Lithium Hydroxide — Chemical Structure, Properties, Industrial Significance, and Applications in Energy Storage and Atmospheric Purification
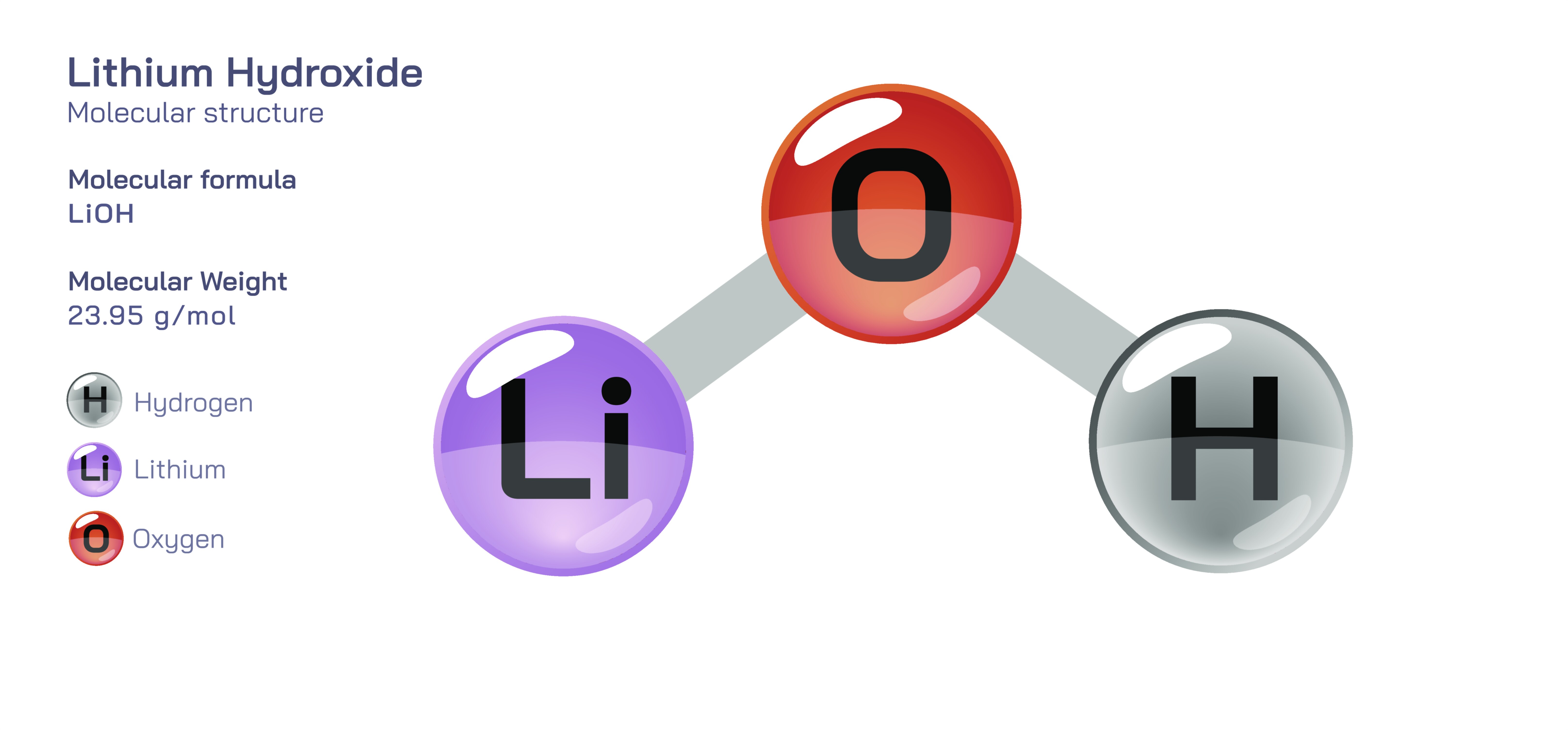
Lithium hydroxide is an inorganic compound with the formula LiOH, composed of lithium ions (Li⁺) bonded to hydroxide ions (OH⁻). Although chemically simple, it has become one of the most strategically important industrial chemicals in the world, largely due to its indispensable role in modern rechargeable lithium-ion batteries, aerospace life-support systems, and high-performance lubricants. Lithium hydroxide showcases how the behaviour of one of the lightest metallic elements can be converted into large-scale technological value through precise chemical interactions. Whether used to enhance the performance of electric vehicles, to maintain breathable atmospheres in space missions and submarines, or to withstand extreme mechanical stress in industrial applications, LiOH demonstrates a unique combination of reactivity, stability, and selectivity that distinguishes it from other alkaline hydroxides.
Structurally, lithium hydroxide is an ionic compound, consisting of Li⁺ bonded electrostatically to OH⁻. The small ionic radius of lithium gives the compound distinctive thermodynamic properties compared with heavier alkali hydroxides. Solid LiOH exists typically as either the anhydrate (LiOH) or the more commonly encountered monohydrate (LiOH·H₂O), where one water molecule becomes part of the lattice. The dehydrating and rehydrating processes between these forms occur readily because lithium ions possess a strong affinity for polar water molecules. This high degree of hydration affinity influences solubility, heat release during dissolution, and the ability of lithium hydroxide to remove acidic gases such as carbon dioxide from air — a feature that makes it invaluable in atmospheric purification systems.
