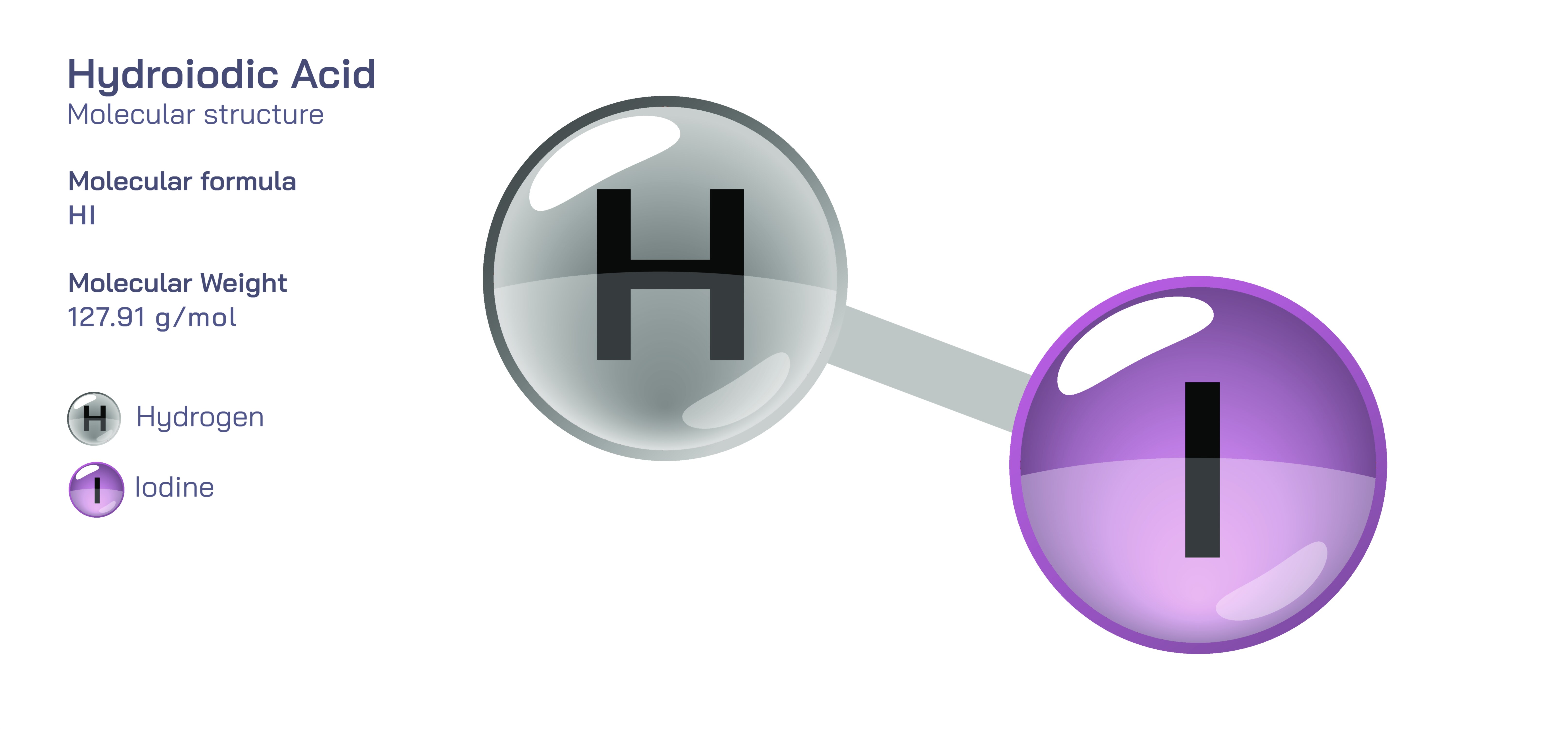
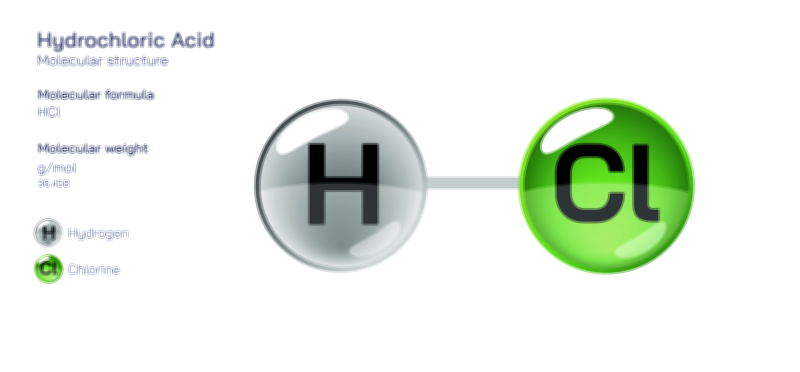
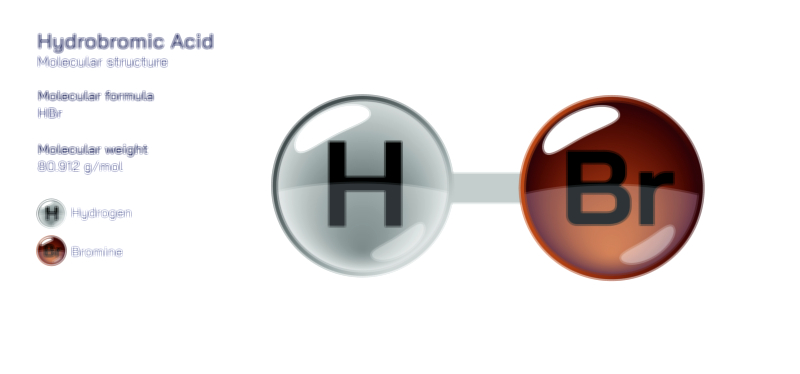
Hydroiodic Acid Chemical Structure — HI Molecule Properties Explained
Hydroiodic acid is one of the strongest and most chemically reactive acids known, and the best way to understand its extraordinary behavior is to begin with its simplest structural identity: at its core, hydroiodic acid is hydrogen iodide, a molecule composed of only two atoms, hydrogen and iodine. Although this formula appears simple, the structural arrangement, bond strength, atomic size contrast, electron distribution, and dissociation behavior give hydroiodic acid unique characteristics that set it apart from other hydrogen halides such as hydrochloric acid (HCl) and hydrobromic acid (HBr). In its gaseous form, hydrogen iodide (HI) exists as a diatomic molecule with a polar covalent bond, but when dissolved in water it transforms dramatically. The molecule undergoes nearly complete dissociation, separating into hydronium ions (H₃O⁺) and iodide ions (I⁻), forming hydroiodic acid. The ease with which this dissociation occurs stems from the chemical nature of the bond between hydrogen and iodine. Iodine’s large atomic radius and diffuse electron cloud weaken the H–I bond compared with its analogs in other hydrogen halides, making the molecule split with remarkable ease when water is present. This phenomenon explains why HI is not only a strong acid, but one of the strongest binary acids in existence — its structure inherently favors proton donation and stabilizes the resulting conjugate base.
The geometry and electron distribution of the hydrogen iodide molecule are responsible for its extreme acidity. Hydrogen, being very small, shares a bond with iodine, which is one of the largest nonmetal atoms on the periodic table. Because iodine’s electron cloud is so large and loosely held, the electron pair shared between hydrogen and iodine is drawn predominantly toward iodine. As a result, the H–I bond becomes highly polarized and energetically unstable. Water molecules, which are strongly polar and include lone electron pairs on oxygen, disrupt the bond immediately upon contact, pulling the hydrogen away as a proton while the iodine atom stabilizes the negative charge distributed across its extensive electron cloud. The iodide ion (I⁻) that forms after dissociation is one of the most stable anions in aqueous chemistry because its negative charge is highly delocalized. This stability is the reason hydroiodic acid is stronger than hydrochloric acid and hydrobromic acid; the more stable the conjugate base, the more strongly the corresponding acid donates protons. The structure of HI therefore drives its function: because the iodide ion is so stable, the molecule has little incentive to keep hydrogen bound, making proton donation practically automatic in solution.
