Raoult’s Law: Relationship Between Vapor Pressure and Solute Concentration in Solutions Explained
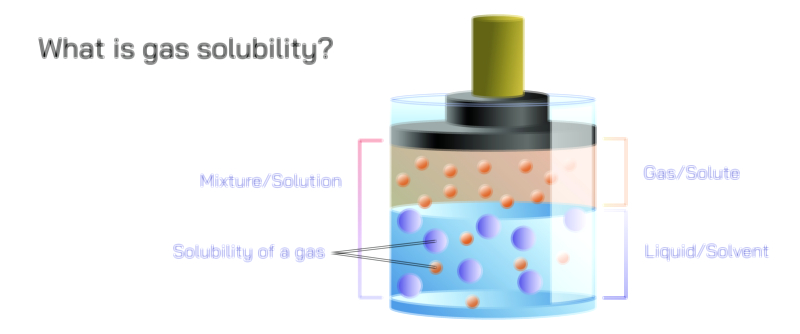
Raoult’s Law describes one of the most fundamental relationships in physical chemistry: how the presence of a solute influences the vapor pressure of a solvent in a solution. Although the concept often appears in textbooks alongside graphs and formulas, its true meaning lies in the molecular behavior of liquids. Vapor pressure is the pressure exerted by solvent molecules that escape from the liquid phase into the gas phase. In a pure solvent, countless molecules near the surface possess enough energy to evaporate, creating a characteristic vapor pressure that reflects the balance between molecules leaving and re-entering the liquid. When a solute is introduced into the solvent—whether salt in water, sugar in ethanol, or any non-volatile substance dissolved in a liquid—the escaping tendency of solvent molecules changes. Raoult’s Law explains how and why vapor pressure decreases as solute concentration increases, linking macroscopic observations to the underlying motion and interactions of molecules.
To understand this relationship, it is important to begin with the concept of a volatile vs. non-volatile solute. A non-volatile solute does not vaporize, meaning its molecules remain in the liquid phase rather than entering the gas phase. When such a solute dissolves in a solvent, it reduces the number of solvent molecules near the surface, simply because some of the surface spots that would normally be occupied by solvent molecules are now taken by solute particles. Fewer solvent molecules at the surface means fewer opportunities for evaporation to occur. Even without calculating anything, this simple physical idea shows that adding solute lowers vapor pressure. The more concentrated the solute becomes, the lower the vapor pressure of the solution becomes. Raoult’s Law captures this reduction in vapor pressure as a predictable, proportional effect that depends on how much of the solution is made up of solvent compared to solute.
A molecular perspective makes the law even clearer. In a pure solvent, each molecule is surrounded by identical neighbors and the only barrier to vaporization is the attraction between like molecules, such as hydrogen bonding in water or dipole interaction in acetone. When a solute dissolves, the solution contains both solute and solvent particles. The introduction of solute disrupts the pure-solvent molecular environment and changes the probability that solvent molecules at the surface can move freely into the vapor phase. The fraction of the surface occupied by solvent corresponds directly to the mole fraction of the solvent in the bulk solution, which becomes the key variable that determines vapor pressure behavior. Raoult’s Law essentially says that the vapor pressure of the solution is proportional to the mole fraction of the solvent. As the mole fraction decreases—because more solute is added—the vapor pressure decreases proportionally. This makes vapor pressure reduction a colligative property, meaning it depends on the number of dissolved particles rather than their chemical identity. A variety of solutes behave similarly: salt, sugar, organic compounds, and polymers all reduce vapor pressure when dissolved in a liquid.
Raoult’s Law is most accurate for ideal solutions, which are mixtures where solute–solvent interactions are similar in energy to the interactions found between pure solvent molecules. In an ideal solution, mixing does not significantly increase or decrease molecular attractions, so the vapor pressure changes smoothly and predictably with composition. Real solutions vary in how closely they follow this ideal pattern. For example, highly polar solutes in non-polar solvents or substances that strongly hydrogen-bond with the solvent can create deviations. When solute-solvent attractions are stronger than solvent-solvent attractions, the vapor pressure falls even more than predicted because solvent molecules are held more tightly in the liquid phase. Conversely, when solute-solvent interactions are weaker, vapor pressure can rise above the predicted value because molecules escape more easily. These deviations do not contradict Raoult’s Law; rather, they reveal the subtleties of intermolecular forces, illustrating how microscopic interactions influence measurable physical properties.
The vapor-pressure lowering effect leads directly to several well-known phenomena in chemistry and everyday life. Because vapor pressure is linked to boiling, the addition of solute raises the boiling point of a liquid. This explains why adding salt to water increases the temperature required for boiling and why antifreeze prevents radiator coolant from boiling over. Likewise, lowering vapor pressure reduces freezing point because solid formation requires solvent molecules to gather in a pure, ordered arrangement that becomes increasingly difficult when solute molecules are mixed in. This explains why salt melts ice on roads and why ocean water freezes at temperatures lower than fresh water. In biological systems, vapor pressure lowering influences osmotic pressure and water movement across cell membranes. In industries ranging from food processing to pharmaceuticals, controlling vapor pressure allows for fine regulation of concentration, storage stability, and evaporation rates. All of these effects trace back to the initial molecular principle described by Raoult’s Law.
Perhaps the most striking aspect of Raoult’s Law is how it reveals that solutions behave in systematic and predictable ways even when composed of different substances. The identity of the solute matters far less than the number of solute particles present because vapor pressure lowering depends on the statistical distribution of molecules at the liquid surface. In this sense, the law serves as a bridge between thermodynamics and molecular theory, demonstrating how microscopic motion determines macroscopic behavior. When chemists measure vapor pressure to determine molar masses, when environmental scientists model evaporation in lakes, or when engineers design industrial distillation processes, they rely on the principle that adding solute reduces the escaping tendency of solvent. Raoult’s Law may appear simple at first glance, but it encapsulates deep insights into the structure of matter and the mathematical order underlying chemical mixtures.
Ultimately, Raoult’s Law captures a universal pattern in solution chemistry: dissolving solute reduces the vapor pressure of the solvent because it reduces the availability of solvent molecules to escape into the gas phase. From that simple physical truth emerges a chain of consequences — boiling point elevation, freezing point depression, osmotic pressure, and countless applications in science and technology. Although the law is built on a microscopic event that cannot be seen with the naked eye, it shapes phenomena that extend from everyday household solutions to advanced industrial chemistry. In describing the relationship between vapor pressure and solute concentration, Raoult’s Law reveals how profoundly the behavior of molecules governs the world around us.