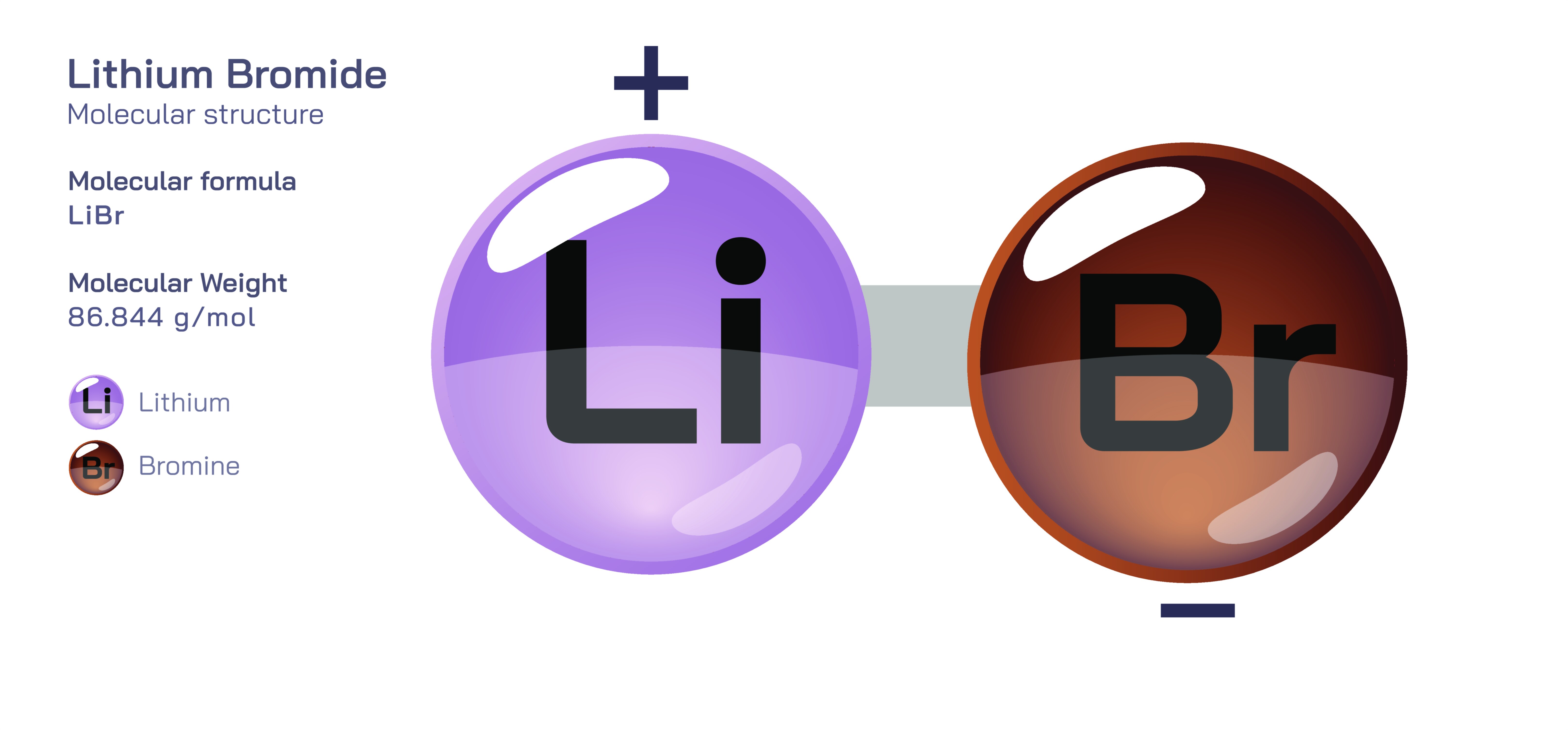
Lithium Bromide — Chemical Structure, Formula, Properties and Applications in Absorption Refrigeration Systems
Lithium bromide is an important inorganic compound widely used in industrial thermal systems, particularly in absorption refrigeration and dehumidification applications, where its unique chemical properties make it exceptionally suited to driving temperature and humidity control without relying on traditional mechanical compression. Chemically represented by the formula LiBr, lithium bromide consists of a lithium cation (Li⁺) and a bromide anion (Br⁻), forming an ionic compound that dissolves readily in water, producing highly concentrated solutions. This simple composition belies a remarkable range of behaviours. The small ionic radius of lithium produces a strong electrostatic attraction to the bromide ion, creating a highly stable salt that is extremely hygroscopic, meaning it has a strong tendency to absorb moisture from the air. Lithium bromide’s affinity for water, high solubility, and strong ability to lower vapour pressure allow it to act as a powerful absorbent in thermal processes, enabling refrigeration and climate-control systems powered not by electricity-intensive compressors but by heat—an energy source that can come from gas burners, waste heat, or solar collectors. Because of this, lithium bromide has become a central component in energy-efficient industrial cooling technologies while exemplifying how chemical principles can be integrated with mechanical engineering to transform thermal management.
The structure of lithium bromide in its solid form reflects typical ionic bonding, where lithium ions are surrounded by bromide ions in a repeating lattice arrangement driven by electrostatic forces. Unlike covalent compounds that show directional bonding, ionic salts like LiBr depend on charge attraction to create extended crystalline structures. The lithium ion, being very small, creates a compact lattice with bromide ions arranged to maximize electrostatic stability. Such ionic packing gives solid LiBr high melting and boiling points, making it resilient under high-temperature industrial applications. When dissolved, however, the crystal lattice separates completely, and lithium and bromide ions become individually solvated by water molecules. This solubility is critical for absorption refrigeration because the performance of the system depends on the ability of the LiBr solution to strongly bind water and remove vapour from low-pressure environments.
