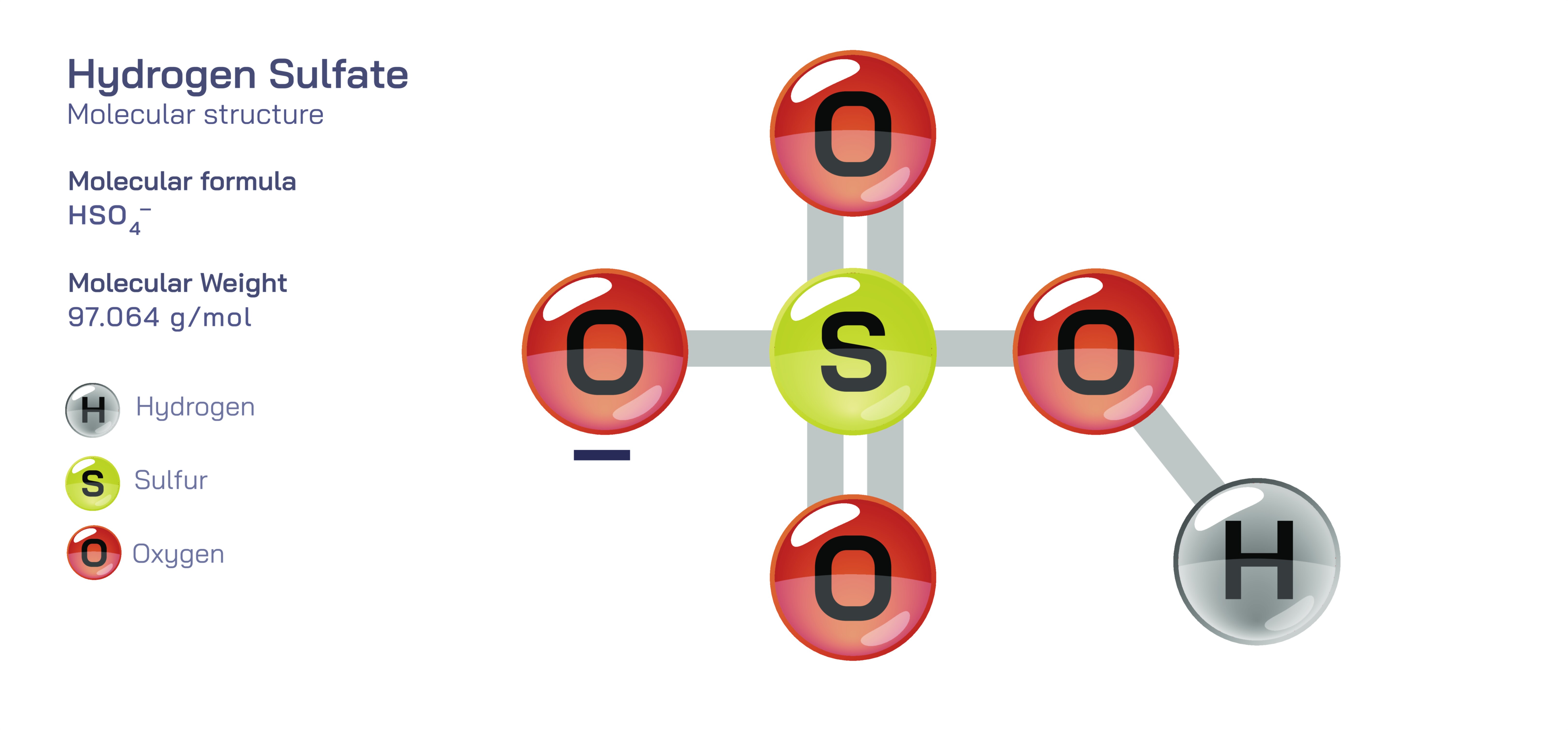
Hydrogen Sulfate Ion Structure — HSO₄⁻ Chemical Diagram and Molecular Behavior Explained
The hydrogen sulfate ion, written chemically as HSO₄⁻, appears at first glance to be nothing more than sulfuric acid missing one proton, yet its structural identity is far more complex than that simple description suggests. It is not merely a residual fragment of an acid; it is a stable, independently functioning polyatomic ion shaped by a precise arrangement of atoms and charge distribution that governs how it reacts in biological systems, environmental chemistry, and industrial processes. Understanding the hydrogen sulfate ion requires looking beneath its formula to the three-dimensional structure of its atoms, the bonds that connect them, and the ways in which the negative charge is held and shared across the molecule. The entire chemical personality of HSO₄⁻ — its ability to donate protons, accept electrons, form salts, participate in buffer systems, and take part in atmospheric and aqueous reactions — emerges from the way sulfur, oxygen, and hydrogen interact within this ion.
