Homogenisation — Process, Principles, and Applications in Science and Industry Explained
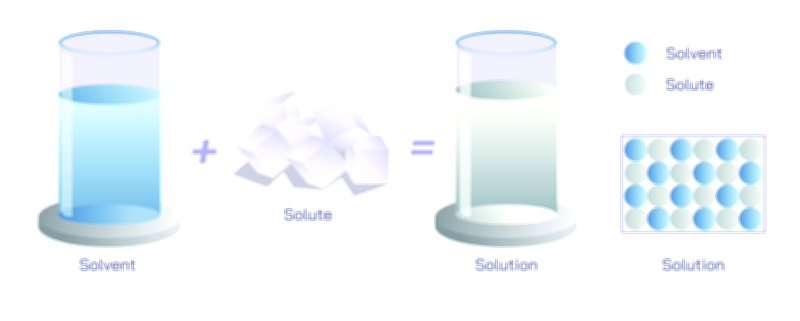
Homogenisation is a process designed to transform a mixture of two or more substances into a uniform and stable system in which the components are distributed evenly throughout the medium without separating over time. Although the term often appears in the context of food processing, particularly in the production of milk, homogenisation is a universal scientific and industrial technique fundamental to chemistry, biology, material science, pharmaceuticals, cosmetics, petrochemicals, biotechnology, and multiple branches of manufacturing. What defines homogenisation is not just mixing but achieving a state in which the size of dispersed particles or droplets becomes so small and so evenly distributed that they can no longer separate under normal environmental conditions. To understand this process deeply, one must explore the scientific principles of particle reduction, dispersion, energy input, intermolecular interaction, and stability that make homogenisation possible and the wide-ranging applications that emerge from these principles.
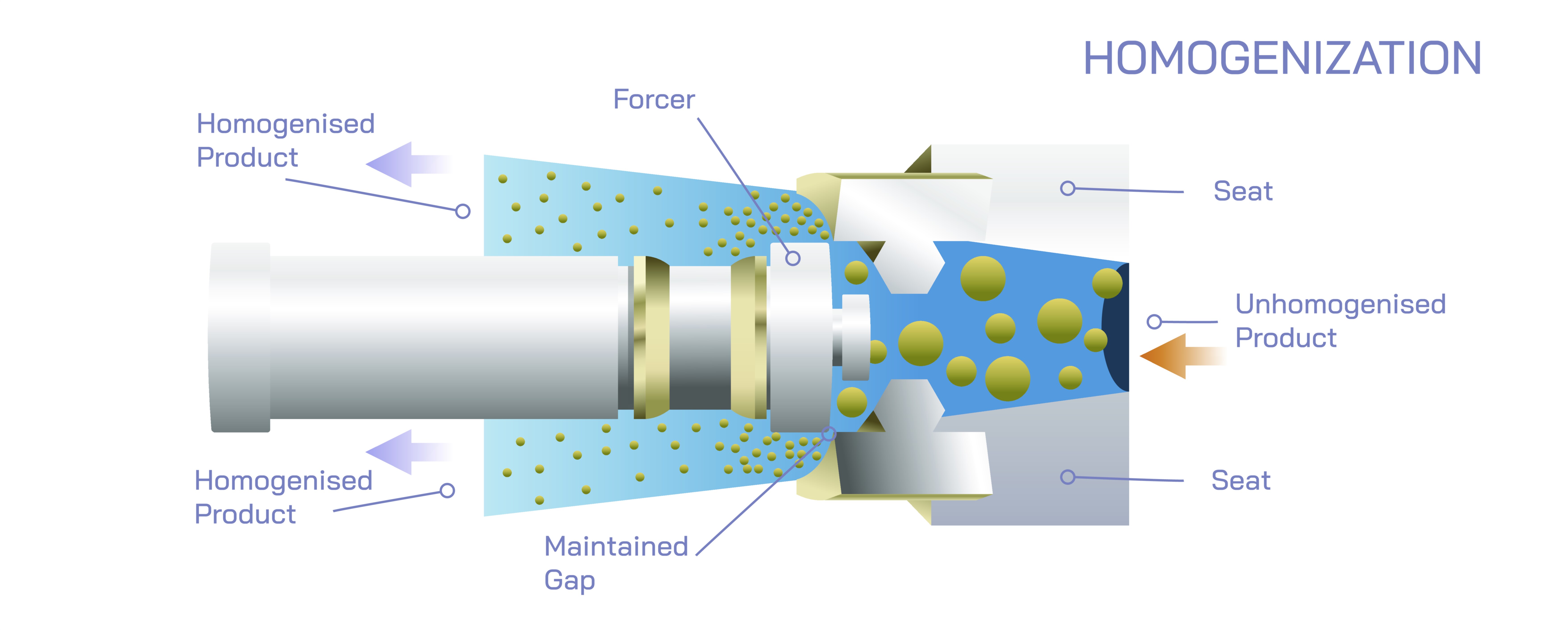
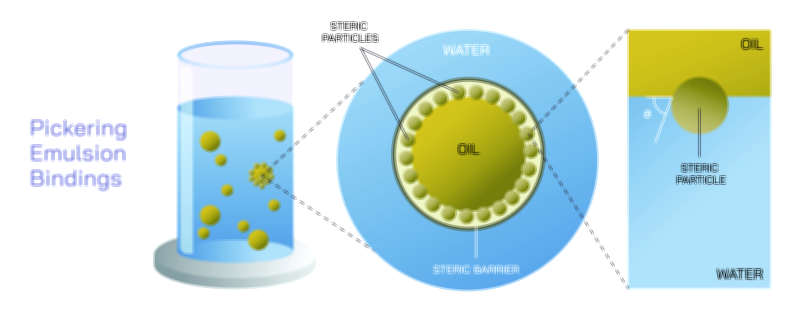
At the core of homogenisation lies the principle of mechanical disruption, in which large particles or droplets are broken down into extremely small ones by applying controlled energy. When two phases, such as oil and water, exist together, surface tension causes them to resist mixing because molecules within each phase tend to remain with their own kind. When homogenisation is applied, intense mechanical forces such as pressure, turbulence, shear, and cavitation break large droplets or particles into micro-scale or even nano-scale sizes. The dramatic increase in surface area following this size reduction allows stabilizing interactions to occur, whether through natural molecular affinity or added emulsifiers and surfactants. The mixture then becomes uniform not because the components naturally blend, but because particle size becomes too small to recombine and because interparticle forces or thermodynamic stability prevent reaggregation. This concept makes homogenisation far more than simple stirring; it is engineering at the microscopic level to control matter in a stable dispersed form.