The defining property that makes lithium bromide so valuable in absorption systems is its extraordinary hygroscopicity. A highly concentrated LiBr solution exerts a very low vapour pressure, meaning that when exposed to moist air or water vapour, it rapidly absorbs water to equalize pressure. This absorption process physically draws water molecules out of the surrounding atmosphere or evaporated refrigerant stream. In refrigeration systems, it allows the refrigerant—usually water—to evaporate at a very low temperature, absorbing heat from the environment and producing a cooling effect. Whereas conventional vapour-compression air conditioners require electrically driven compressors to force refrigerant evaporation and condensation, lithium bromide absorption systems rely instead on differences in solution concentration driven by heat. When a low-concentration LiBr solution absorbs water vapour, it becomes diluted. Heat is then applied in a generator or desorber to expel the absorbed water, reconcentrating the LiBr solution. The expelled refrigerant vapour is condensed and recirculated, while the reconcentrated solution returns to the absorber to repeat the cycle. This closed-loop process allows cooling to occur while using heat as the input energy rather than mechanical work.
This ability to enable refrigeration using heat rather than electricity has profound implications for energy efficiency and environmental sustainability. Large absorption chillers using lithium bromide are common in commercial buildings, district cooling systems, manufacturing plants, and industrial complexes that produce waste heat. By using excess thermal energy from boilers, combined heat-and-power installations, diesel generators, and even solar thermal collectors, these systems reduce reliance on grid electricity and lower carbon emissions. Lithium bromide systems have no ozone-depleting or greenhouse-gas refrigerants circulating under high pressure, instead relying on water as the working refrigerant, making them attractive alternatives in sustainable engineering and climate control. They operate quietly, have fewer moving parts than mechanical chillers, and often provide longer operational life due to reduced mechanical wear. Air-conditioning systems in hospitals, data centres, office buildings, chemical plants, and large hotels frequently use LiBr technology, especially when thermal energy savings offset the initial cost of installation.
Lithium bromide’s ability to absorb water also underpins its role as a desiccant in industrial dehumidification. Concentrated LiBr solutions or impregnated solid materials are used to remove moisture from air streams in drying applications, humidity-controlled manufacturing environments, pharmaceutical production rooms, textile factories, and storage facilities for moisture-sensitive goods. In HVAC systems, LiBr dehumidification improves indoor-air quality while reducing the cooling load on mechanical refrigeration units. Some modern energy-recovery ventilation systems incorporate lithium bromide in liquid desiccant modules to regulate humidity while exchanging heat between incoming and outgoing air. These applications rely on LiBr’s strongly exothermic water-absorption properties—when water vapour binds to the solution, heat is released, and the system must be engineered to dissipate this heat while maintaining stability of the salt solution.
Although its major industrial use is in absorption refrigeration, lithium bromide also serves multiple roles across chemistry and materials science. It is used as a catalyst and reagent in organic synthesis, particularly in reactions that require strong ionic environments or coordination with polar solvents. Because of the small size and high charge density of Li⁺, lithium bromide is employed in some pharmaceutical manufacturing processes to influence molecular rearrangement or promote dehydration reactions. In metallurgical processes, LiBr can appear in flux mixtures to improve molten-metal fluidity and reduce oxide formation. In the field of electronics and battery development, lithium bromide has been investigated as an electrolyte component due to the reactivity of lithium ions and the stability of bromide salts in certain solvent systems, although safety considerations have limited some applications. LiBr is also used in specialized photographic chemical solutions and in heat-storage salt mixtures for industrial thermal management.
The handling of lithium bromide requires careful safety management due to its corrosive nature when dissolved in water and its potential to cause irritation or chemical burns through prolonged skin or eye contact. It is not considered highly toxic compared with many industrial chemicals, but concentrated solutions can damage metals and cause equipment corrosion without proper material selection. Absorption chiller systems therefore use corrosion inhibitors combined with LiBr to protect internal steel components. Proper storage, ventilation, and closed-system circulation prevent LiBr from absorbing atmospheric moisture uncontrollably, which could result in clogging, crystallization or pressure imbalances. Modern industrial guidelines treat lithium bromide as a controlled industrial material that is safe when handled within engineered systems but unsuitable for uncontrolled release or direct domestic exposure.
What ultimately makes lithium bromide scientifically and technologically significant is the combination of molecular simplicity and industrial versatility. At the atomic level, it exists only as lithium and bromide ions; at the chemical level, it behaves as a powerful water-binding agent; and at the engineering level, it becomes the driving force behind some of the most energy-efficient cooling systems humans have developed. Its structure and ionic behaviour explain why it dissolves easily, absorbs water aggressively, and modifies vapour pressure so dramatically. Its hygroscopic nature explains why it underpins dehumidification and climate-control systems. Its chemical stability explains why it can operate at high temperatures in cyclic thermal absorption processes. All these characteristics together transform lithium bromide from a simple inorganic salt into a cornerstone of sustainable refrigeration and industrial humidity regulation.
In essence, lithium bromide demonstrates how a fundamental chemical property—the attraction between ions and water molecules—can be magnified into a large-scale engineering advantage. By enabling cooling and dehumidification through the absorption of water rather than the compression of refrigerant gases, lithium bromide helps replace high-power mechanical systems with quiet, efficient thermochemical alternatives. Through its structure, formula and unique absorption behaviour, LiBr stands at the intersection of chemistry, thermodynamics, and sustainable industrial technology, illustrating how molecular interaction can be scaled to reshape energy use in buildings, factories and climate-control networks around the world.