Octet Rule — Concept, Principles, and Applications in Chemistry
The octet rule is a fundamental principle in chemistry that explains how atoms achieve stability by having eight electrons in their outermost electron shell. This rule is based on the observation that atoms tend to be most stable when their valence shell is filled with eight electrons, resembling the electron configuration of noble gases. Understanding the octet rule is essential in predicting chemical bonding, molecular structure, and reactivity in elements.
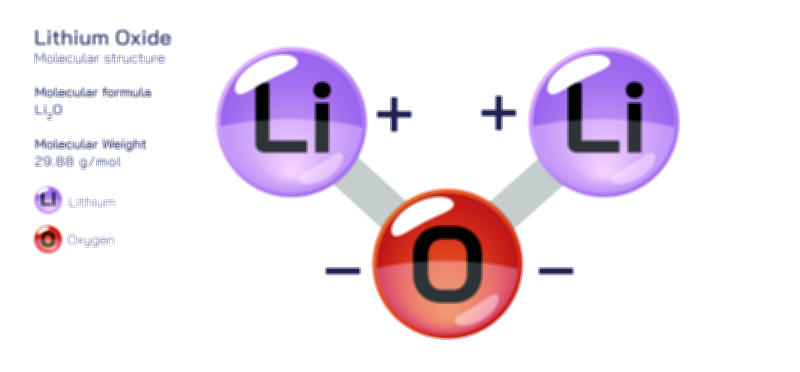
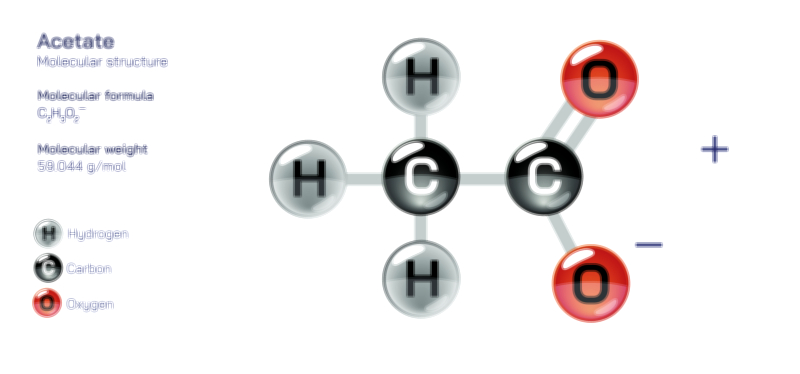
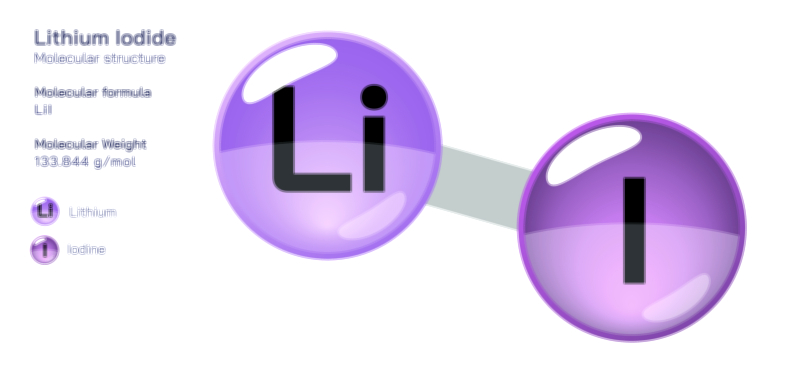
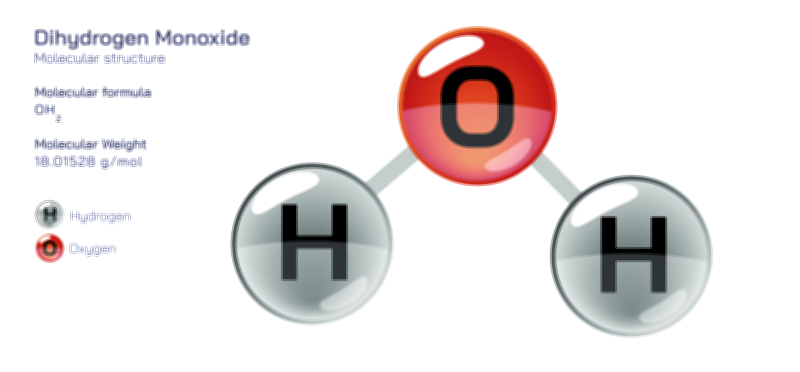
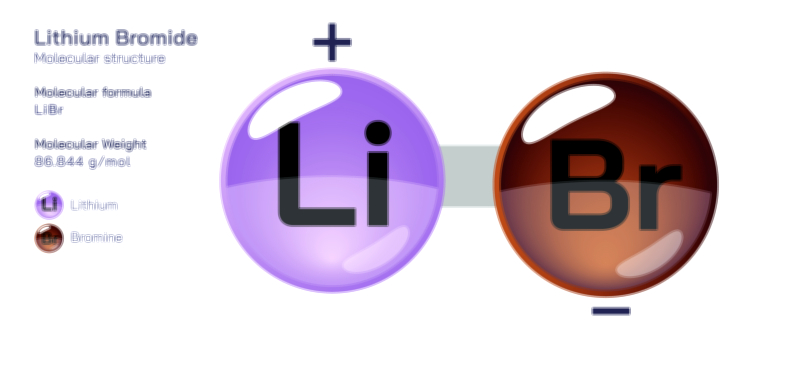
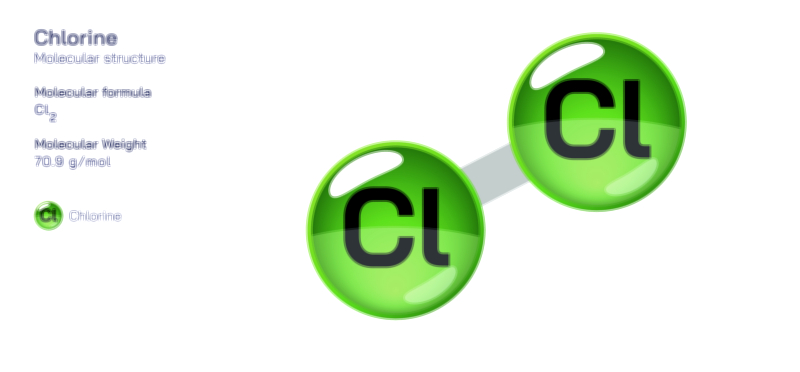
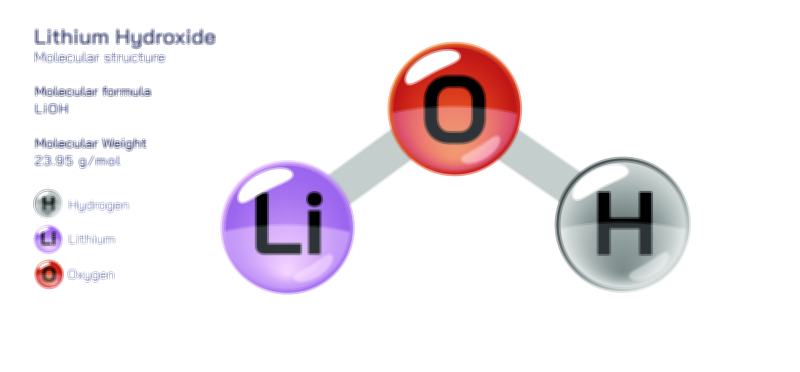
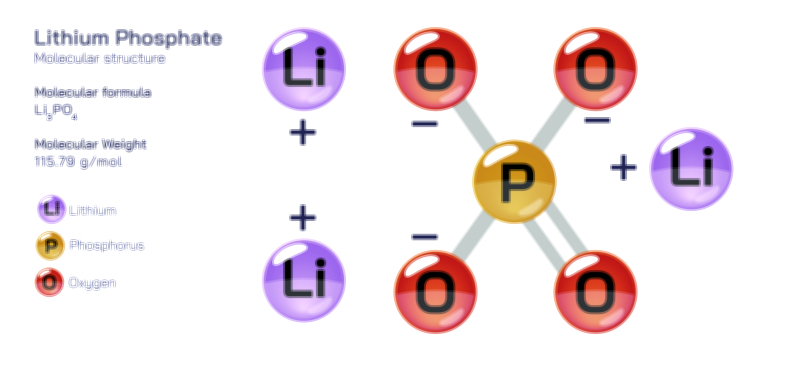
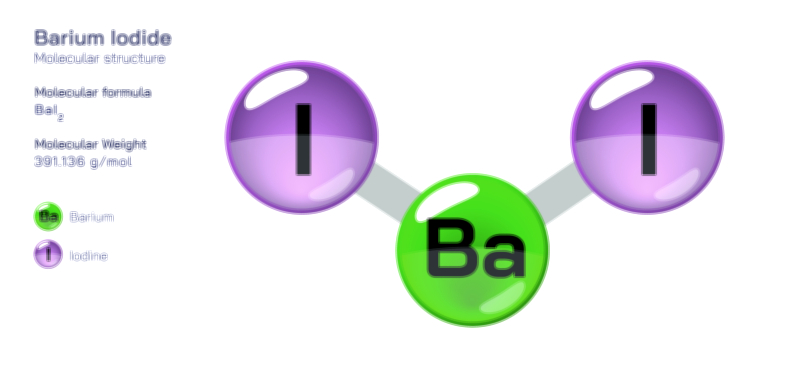
