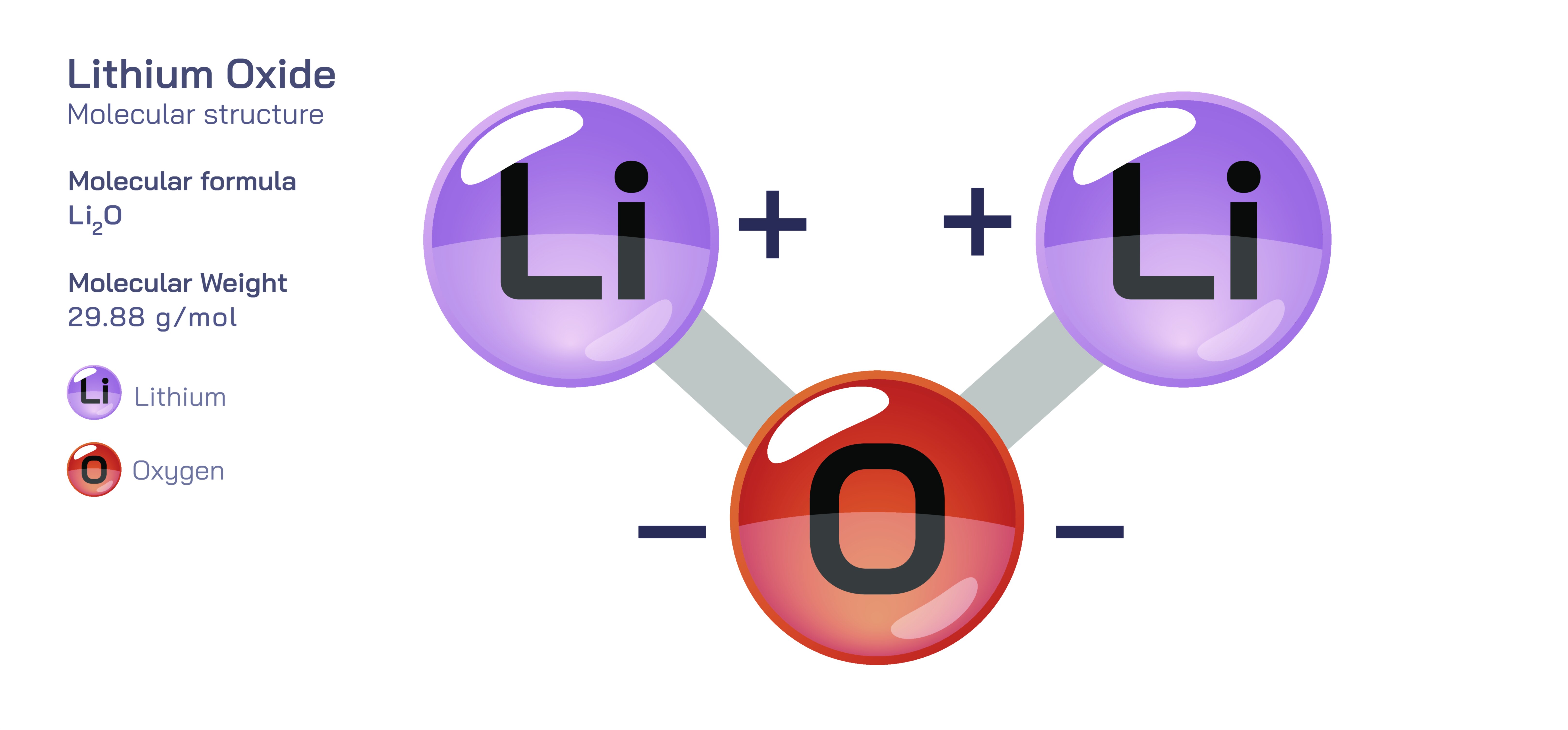Lithium Oxide — Chemical Structure, Physical and Chemical Properties, Reactivity, Industrial Importance, Ceramic and Glass Applications, and Role in Lithium-Ion Technology
Lithium oxide is an inorganic compound with the formula Li₂O, consisting of two lithium cations (Li⁺) bonded to one oxide anion (O²⁻). Although it appears to be a simple binary oxide, its chemistry and applications extend deeply into materials science, ceramics engineering, metallurgy, energy storage, and battery manufacturing. Among lithium compounds, Li₂O occupies a unique position because it is not only a product of high-temperature reactions involving lithium salts and lithium hydroxide, but also a crucial intermediate used to manufacture advanced materials—particularly those that rely on light weight, thermal robustness, ionic mobility, and structural stability. Lithium oxide’s significance arises from the intersection of its ionic bonding, strong basicity, high reactivity with water, and the ability of the Li⁺ ion to influence melting behaviour, hardness, crystal structure, and electrochemical processes.
