Aufbau Principle Showing Electron Configuration, Atomic Orbital Filling Order, and Energy Level Distribution
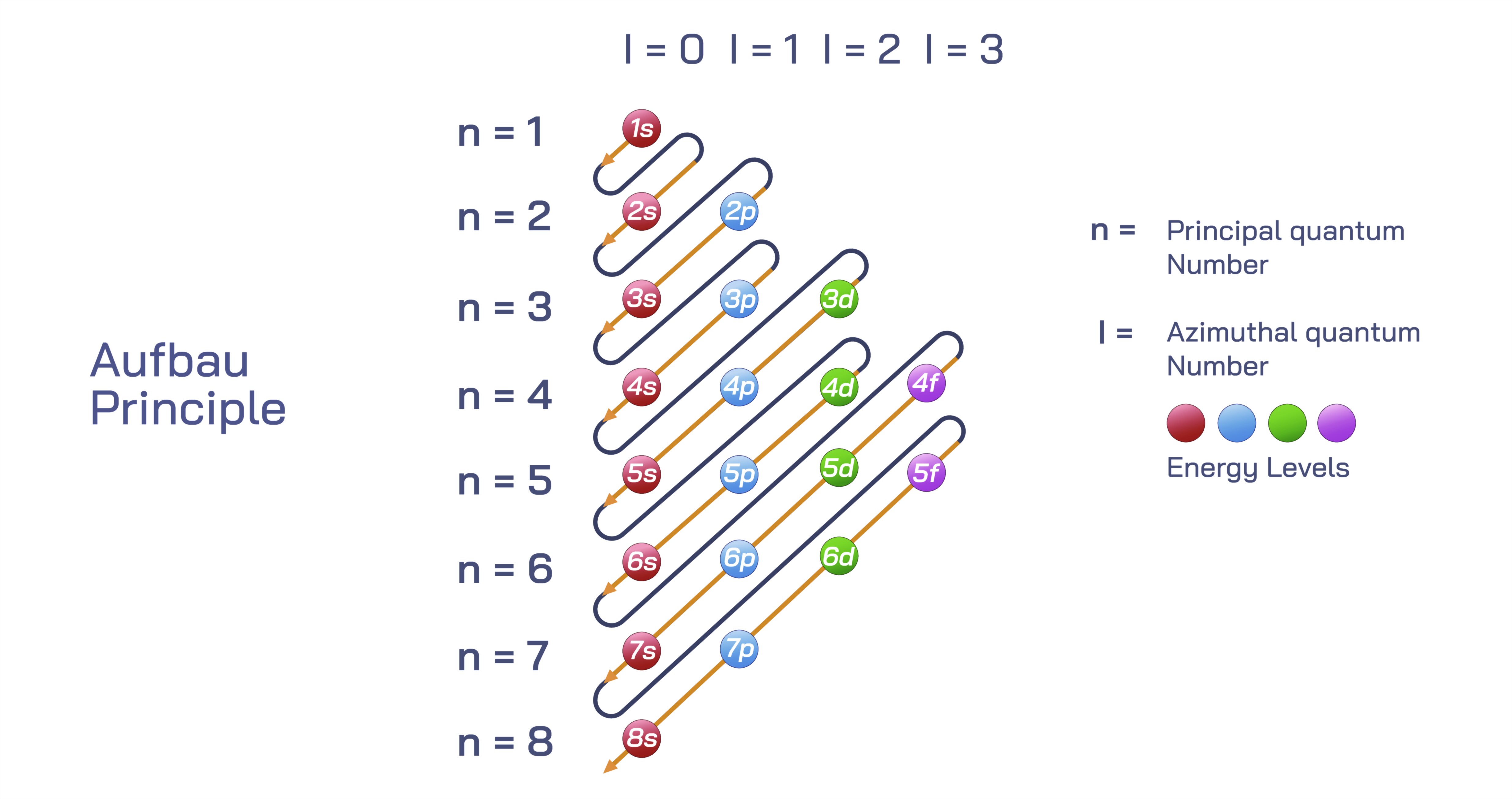
The Aufbau principle is one of the most fundamental concepts in atomic chemistry because it explains how electrons gradually occupy the available orbitals of an atom in an organized and predictable manner as the atomic number increases. The term comes from the German word “Aufbau,” meaning “building up,” and that meaning reflects its scientific purpose: with every additional proton placed into the nucleus of a growing atom during the evolution of the periodic table, an extra electron joins the electron cloud surrounding the nucleus, and it must occupy a space defined by energy requirements. Electrons do not enter orbitals randomly, nor do they scatter themselves in regions of equal probability throughout the atom. Instead, their positions follow a specific order based on relative energy levels, stability patterns, and quantum rules that govern their distribution. Understanding the Aufbau principle gives students a deep appreciation of atomic structure, chemical reactivity, the layout of the periodic table, and the nature of bonding, because it reveals how atoms establish their electron configuration and why that configuration determines the identity and behavior of each element.
