Standard Hydrogen Electrode (SHE: Structure and Working Principle Explained)
The Standard Hydrogen Electrode, commonly abbreviated as SHE, occupies a central role in electrochemistry as the universal reference against which all other electrode potentials are measured. Its purpose is not rooted in ordinary industrial use but in establishing a reproducible and stable zero point on the electrochemical scale, enabling scientists to compare the oxidizing and reducing tendencies of all known substances. When electrochemists describe a half-cell as possessing a certain reduction potential — whether copper, zinc, chlorine, or any other redox pair — that value is determined relative to the Standard Hydrogen Electrode. SHE therefore forms the foundation of the electrochemical series, allowing insight into spontaneous reaction likelihood, energy output from batteries, corrosion behavior, redox equilibria in biochemical systems, and industrial electrolysis. Understanding SHE requires examining its physical structure, the chemical reactions that occur at its surface, and the controlled environmental conditions that give it unmatched reproducibility.
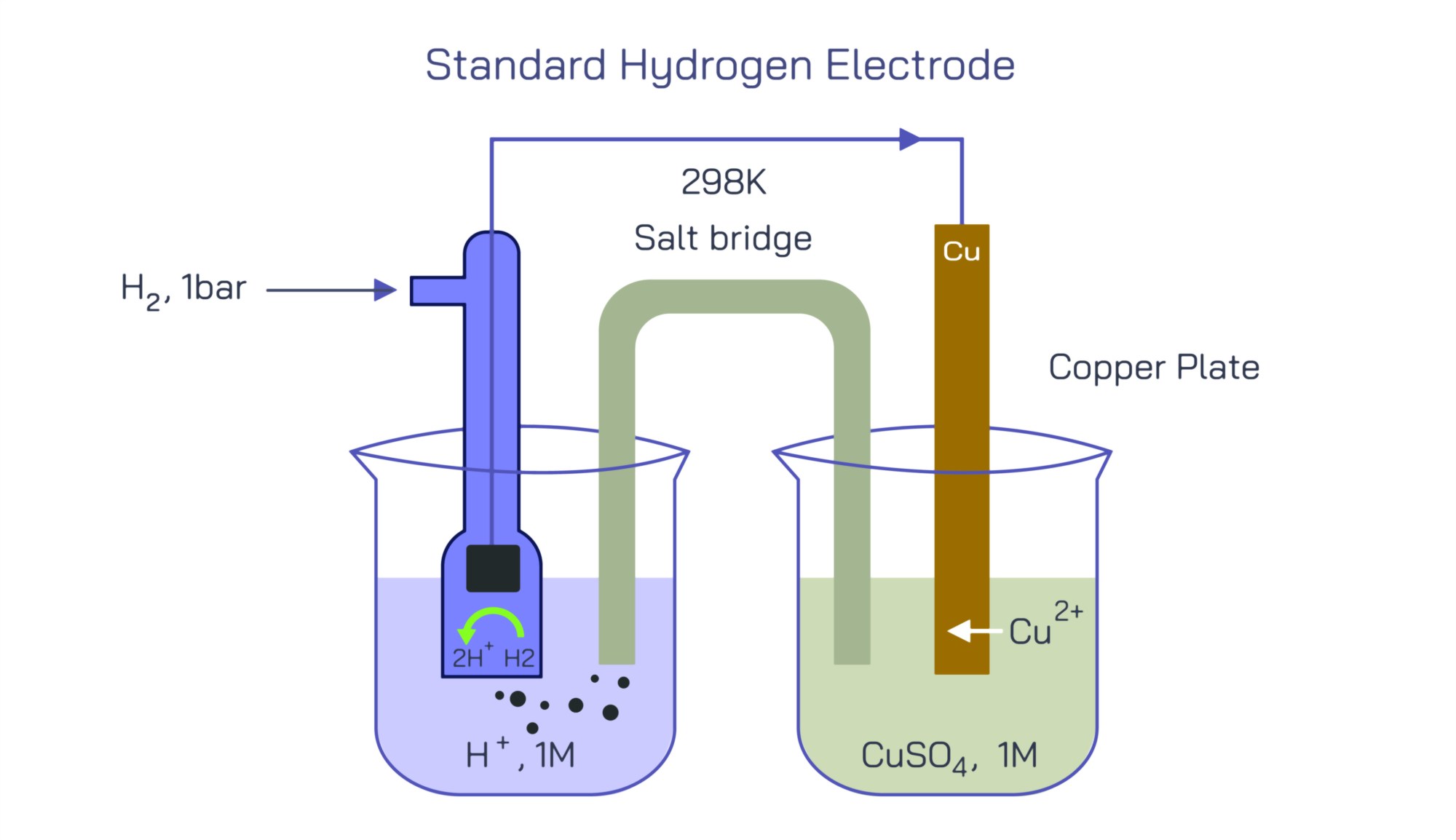
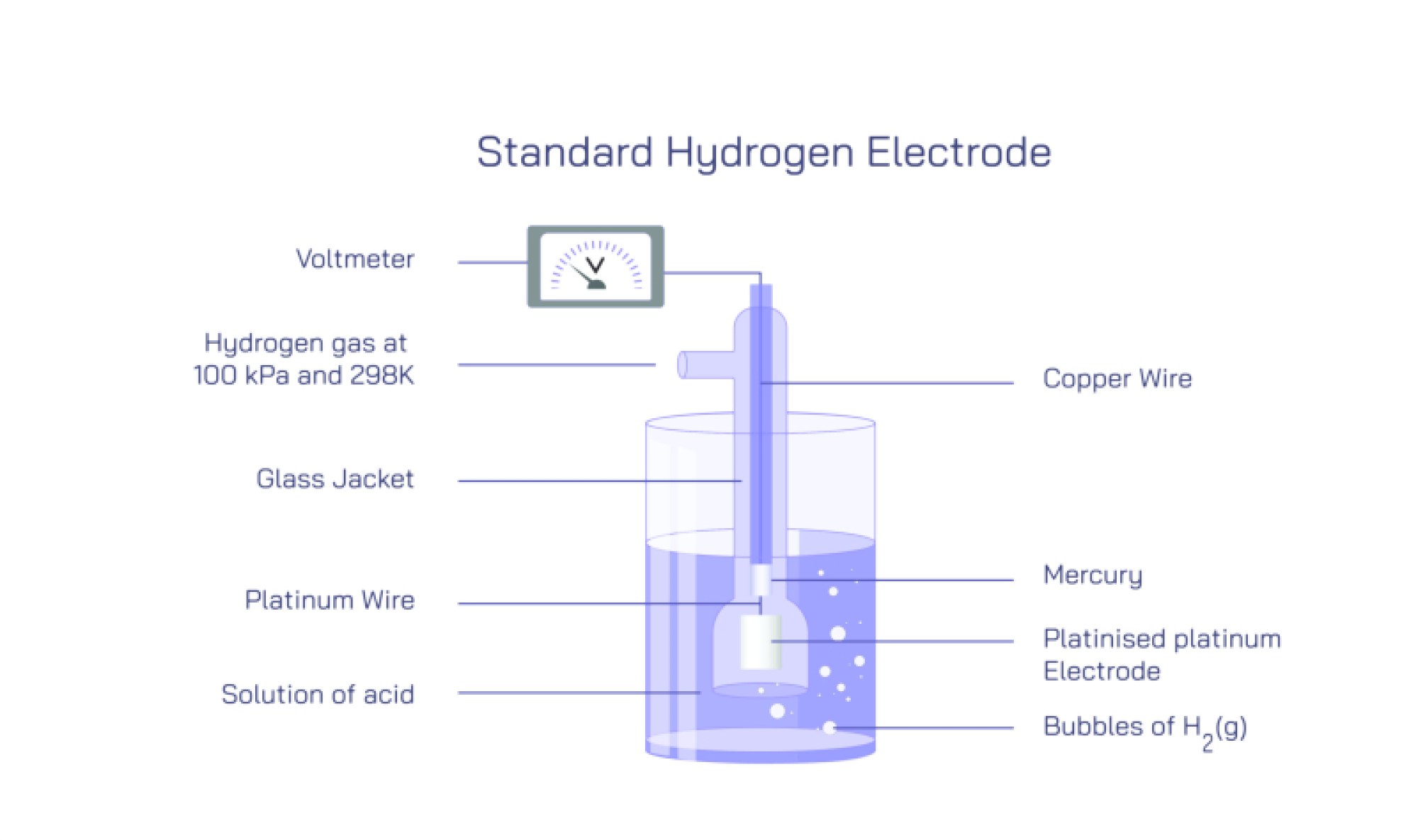
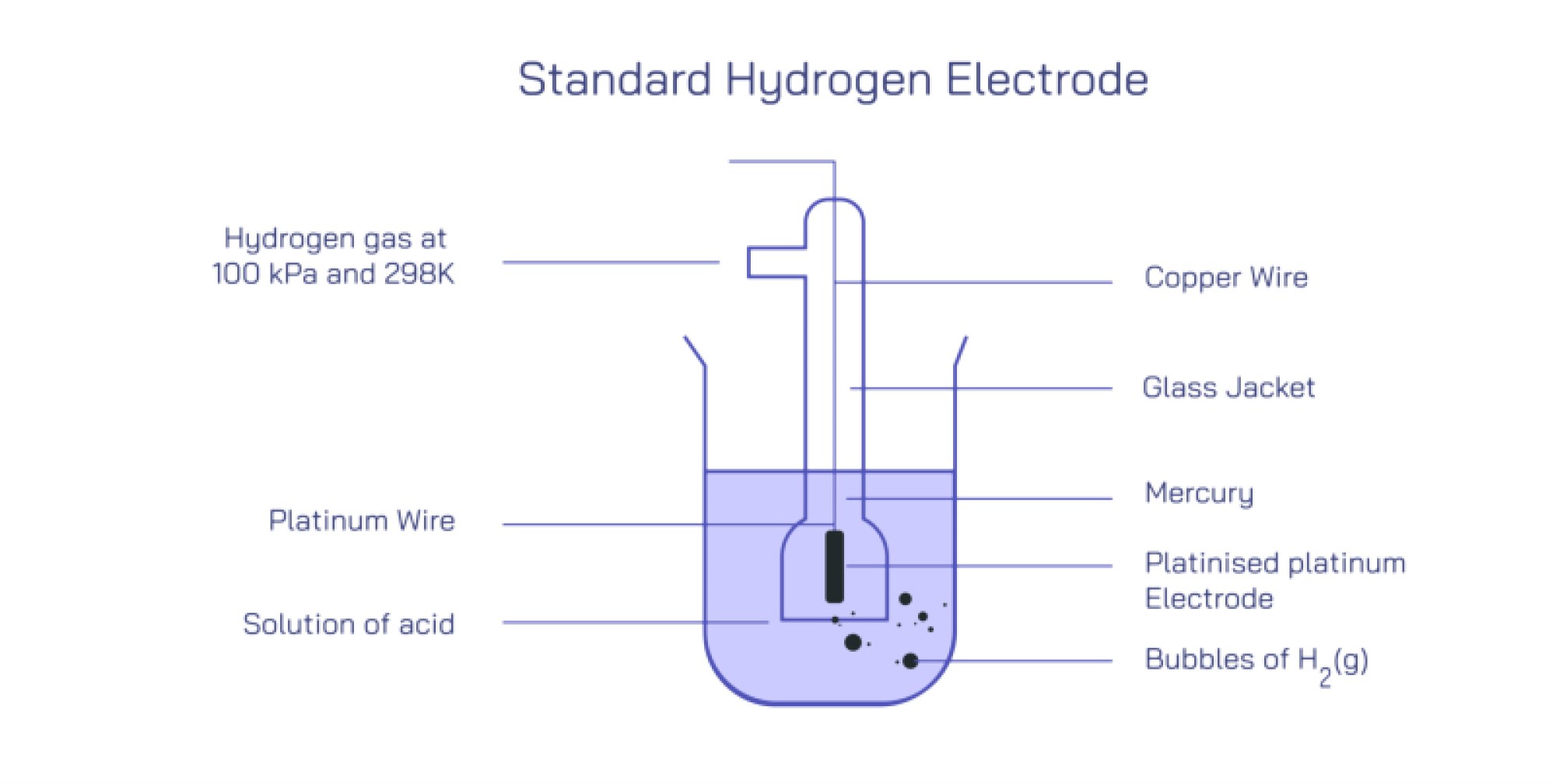
Structurally, the Standard Hydrogen Electrode is built with precision to ensure full consistency in every laboratory and measurement setup. At its core is a platinum electrode, typically a thin sheet, foil, or platinized platinum black surface, partially immersed in a solution containing hydrogen ions at a standard concentration of 1 mol per liter (1 M H⁺). The platinum surface is roughened with platinum black to drastically increase its surface area, which enhances the adsorption of hydrogen molecules and ions and ensures rapid attainment of equilibrium. The electrode does not participate chemically in the reaction; it serves as an inert conductor that facilitates electron exchange between the external circuit and hydrogen species in the solution. Surrounding the platinum electrode, pure hydrogen gas at exactly 1 atmosphere pressure is continuously bubbled over the surface. The bubbling maintains direct contact between dissolved hydrogen and the electrode and prevents buildup of concentration gradients. The entire system operates at standard temperature, conventionally 25 °C (298 K). These tightly controlled parameters together establish the "standard state" for measurement. When SHE is connected to any other half-cell in an electrochemical circuit, the two form a complete galvanic cell, and the resulting voltage difference reveals the other electrode’s potential relative to SHE.