The process of homogenisation can be carried out using many different devices, each harnessing energy in a unique way to disrupt structures. High-pressure homogenisers force a mixture through a narrow valve at extremely high velocity, causing sudden pressure drops, impact forces, and turbulence that tear droplets apart. Ultrasonic homogenisers use high-frequency sound waves to generate cavitation bubbles that collapse violently and create shear forces capable of breaking apart particles. Rotor–stator homogenisers use rapidly spinning impellers to generate zones of high shear where particles collide and disperse. The unifying element across all these systems is the conversion of mechanical energy into microscopic disruptive events that reduce particle size and drive the components toward a uniform state. The efficiency of the process depends on the intensity of applied energy, the properties of the materials, and the degree of stability required for the final product.
One of the most widely recognized applications of homogenisation occurs in the dairy industry, where milk is processed to prevent the cream layer from separating. In raw milk, fat globules are large and buoyant, so they gradually rise to the surface during storage. When milk undergoes high-pressure homogenisation, the fat droplets are broken down into tiny globules that remain suspended throughout the liquid, eliminating visible separation. What makes this process function so effectively is that the reduced droplet size changes the physical behaviour of fat in the aqueous phase, while proteins in the milk act as natural stabilisers by coating the fat droplets and preventing them from clumping together. Through this microscopic transformation, a once unstable mixture becomes uniformly smooth, stable, and more visually appealing while also improving digestibility and sensory texture.
Beyond milk, the food industry relies extensively on homogenisation to improve texture, stability, and mouthfeel in products such as sauces, salad dressings, nut milks, ice creams, beverages, spreads, creams, and confectionary fillings. In ice cream, homogenisation breaks and redistributes fat globules so they interact with air bubbles and ice crystals in ways that give the final product its creamy structure rather than a coarse, icy texture. In sauces and dressings, homogenisation ensures that oil droplets remain suspended without separating, even after long storage periods. In plant-based milk alternatives, homogenisation disrupts cellulose fibres and oil droplets to create smooth consistency and uniform visual appearance. These transformations illustrate that homogenisation is as much about sensory experience and consumer product quality as it is about chemical and physical stability.