Structurally, lithium oxide adopts a cubic antifluorite arrangement in the solid state. In this lattice, oxide ions sit in a face-centred cubic arrangement while lithium ions occupy all tetrahedral interstitial positions. The oxide ion, being doubly charged and larger than lithium, dominates the geometric arrangement, while the lithium ions are distributed throughout the lattice to balance charge. The antifluorite structure is particularly notable for its ionic mobility and symmetrical packing, enabling Li₂O to behave not only as a stable oxide but also as a material capable of supporting ion transport under elevated temperatures. Its melting point is high, its structure is rigid, and its density is low due to the extremely light atomic mass of lithium. The lattice configuration also influences how Li₂O behaves when incorporated into glassy, ceramic, or molten matrices, often modifying viscosity, thermal expansion, and crystallinity.
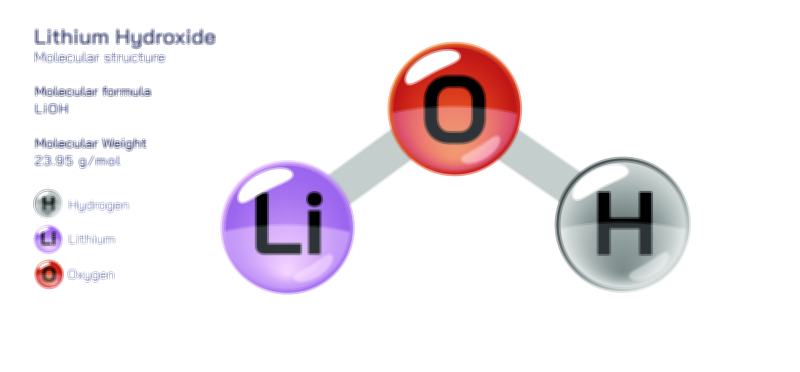
Lithium oxide is typically produced industrially through the thermal decomposition of lithium carbonate (Li₂CO₃) or lithium hydroxide (LiOH), a process that releases carbon dioxide or water vapour. This reaction requires high heat because lithium salts are thermally stable, and the resulting Li₂O appears as a white, hygroscopic crystalline solid. Its behaviour in air and moisture immediately reveals its strong basicity. Lithium oxide reacts vigorously with water to form lithium hydroxide:
Li₂O + H₂O → 2 LiOH
This reaction is highly exothermic, illustrating how strongly Li₂O seeks to bind water through the lithium cation’s intense hydration energy. Because of this reactivity, lithium oxide is not stored in humid environments and is handled in sealed or controlled atmospheres in high-purity industrial settings. In the presence of carbon dioxide, Li₂O absorbs CO₂ to form lithium carbonate, making it chemically useful in gas-capture and purification systems designed to remove acidic gases from closed atmospheres. This property forms the basis of research into Li₂O and lithium ceramic materials for CO₂ sorption, regenerative air purification, and spaceflight life-support systems.
One of the most significant industries that depend on lithium oxide is the ceramics and glass industry. When Li₂O is incorporated as a flux or modifier in glass batches, it reduces the melting temperature dramatically, improves flow characteristics of the melt, and enhances the formation of uniform microstructures. This makes glass processing more energy-efficient while improving clarity and mechanical strength. Lithium oxide also increases resistance to thermal shock, making it valuable for cookware, laboratory glassware, heat-resistant windows, stove-top surfaces, and specialty architectural glass. In ceramics, lithium oxide influences crystal growth and sintering dynamics, allowing production of glazes and porcelain materials that are smoother, stronger, and less prone to cracking during firing. Lithium compounds derived from Li₂O are used in lithium aluminium silicate (LAS) and spodumene-based ceramic systems, which are known for exceptional thermal stability and are widely used in aerospace, cookware, and electronic substrates.