At the platinum surface, hydrogen undergoes a reversible redox reaction, meaning the electrode can switch freely between oxidation and reduction depending on which direction electrons flow. The reaction that defines SHE is
H₂(g) ⇌ 2H⁺(aq) + 2e⁻
If electrons are supplied to the electrode from an external circuit, hydrogen ions gain electrons, producing hydrogen gas — a reduction process. If electrons leave the electrode toward the external circuit, hydrogen molecules dissolve to form hydrogen ions — an oxidation process. What makes SHE uniquely powerful is that the exact moment of equilibrium, when oxidation and reduction tendencies are perfectly balanced, is reproducible under standard conditions and possesses a potential defined as 0.00 volts by international agreement. This does not mean that SHE lacks reactivity or exerts no driving force; rather, its value provides the zero reference against which other electrode potentials are interpreted. A half-cell with a positive potential relative to SHE has a greater tendency to gain electrons (undergo reduction), whereas one with a negative potential has a greater tendency to lose electrons (undergo oxidation). Thus, the SHE acts as the anchor point for the entire redox landscape of chemistry.
The operation of SHE depends on the subtle interplay between hydrogen adsorption on platinum, proton concentration in solution, gas pressure, and electron transfer rates. The platinum’s role is essential not only because it is chemically inert but also because it can bind hydrogen atoms temporarily in a way that promotes rapid exchange between molecular hydrogen, dissolved protons, and electrons. At the microscopic level, hydrogen molecules dissociate on the platinum surface into adsorbed atoms, allowing electron exchange before recombining and desorbing into solution or gas phase. These reversible micro-transformations ensure that the electrode reaches a stable equilibrium potential very quickly. Without the platinum catalyst, hydrogen exchange would proceed too slowly for SHE to function reliably. Likewise, the bubbling of hydrogen gas keeps the concentration of dissolved hydrogen constant, and the strict 1 M proton concentration ensures that the electrode’s potential does not fluctuate due to pH changes. Any deviation from the standard conditions — lower or higher acidity, reduced gas flow, altered temperature, or contamination of the platinum surface — would shift the equilibrium potential and eliminate its value as a reference.
The role of the Standard Hydrogen Electrode becomes clearer when you consider how it integrates into actual electrochemical measurements. To determine the electrode potential of another half-cell, such as Cu²⁺/Cu or Zn²⁺/Zn, that cell is connected to SHE and the resulting voltage is recorded under standard conditions. If electrons spontaneously flow from the metal electrode to the SHE, the metal electrode undergoes oxidation and its standard reduction potential is negative relative to zero. If electrons flow from the SHE to the other electrode, the metal ion is reduced and its standard reduction potential is positive. Through decades of experiments, this system has yielded the full electrochemical series covering metals, nonmetals, organic redox systems, biochemical redox pairs, and industrial electrolytes. Every value in that series, every prediction of whether a reaction will occur or whether a battery will supply useful power, depends on the existence of a globally uniform reference — the Standard Hydrogen Electrode.
Although SHE is conceptually straightforward and scientifically essential, it is seldom used outside controlled laboratory environments because maintaining pure hydrogen gas flow, a perfectly platinized electrode, and contamination-free acidic medium is not practical in routine measurements. Safer, more portable reference electrodes — such as the calomel electrode and silver/silver chloride electrode — are employed in most applications and calibrated relative to SHE. Yet even those substitutes retain meaning only because SHE defines the zero point to which they are compared. Thus, the Standard Hydrogen Electrode remains the theoretical backbone of electrochemical studies, linking every laboratory experiment, industrial process, and redox interaction to the same unchanging voltage scale.
The profound importance of SHE lies not in its external appearance — a simple hydrogen-bubbling beaker with a piece of platinum foil — but in its ability to anchor the entire framework of electrochemical energy. Batteries, fuel cells, metal corrosion, biochemical redox reactions, electrode plating, and industrial electrolysis all depend on predictable electron flow. By providing the absolute reference for electron potential, the Standard Hydrogen Electrode acts as the ruler of electrochemistry. It reveals how energetically substances compete for electrons, whether electrons move from metal to solution or vice versa, and how much work those electrons can perform once in motion. The deliberate precision of its structure and operating principles ensures that wherever electrochemistry is practiced — from academic laboratories to industrial energy research — all potentials speak the same universal language, measured from the constant and reliable zero established by the Standard Hydrogen Electrode.