Lithium hydroxide dissolves readily in water, producing a strongly alkaline solution that releases heat due to high hydration energy. In solution, Li⁺ becomes tightly surrounded by several layers of water molecules, creating stable hydrate complexes. Because lithium ions interact more strongly with water than sodium or potassium ions, LiOH has a stronger ability to retain moisture and form stable gels, a property that contributes to its usefulness in heat-resistant greases. As a solid, lithium hydroxide is white and crystalline, with a melting point that reflects the strength of ionic interactions in the lattice. Heating converts the monohydrate to anhydrous LiOH and, at even higher temperatures, decomposes the hydroxide into lithium oxide (Li₂O) while releasing water vapour — a behaviour that underlies its use in high-temperature ceramic and metallurgical processes.
The global importance of lithium hydroxide has risen sharply due to its essential role in lithium-ion battery chemistry. Cathode materials in many high-performance rechargeable batteries — particularly nickel-rich compositions such as NCM (nickel-cobalt-manganese) and NCA (nickel-cobalt-aluminium) — require lithium hydroxide for their synthesis. Compared to lithium carbonate, another lithium salt used in battery material production, lithium hydroxide reacts more efficiently with transition-metal oxides at lower temperatures and produces superior cathode uniformity. These characteristics directly influence charging speed, thermal stability, capacity retention, and cycle lifespan of advanced battery systems. Electric vehicles, grid-scale renewable-energy storage, aerospace power units, and portable electronics increasingly rely on cathodes derived from LiOH due to their ability to support high-density energy storage without rapid degradation. As the world shifts toward electrified transportation and large-scale energy storage solutions, lithium hydroxide has become a strategic raw material in global energy supply chains.
Beyond batteries, lithium hydroxide is widely used in air purification and life-support systems, particularly in enclosed environments where carbon dioxide accumulation poses a threat. LiOH absorbs CO₂ through a strong acid-base reaction that forms lithium carbonate (Li₂CO₃) and water. Because it performs this conversion rapidly, cleanly, and without requiring electrical input, lithium hydroxide has historically been used in submarines, spacecraft, underwater habitats, industrial respiratory systems, and emergency oxygen equipment. Its role became well known during NASA’s Apollo 13 mission, when the safe return of astronauts depended on efficient carbon-dioxide scrubbing using LiOH canisters. This characteristic continues to make lithium hydroxide a critical material for space exploration, naval operations, and emergency response technologies where breathable air must be maintained in sealed environments.
Lithium hydroxide is also indispensable in the production of lithium-based greases, especially lithium stearate greases, which dominate global industrial lubrication. These greases are valued for their water resistance, mechanical stability, low-temperature flexibility, and ability to retain lubricating performance under high heat and heavy mechanical loads. As a thickening agent, lithium hydroxide reacts with fatty acids to form soaps that bind and stabilize base oils. Such greases are used in automotive components, heavy machinery, ball bearings, wind-turbine gears, aircraft systems, mining equipment and marine hardware — environments where lubricants must withstand both temperature extremes and mechanical shock. The growth of manufacturing, transportation, renewable-energy systems and heavy-industry sectors has maintained lithium hydroxide as a critical feedstock for lubrication chemistry even as the battery market continues to expand.
In addition to these major uses, lithium hydroxide appears across other specialized industries. In ceramics and glass manufacture, it modifies fusion characteristics and improves resistance to thermal shock. In alkaline fuel-cell technologies, it is used as a catalyst and electrolyte component in research systems aimed at sustainable energy conversion. In polymer and chemical synthesis, lithium hydroxide serves as a strong base with predictable selectivity, allowing controlled deprotonation reactions and polymer modifications. In corrosion-control applications, LiOH can be added to industrial fluids to maintain alkaline environments that reduce metal oxidation, especially in power-plant cooling systems. Because the lithium ion has unique solvating behaviour compared with other alkali ions, lithium hydroxide often provides performance advantages that cannot be matched by sodium or potassium hydroxide in many industrial processes.
Handling lithium hydroxide requires careful precautions because it is highly caustic. Contact with skin or eyes can cause burns, and accidental inhalation of dust or ingestion is hazardous. Because lithium hydroxide absorbs water and CO₂ from the atmosphere, it must be stored in airtight containers to prevent degradation and to maintain purity for use in high-precision applications like battery cathode production. Industrial processes therefore involve moisture-controlled environments, corrosion-resistant storage materials, and safety measures to prevent direct human exposure. Despite its hazards as a strong base, controlled handling ensures that lithium hydroxide can be used safely in large-scale installations ranging from automotive factories to space-flight life-support systems.
The wide variety of applications that rely on lithium hydroxide — from carbon-dioxide scrubbing systems deep in the ocean or high above Earth, to high-performance lithium-ion batteries powering the transition to sustainable transportation, to lubrication systems supporting heavy industry — demonstrates how a single chemical compound can transform multiple technological fields. Its performance is rooted in its chemical structure: the small Li⁺ cation binds strongly to water, coordinates predictably with oxygen atoms, forms stable soaps and ceramics, and reacts efficiently in battery precursor synthesis. These microscopic properties scale upward into global technological influence.
In summary, lithium hydroxide stands as a material that connects chemistry directly to the modern world’s most crucial engineering achievements. Its ability to stabilize and enhance battery cathodes supports the expansion of electric mobility and renewable-energy storage. Its CO₂-absorption behaviour sustains life in confined environments where atmospheric control is not optional. Its lubrication chemistry protects the machinery that drives industrial progress. Through its structure, reactivity, and exceptional selectivity, LiOH has become far more than a simple hydroxide — it is a cornerstone of aerospace safety, industrial reliability, and the global shift toward sustainable energy systems.