Atoms achieve a complete octet by gaining, losing, or sharing electrons. In ionic bonding, atoms transfer electrons to or from other atoms. Metals tend to lose electrons to form positively charged cations, while nonmetals gain electrons to form negatively charged anions. This electron transfer creates electrostatic attraction between the oppositely charged ions, resulting in stable ionic compounds. In covalent bonding, atoms share electrons with other atoms to complete their valence shells. Shared pairs of electrons allow each atom in the molecule to achieve an octet, forming stable covalent molecules.
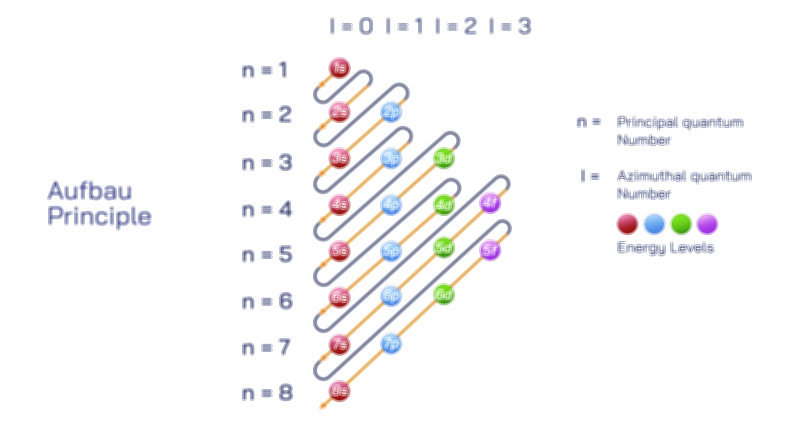
The octet rule also explains the behavior of various elements in the periodic table. While most main-group elements obey the rule, exceptions exist for hydrogen, helium, lithium, and beryllium, which achieve stability with two or four electrons in their outermost shell. Some elements in the third period and beyond can have an expanded octet, holding more than eight electrons, due to the availability of d-orbitals. The rule is widely used in drawing Lewis dot structures, predicting molecular shapes, and understanding chemical reactions.
Applications of the octet rule extend beyond simple compounds. It helps chemists design molecules, explain chemical reactivity, and predict molecular geometry using models like VSEPR theory. In organic chemistry, it guides the formation of functional groups and stable molecules. In inorganic chemistry, the rule explains ionic crystal formation and lattice energy. Understanding the octet rule is foundational for further studies in chemistry, including chemical thermodynamics, reaction mechanisms, and materials science.
In conclusion, the octet rule is a key concept that describes how atoms attain stability by completing their valence shell with eight electrons. It explains the formation of ionic and covalent bonds, electron transfer, and electron sharing, as well as predicting molecular structure and chemical reactivity. While there are exceptions, the octet rule remains central to understanding chemical bonding, molecular behavior, and the principles of modern chemistry, providing a framework for studying atoms, molecules, and reactions.