Beyond glass and ceramics, Li₂O plays a crucial role in metallurgy, especially in the refining and alloying of non-ferrous metals. When added to molten phases, lithium oxide enhances slag fluidity, reduces viscosity, and improves removal of impurities during metal purification. In aluminium production, it helps lower operating temperatures and increases bath stability. The strong affinity between lithium and oxygen also makes Li₂O useful in metal-surface treatments and high-temperature industrial lubricants, where lithium soaps resist oxidation and maintain performance under mechanical stress. The thermal strength and reactivity of lithium oxide make it a valued additive in high-temperature metallurgical processing where conventional fluxes are insufficient.
Lithium oxide is also at the core of advanced nuclear and fusion energy research. Compounds derived from Li₂O are investigated as breeder materials for nuclear fusion reactors. When exposed to high-energy neutrons, lithium atoms can react to produce tritium—an essential fuel for future fusion power systems. Lithium ceramics based on Li₂O are engineered to contain lithium isotopes in forms that promote tritium production, heat transfer, and radiation stability. The oxide’s low atomic number and high thermal conductivity make it a candidate for fusion blanket materials, contributing to both energy generation and structural heat management. Although fusion technology remains under development, the importance of lithium oxide to this field has made it the subject of significant materials-science research.
In the rapidly expanding energy-storage sector, lithium oxide also plays an indirect but critical role. Many lithium-ion cathode materials—including layered oxides (NCM, NCA), spinel oxides (LMO), and high-voltage solid-solution cathodes—are manufactured using lithium oxide precursors or intermediate lithium-oxide-forming reactions. Even when lithium carbonate or lithium hydroxide is used as the starting material, the high-temperature synthesis of cathode powders often proceeds through transient Li₂O phases that dictate final structure and electrochemical performance. In solid-state batteries, thin lithium-oxide-based interlayers can enhance adhesion, ionic conductivity and interface stability between lithium metal anodes and solid electrolytes. The presence of Li₂O at grain boundaries or interphases influences dendrite suppression, charge cycling efficiency and thermal resilience, further underscoring how the oxide participates in energy systems not only as a chemical compound but as a structural and ionic mediator.
Lithium oxide also appears in chemical manufacturing, catalysis and reagent chemistry. Because it is a strong base and an oxygen donor, Li₂O is used to adjust alkalinity in industrial chemical reactors, to modify melt characteristics in high-temperature catalytic systems, and to synthesize specialty lithium salts. It has been investigated for ceramic-supported catalysts, solid-base catalysis in biodiesel conversion, and heterogeneous catalytic systems where its basicity and thermal stability contribute to reaction selectivity.
Safety, handling and storage of lithium oxide require care because of its strong reactivity with water and carbon dioxide. Direct contact can irritate skin and eyes due to conversion into caustic lithium hydroxide. High-purity industrial processing therefore relies on sealed containers, inert or moisture-controlled environments and corrosion-resistant materials. Despite its hazards at the point of handling, its behaviour is predictable and manageable within engineered systems, enabling safe industrial usage on large scales.
Lithium oxide ultimately is a compound that reveals how a small, light alkali metal paired with oxygen can produce an industrial material of disproportionate global importance. Its antifluorite structure dictates ion mobility and reactivity; its strong basicity explains moisture sensitivity and gas-absorption capability; its ability to modify glass and ceramic networks leads to advanced heat-resistant materials; and its interactions with transition-metal oxides facilitate the creation of high-performance battery cathodes. As industries continue to transition toward renewable energy, electrified transportation, thermally stable advanced materials and potentially fusion-based power, Li₂O remains positioned not as a common oxide but as a cornerstone of modern materials technology.
Through its chemical framework and technological influence, lithium oxide demonstrates how atomic-scale properties can expand into global industrial transformation, shaping energy storage, materials science, and high-temperature engineering across the world.