Structurally, the hydrogen sulfate ion consists of a sulfur atom centrally bonded to four oxygen atoms arranged in a tetrahedral geometry. One of these oxygen atoms is connected to a hydrogen atom, while the other three remain unprotonated. The ion carries a single negative charge, yet that charge is not rigidly localized on one oxygen atom. Instead, it is delocalized across the molecule through resonance, meaning that the charge is spread over the oxygen atoms through overlapping orbital structures rather than sitting in a single fixed position. This delocalization helps stabilize the ion significantly, allowing hydrogen sulfate to exist comfortably in aqueous environments and enabling it to serve as a conjugate base of sulfuric acid. Although sulfuric acid itself is a strong acid that dissociates almost completely, hydrogen sulfate still retains the ability to donate a proton and convert into sulfate, SO₄²⁻, which is one of the most stable oxyanions found in nature. This ability to both carry and release a proton — the defining hallmark of hydrogen sulfate — positions it midway between reactivity and stability, not as an unstable fragment but as a controlled intermediate between an acid and its fully deprotonated conjugate base.
The structural arrangement of the oxygen atoms around the sulfur atom determines not only the shape but also the chemistry of the hydrogen sulfate ion. The S–O bonds in the ion are not equivalent in the classical sense because one oxygen bears a hydrogen while the others do not, yet the molecular charge is distributed in such a way that the entire structure remains symmetrical in its electronic character. This electronic symmetry explains why hydrogen sulfate behaves so predictably in aqueous systems: when dissolved in water, it can release a proton to become sulfate, or it can act as a weak base by accepting a proton to reform sulfuric acid. In real-world solutions, the ion does not float in isolation but constantly participates in an equilibrium defined by pH, concentration, and surrounding ions. When the environment is highly acidic, hydrogen sulfate holds onto its hydrogen and behaves more like a neutral acid derivative. As pH begins to rise, the ion donates its hydrogen to water, shifting toward sulfate as the stable form. These transformations make hydrogen sulfate an important buffering species in natural waters, industrial chemical systems, and biological fluids where acid-base balance must remain controlled rather than swinging wildly between extremes.
Hydrogen sulfate becomes especially important when considering soluble salts. When paired with positively charged ions such as sodium, potassium, ammonium, calcium, or magnesium, hydrogen sulfate forms compounds known as bisulfates. These salts are widely used in laboratories, water treatment systems, and industrial processes because of their unique acidity and solubility properties. Unlike sulfate salts, which are nearly pH-neutral, bisulfates retain the ability to release protons, allowing them to adjust acidity without the volatility or corrosive fumes associated with sulfuric acid. Household compounds such as sodium bisulfate are used in swimming pool pH control and cleaning products because the hydrogen sulfate ion provides acidity in a solid, easily handled form. This example shows that the ion is not simply a microscopic theoretical structure but a practical and versatile chemical tool.
Beyond industrial relevance, hydrogen sulfate plays a significant role in natural ecosystems, particularly in atmospheric and geochemical cycles. When sulfur-containing compounds rise into the atmosphere from natural or human activities and interact with moisture, oxidation processes convert them into sulfuric acid, which then dissociates into hydrogen sulfate and sulfate ions in water droplets. These ions contribute to acidity in clouds and rainfall, which in turn affects soil chemistry, aquatic habitats, and mineral weathering. The persistence and mobility of hydrogen sulfate in water are key factors in how ecosystems adapt to chemical changes. In natural waters, hydrogen sulfate and sulfate act as part of a biochemical network that supports the sulfur cycle — the global transformation of sulfur between rocks, soil, oceans, organisms, and the atmosphere. Although hydrogen sulfate itself does not form biological macromolecules, its presence influences microbial metabolism and biochemical pathways, particularly in environments where acidity regulates metabolic activity.
From a molecular composition perspective, hydrogen sulfate represents a balance between strong chemical bonding and reactive potential. Its sulfur atom, surrounded by four electronegative oxygens, sits in a high-oxidation state and creates a highly polarized ion. This electronegativity distribution gives hydrogen sulfate the tendency to participate in reactions that involve proton transfer, electron flow, and ionic bonding. However, unlike sulfuric acid, hydrogen sulfate does not release unstable vapors and is significantly safer to handle, which explains its widespread use in regulated settings. The ion is stable enough to store and transport but still reactive enough to influence pH and participate in synthesis.
The hydrogen sulfate ion therefore stands as a perfect example of how structural chemistry governs function. Its tetrahedral geometry, delocalized charge, capacity to donate or retain hydrogen, and compatibility with a vast range of cations define its behavior across laboratory science, environmental systems, biological chemistry, and industrial processes. The molecule is not simply a remnant of sulfuric acid; it is a bridge between acidity and neutrality, a buffering agent, a reactive intermediate, and a structural unit in countless compounds. Every atom and bond in the HSO₄⁻ ion supports the balance between stability and reactivity that makes it both functional and ubiquitous. Understanding this ion means appreciating that even the smallest structural variations — a hydrogen attached to one oxygen rather than none — are enough to transform the chemical identity of a system, shaping how matter behaves throughout both technology and nature.