In the pharmaceutical field, homogenisation plays a critical role in drug delivery and formulation. Many active pharmaceutical ingredients are poorly soluble in water and must be delivered in suspension or emulsion form. By reducing particle size to the micro or nano scale, homogenisation increases surface area and enhances dissolution rates, allowing drugs to be absorbed more efficiently into the body. Homogenised nanosuspensions and nanoemulsions are used to improve the bioavailability of medications that would otherwise be ineffective in the digestive system. In topical creams and ointments, homogenisation ensures that both the active and inactive components remain evenly distributed, providing consistent dosing and smooth application. In injectable formulations, homogenisation produces fine dispersions that are safe for intravenous administration and do not create blockages or uneven deposition within tissues. The process therefore connects physical particle manipulation to direct therapeutic impact.
In cosmetics and personal care, homogenisation gives lotions, shampoos, conditioners, serums, and makeup their characteristic textures and stability. Creams must maintain a uniform consistency without oil separation, and emulsions must remain smooth over long storage periods despite temperature fluctuations and repeated consumer handling. Homogenisation not only creates uniform droplet distribution but also influences how products feel on the skin, determining whether a lotion spreads easily, absorbs quickly, or creates a protective film. In perfume and fragrance formulations, homogenisation ensures the even dispersion of aromatic oils in alcohol or other carrier systems, providing consistent scent performance from the first spray to the last.
Homogenisation is also critical in biotechnology and microbiology for cell disruption and extraction of intracellular material. When used intentionally to break biological cells, homogenisation releases proteins, DNA, enzymes, organelles, metabolites, and other cellular contents for research or industrial processing. In this case, the goal is not stability of a dispersion but controlled rupture of biological membranes. The same principle applies in biofuel production, where algae or biomass must be disrupted to release lipids for conversion into fuel. Here, the mechanical forces of homogenisation become tools to access biological resources locked inside cell structures.
In chemical and petrochemical industries, homogenisation ensures uniformity in complex mixtures such as lubricants, cutting fluids, polymer dispersions, fuel additives, bitumen emulsions, and paints. Many of these products require tightly controlled particle sizes to prevent sedimentation, clogging, or changes in performance over time. A lubricant with poorly dispersed additives can lead to engine wear; an ink with unstable pigment distribution can cause streaking; and a coating with uneven dispersion can produce weak or uneven protective films. Homogenisation prevents these failures by generating dispersions that remain stable for long periods under mechanical stress, temperature variation, and storage conditions.
Environmental science and wastewater treatment also utilise homogenisation when samples must be broken down into uniform consistency for accurate testing or when sludge mixtures must be conditioned for optimal digestion and processing. Stable dispersions allow chemical, biological, and physical treatment techniques to work more efficiently and predictably. In research laboratories, homogenisation is often the first step of sample preparation, ensuring that subsequent analysis reflects the complete sample rather than unevenly distributed components.
The principle underlying all these diverse applications is constant: homogenisation controls matter at the microscopic scale to achieve uniformity, stability, or controlled disruption depending on the purpose. What distinguishes homogenisation from simple mixing is the energy required to overcome natural separation forces, whether they arise from density differences, surface tension, hydrophobicity, cellular structure, or intermolecular bonding. Homogenisation transforms materials physically so that their behaviour in the macroscopic world — whether as food, medicine, lubricant, fuel, cosmetic, or biological extract — becomes predictable and functional.
Ultimately, homogenisation represents one of the clearest examples of how manipulating particle size and distribution can change the identity, performance, and usefulness of a material without altering its basic chemical composition. By applying mechanical forces at the microscopic level, industries shape large-scale attributes such as stability, texture, absorption, flow, combustion, interaction, and shelf life. Homogenisation shows that the physical organisation of matter is as important as chemical structure when determining how substances behave in real-world systems. Through this perspective, homogenisation becomes not only a process but a principle that bridges science and industry by demonstrating how control at the smallest scale creates value at the largest.