Chlorine — Halogen Chemical Element
Chlorine is one of the most influential and widely encountered halogen chemical elements, shaping human civilization, environmental chemistry, industrial production, biological systems, and public health in ways that stretch far beyond its simple atomic identity. Recognized by its place in Group 17 of the periodic table, chlorine occupies a unique intersection between high reactivity, essential biological roles in ionic form, and transformative applications across science and technology. In its elemental state, chlorine is a pale greenish-yellow gas with a sharp, assertive odor, instantly recognizable even in small concentrations. This gaseous form is profoundly reactive, and its behavior is rooted in the fundamental electronic structure that defines halogens: seven electrons in the outer shell, leaving the atom one electron short of achieving stable configuration. This powerful tendency to gain an electron governs chlorine’s interactions, shaping everything from the disinfecting of drinking water to the formation of salts, plastics, solvents, medicines, and even the biochemical functioning of the human body. To understand chlorine is to understand both the extraordinary versatility of halogens and the profound ways in which elemental chemistry intertwines with life and industry.
Chlorine’s atomic structure is central to its personality as a chemical element. With seventeen protons in its nucleus and a surrounding cloud of electrons arranged in shells that mirror halogen characteristics, it stands as a highly electronegative, electron-hungry element. This intense desire for stability drives chlorine to react vigorously with a wide range of substances, forming robust ionic and covalent bonds. In its diatomic molecular form, Cl₂, chlorine gas is composed of two chlorine atoms sharing electrons to complete their outer shells. This form is reactive enough to participate in oxidation processes that release significant energy, which makes the gas extremely useful industrially but also dangerous in uncontrolled environments. Chlorine’s nature as a powerful oxidizer explains why it is so effective at destroying microorganisms, bleaching materials, and participating in chemical transformations that create numerous compounds essential to modern life.
The most widespread and biologically significant form of chlorine is the chloride ion, the reduced and stable version of the element. Chloride ions are abundant on Earth, permeating oceans, soils, minerals, and living organisms. In seawater, chloride makes up a significant portion of dissolved salts, contributing to the chemistry of marine systems and influencing global weather patterns through evaporation and saltwater circulation processes. For living beings, chloride ions play irreplaceable roles in maintaining fluid balance, nerve impulse transmission, digestion, and respiration. In the human body, chloride is one of the primary electrolytes, working alongside sodium and potassium to regulate osmotic pressure and maintain hydration at the cellular level. It helps generate stomach acid, supports the movement of carbon dioxide in the bloodstream, and contributes to electrical stability in nerve cells. These essential functions show that while elemental chlorine gas is highly dangerous, its ionic form is indispensable to life, demonstrating the remarkable divergence in behavior that occurs through chemical bonding and oxidation-state shifts.
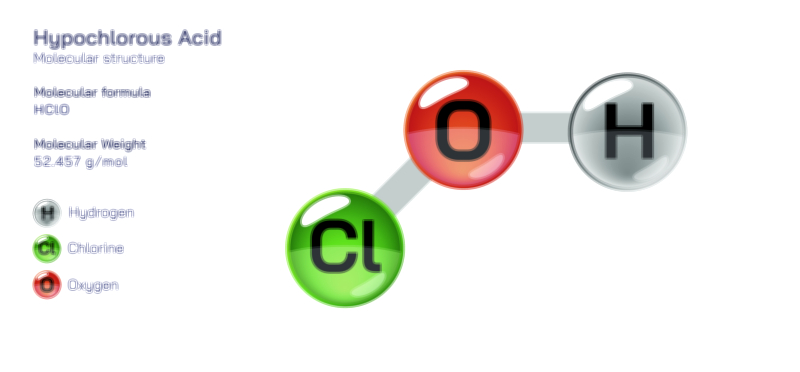
Chlorine’s industrial importance cannot be overstated. One of the most transformative aspects of its history is its role in water purification. During the early twentieth century, waterborne diseases such as cholera, typhoid, and dysentery claimed countless lives due to contaminated drinking water. When chlorine-based disinfection was introduced into public water supplies, the impact on global health was immediate and profound. Chlorine reacts with water to form hypochlorous acid, a powerful antimicrobial agent capable of destroying bacteria, viruses, and other pathogens. This simple chemical interaction revolutionized sanitation, drastically reducing disease rates and enabling large, growing populations to access safe drinking water. Today, chlorine-based disinfectants remain essential in municipal water treatment, swimming pool maintenance, and emergency sanitation efforts. The legacy of chlorine in public health underscores how a halogen element can shift human society by providing a reliable means to control infections that once devastated communities.