These structural features translate directly to the chemical behavior of hydroiodic acid. Because it dissociates fully, hydroiodic acid produces high concentrations of hydronium ions, creating intense acidity capable of protonating even weak bases and rapidly breaking down oxides and carbonates. HI’s reducing power also sets it apart from many other mineral acids. The iodide ion, being highly polarizable and electron rich, can donate electrons in chemical reactions, meaning hydroiodic acid can function not only as a proton source but also as a reducing agent. This property becomes particularly significant in organic chemistry, where hydroiodic acid is used to convert alcohols to alkyl iodides or to remove oxygen-containing functional groups by replacing them with hydrogen. The reason HI accomplishes these reactions so effectively is that iodine can stabilize transitional negative charges during substitution and reduction. In industry and chemical synthesis, this dual identity as a strong acid and strong reducing agent makes hydroiodic acid invaluable, especially when powerful reactivity is needed under controlled conditions.
However, the same structural traits that give hydroiodic acid its chemical value also make it hazardous to biological tissue and the environment. When hydroiodic acid contacts living systems, the immediate release of protons causes corrosive burns that break down cellular components by denaturing proteins, dissolving lipid membranes, and disrupting DNA. Unlike weaker acids, which may act slowly, hydroiodic acid inflicts damage rapidly because its extremely high proton activity overwhelms biological buffering systems. The iodide ion itself is not corrosive, but because proton release happens instantly, tissue damage begins immediately upon exposure. Inhalation of hydrogen iodide gas or vapors of hydroiodic acid can lead to severe respiratory injury, as moisture in the airway converts the gas into concentrated acid on the sensitive tissue lining the lungs. Ingestion produces dangerous internal corrosive injury extending from the mouth and throat down the digestive tract. Even diluted solutions of hydroiodic acid possess strong acidity, which means accidental exposure of any kind requires immediate first aid with prolonged rinsing and medical treatment.
From a materials perspective, hydroiodic acid is equally aggressive. Metals, concrete, plastics, and many ceramic materials degrade under prolonged exposure depending on concentration and temperature. When HI reacts with certain metals, it can generate hydrogen gas, increasing the risk of flammability or explosion in confined spaces. The vapors themselves can corrode equipment and infrastructure, meaning hydroiodic acid must be stored in containers specifically engineered to resist its attack. Temperature control is another aspect of safe storage, because the hydrogen iodide molecule has a tendency to dissociate and release gaseous HI at elevated temperatures. This volatility requires careful handling, ventilation, and monitoring in laboratories and industrial facilities.
The reactivity of hydroiodic acid extends into environmental chemistry as well. If released into water systems, its acidity can rapidly lower pH and cause severe harm to aquatic organisms. Rapid protonation of biological molecules and oxidation–reduction interactions can disrupt metabolic processes in fish, microorganisms, and plant life. Thankfully, the iodide ion is relatively stable and, once the protonation damage is buffered by the environment, iodide becomes the dominant form and poses significantly less biological threat. Even so, proper disposal and regulation are essential to prevent ecological disruption. For this reason, hydroiodic acid is typically neutralized or converted to stable salts before disposal.
Because of its intensity, hydroiodic acid illustrates how molecular structure determines chemical identity and how this identity defines both practical applications and risks. The H–I bond, though simple in chemical notation, creates a chain of consequences because of its unusual weakness and the immense stability of the iodide ion. In the world of chemistry, HI ranks among the strongest proton donors not because of size, mass, or complexity, but because its structure makes proton loss effortless and energetically favorable. This characteristic enables significant scientific and industrial advances, particularly in synthesis routes where other acids fail due to insufficient reactivity. At the same time, it demands caution, expertise, and respect from anyone handling it, because its molecular behavior does not forgive mistakes or mishandling.
In the end, hydroiodic acid stands as a clear example of how structure at the molecular level governs everything that follows — its strength as an acid, its ability to act as a reducing agent, its corrosive effect on living cells and industrial materials, and its essential functions in chemistry laboratories and manufacturing. The hydrogen–iodine bond defines its entire personality: a weak bond that breaks easily in water, releasing hydrogen as a proton and leaving behind an exceptionally stable iodide ion. From this simple arrangement arises one of the most powerful acids known to chemistry, illustrating how the relationship between atoms determines not only how matter behaves but how humans must interact with it responsibly, strategically, and safely.