In a neutral atom, the nucleus attracts electrons through electrostatic forces, but electrons also repel one another. This interplay of forces creates a structured, layered arrangement of orbitals and shells. Each orbital represents a region of space where electrons are most likely to be found, and each orbital has a unique energy level associated with it. According to the Aufbau principle, electrons fill the lowest available energy orbitals first before moving to higher ones. This ensures that atoms achieve the most stable configuration possible with the least amount of energy expenditure. Even though the periodic table appears flat and orderly in two dimensions, the actual distribution of electrons requires imagining three-dimensional wave patterns arranged around the nucleus. These patterns are grouped into shells or principal energy levels and subdivided into sublevels identified as s, p, d, and f. Each sublevel has one or more orbitals that can accommodate a limited number of electrons, and so electron configuration unfolds progressively, one orbital at a time, following this energy ladder.
The Aufbau principle is often represented visually through a diagram in which diagonal arrows cascade downward across the s, p, d, and f orbitals. Although this graphic is abstract, its message is straightforward: the next available electron follows the arrow to the next lowest energy position rather than simply moving to the next shell numerically. This is why electrons do not always fill orbitals in numerical order such as 1s, 2s, 2p, 3s, 3p, 3d, and so on. Instead, orbitals overlap in energy based on their distance from the nucleus and their spatial probability distributions. As a result, some higher numbered shells are actually lower in energy than sublevels of shells closer to the nucleus. Because the Aufbau principle requires electrons to reside in the lowest possible energy state, orbitals such as 4s may fill before 3d, even though 3d belongs to the third principal shell. This critical detail not only reveals how electrons behave but also demonstrates why the periodic table has its unique block structure, where the s-block, p-block, d-block, and f-block correspond to the filling of these orbital groups.
The principle becomes especially meaningful when considering the Pauli exclusion rule and Hund’s rule, because they shape how electrons distribute inside each orbital and sublevel. Although the Aufbau principle explains the order in which sublevels fill, the Pauli rule states that no two electrons in the same atom can have identical sets of quantum numbers, meaning that each orbital can hold only two electrons and these electrons must differ in spin direction. Meanwhile, Hund’s rule teaches that when multiple orbitals of the same energy are available, such as the three p orbitals or five d orbitals, electrons fill them individually first, maintaining parallel spin to minimize repulsion before pairing up. Together, these rules govern the fine structure of electron configuration and ensure that electrons occupy positions that maximize stability, minimize repulsion, and maintain the lowest possible energy.
As atomic number increases across the periodic table, electron configuration becomes more complex, yet it still adheres to the same foundation. Hydrogen begins building with one electron in the 1s orbital. Helium completes the first shell with two electrons. Lithium begins the second shell with 2s, and then elements progress across the s and p orbitals until neon fills the 2p sublevel. Sodium begins the 3s orbital, and the pattern repeats. Even when the d and f orbitals enter the picture, seeming to disrupt the pattern numerically, the order remains consistent with energy priority. The filling of 4s before 3d explains why the transition metals behave chemically differently from s-block elements and why the d-block sits in the middle of the periodic table. When the lanthanides and actinides appear in the f-block, their underlying electron configurations follow the same building principle, though they involve more complex overlap patterns between f, d, and s orbitals.
The phenomenon of stability emerges clearly through the Aufbau principle when examining noble gases. Each noble gas completes an electron shell, giving it a configuration that is extraordinarily stable due to a fully occupied set of low-energy orbitals. Other elements strive, through chemical bonding, to achieve similar stability by sharing, losing, or gaining electrons. This is why the Aufbau principle is far more than a theoretical or mathematical guideline; it becomes the foundation that explains why atoms participate in chemical reactions at all. Atoms with incomplete valence shells seek to reach lower energy states, while those with filled valence shells maintain their structure with little tendency to react. The architecture of these filling patterns explains periodic properties such as electronegativity, ionization energy, electron affinity, oxidation state tendencies, and patterns in atomic and ionic radii.
Because the Aufbau principle describes the way electrons populate orbitals as atomic number increases, it becomes a blueprint for reading and predicting electron configurations. Every electron added to a neutral atom transforms its energy distribution and chemical personality. The predictable sequence in which orbitals fill gives meaning to the arrangement of the periodic table itself. Each row completes a major shell, each block represents a sublevel pattern, and each column showcases a recurring electron configuration in the valence shell. This explains why elements in the same group exhibit similar chemical behaviors: they share the same number and arrangement of valence electrons, even though their inner electron structures grow increasingly complex as new orbitals fill.
Even in ions, the Aufbau principle influences behavior, but with special exceptions that emerge when the delicate balance of orbital energy shifts. Transition metal ions often lose electrons first from the 4s sublevel rather than from the 3d sublevel after both have been filled, because electron repulsion and shielding effects alter the energy ordering when electrons begin to populate the d orbitals. These details demonstrate that although the Aufbau principle provides the baseline filling order, electron occupation is dynamic and sensitive to subtle changes in energy landscape as electrons are added or removed. Nonetheless, the core principle remains consistent: electrons always occupy the most energetically favorable arrangement available.
The Aufbau principle therefore acts as the organizational backbone of atomic chemistry. It explains why the periodic table has the shape that it does, why atoms form particular ions, why they bond in certain ways, and why elements express repeating patterns in their chemical properties. It connects the quantum mechanical interpretation of atomic orbitals with the observable behavior of matter. By understanding how electrons fill orbitals layer by layer, one gains a deeper understanding of the intricate yet beautifully ordered nature of atoms, where energy minimizes itself step by step to build up the rich diversity of chemical elements that define the material world.