Beyond its disinfecting power, chlorine is fundamental to the chemical manufacturing industry. Its production through the electrolysis of brine—a process that also yields sodium hydroxide and hydrogen—supports the chlor-alkali industry, one of the foundational sectors in modern manufacturing. Chlorine serves as a precursor for the synthesis of countless compounds, including plastics, solvents, adhesives, coatings, pharmaceuticals, dyes, and advanced materials. Perhaps the most recognizable chlorine-derived material is polyvinyl chloride (PVC), a versatile polymer used in pipes, flooring, electrical insulation, medical equipment, packaging, and an array of consumer goods. PVC’s durability, chemical resistance, and adaptability in design stem directly from the properties chlorine imparts to the polymer. Without chlorine, vast sectors of construction, healthcare, and manufacturing would lose a material that has become integral to modern infrastructure.
Chlorine also plays a significant role in organic chemistry, where its reactivity allows it to substitute for hydrogen atoms in hydrocarbons, producing chlorinated organic compounds that serve as intermediates in the formation of more complex molecules. These compounds facilitate the synthesis of anesthetics, sterilants, pesticides, herbicides, pharmaceuticals, and flame retardants. Although some chlorinated organics have raised environmental concerns, particularly due to persistence or bioaccumulation, ongoing research and regulation have guided safer production and disposal methods, ensuring responsible use while preserving the benefits chlorine chemistry offers. The versatility of chlorine in molecular transformations highlights its importance in shaping both everyday consumer products and advanced scientific technologies.
In environmental systems, chlorine demonstrates a dual character. On one hand, chloride ions are harmless and abundant, contributing to natural cycles without posing toxicity at normal levels. On the other hand, reactive chlorine species can participate in atmospheric processes that influence air quality. Certain chlorine-containing compounds can contribute to smog formation or, in specific cases involving halogen radicals, participate in reactions that affect ozone concentrations. Though elemental chlorine itself is not a major stable component of the atmosphere, the chemistry associated with chlorine-containing substances informs important aspects of environmental science. Researchers study chlorine not only for its immediate industrial use but also for its influence on atmospheric processes, coastal chemistry, and interactions between seawater aerosols and sunlight.
In medicine, chlorine’s influence extends beyond water disinfection. Chlorine-containing compounds appear in numerous pharmaceuticals and antiseptics. Some antibiotics, antifungals, and antiviral drugs incorporate chlorine atoms to enhance stability, strengthen molecular binding to biological targets, or modify pharmacokinetics. The presence of chlorine in medicinal chemistry showcases the element’s ability to alter molecular behavior in ways that support therapeutic design. Meanwhile, chlorine-based disinfectants are indispensable in hospital environments, surgical settings, and laboratories, where sanitation must be maintained at the highest standard.
Chlorine’s history includes cautionary lessons that reflect the broader theme of scientific responsibility. During World War I, chlorine gas was weaponized and used on the battlefield, causing devastating injuries due to its reactivity with moisture in the lungs, forming corrosive compounds that impaired breathing. Although global agreements now prohibit such use, this grim past remains part of chlorine’s legacy, reminding society that powerful chemistry must be managed with moral and regulatory oversight. Most modern handling of chlorine gas now occurs under strict safety protocols involving containment systems, gas detection, ventilation, protective equipment, and emergency preparedness. Industrial facilities storing or transporting chlorine implement rigorous controls to prevent accidental exposure, understanding that while chlorine is essential, it must be handled with respect.
In materials science, chlorine’s role extends to etching, bleaching, and surface treatment. Its ability to break down color-bearing compounds makes it a powerful bleaching agent used historically in paper production, fabric treatment, and cleaning formulations. While alternative bleaching technologies have emerged, chlorine-based compounds remain significant in contexts where strong oxidizing power is required. Its use in metallurgy further expands its impact; chlorine can help purify metals, extract desirable elements from ores, or remove impurities in high-temperature processes.
Culturally, chlorine has shaped modern expectations of cleanliness and hygiene. The scent of chlorinated pools, the assurance of safe tap water, and the widespread use of disinfectants all reflect chlorine’s presence in daily life. In households, chlorine-derived cleaners and sanitizing agents contribute to food safety, infection control, and general maintenance. While most individuals may only encounter chlorine indirectly, the reliability and safety of modern sanitation systems depend deeply on its chemical behavior.
Ultimately, chlorine stands as a remarkable example of how a single element can embody opposing yet complementary qualities—harmful in one form, life-sustaining in another; highly reactive as a gas, essential and stable as an ion; dangerous when uncontrolled, indispensable when applied responsibly. Its chemistry is foundational not only to scientific understanding but also to the functioning of global infrastructure, public health systems, biological life, and vast industrial networks. Chlorine’s story reveals how the periodic table is more than a chart of atoms—it is a tapestry of elements whose behaviors influence every aspect of existence, demonstrating that even the most reactive substances can become keystones of civilization when knowledge, respect, and scientific skill converge.