediation strategies in which microbial communities are stimulated to transform chromate into safer forms. Additionally, chromate participates in photochemical reactions in the environment, where sunlight-driven processes can influence its oxidation state and mobility. Understanding these pathways helps environmental scientists model pollutant behavior and design remediation methods that reflect real-world chemical dynamics.
In material science, chromate’s ability to form complex, durable coatings has influenced alloys, catalysts, and specialty materials. Chromate-treated metals have been used in aircraft structures, electronics casings, and high-reliability components where failure could have severe consequences. Although alternative technologies continue to emerge—such as trivalent chromium coatings, sol-gel materials, and organic-inorganic hybrid barriers—chromate remains a benchmark for corrosion protection, setting standards for performance that new materials strive to match. This enduring relevance reflects the depth of chemical insight that chromate-based systems provide, demonstrating the delicate balance between effectiveness and environmental responsibility in modern engineering.
The compound also holds historical significance. Before widespread knowledge of its hazards, chromate pigments appeared in iconic artworks, contributing vivid yellows to paintings in the nineteenth and early twentieth centuries. These pigments influenced artistic movements but also posed risks to artists and conservators, prompting ongoing efforts in art restoration to stabilize chromate-based materials and prevent degradation. Its industrial uses shaped the growth of metal finishing, synthetic dye production, and chemical processing during the industrial revolution, leaving a legacy that continues to inform regulatory frameworks and chemical safety standards.
Ultimately, chromate embodies the dual nature of inorganic chemistry—offering extraordinary utility while demanding rigorous respect. Its simple tetrahedral structure belies a powerful oxidative character that influences corrosion resistance, analytical techniques, industrial synthesis, environmental fate, and toxicology. As a compound that both advances technology and challenges environmental stewardship, chromate serves as a reminder that understanding chemical behavior is essential for making informed decisions about how substances are used, managed, and replaced. Through its reactivity, versatility, and complex interactions with natural systems, chromate continues to play a central role in the scientific, industrial, and environmental narrative of modern inorganic chemistry.