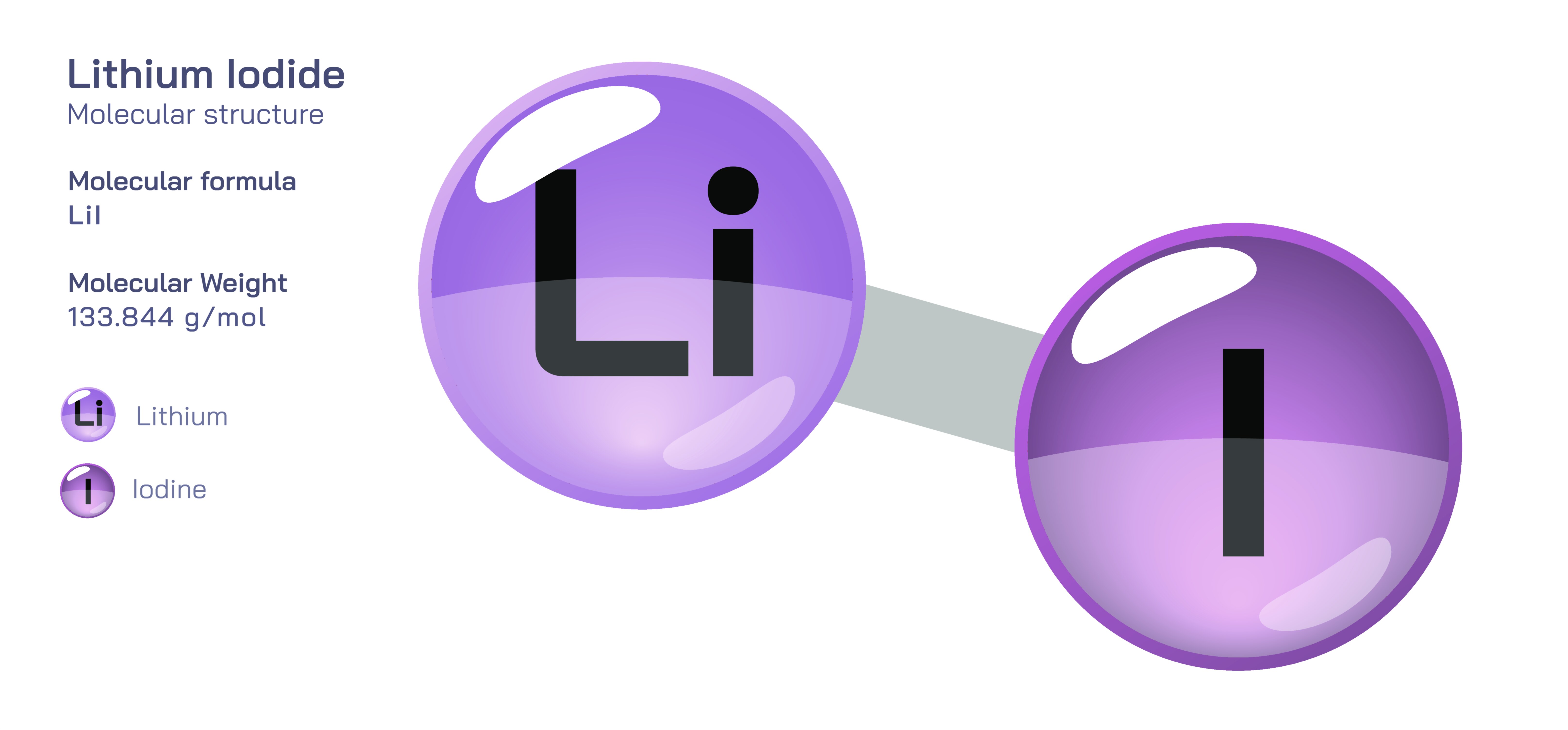
Lithium Iodide — Chemical Structure, Physical and Chemical Properties, Hygroscopic Behaviour, Electrochemical Role, and Industrial & Technological Applications
Lithium iodide is an inorganic compound with the formula LiI, composed of a lithium cation (Li⁺) and an iodide anion (I⁻). Although chemically simple in appearance, its behaviour is defined by the extreme softness and polarizability of the iodide ion paired with the exceptionally small ionic radius and strong solvation tendency of the lithium ion. This combination produces a salt that is highly soluble, strongly hygroscopic, and unusually reactive in certain environments. Lithium iodide holds relevance across diverse fields, including solid-state battery technology, organic synthesis, pharmaceutical chemistry, radiation detection, optical materials, and high-performance electrolytes. The compound illustrates how the pairing of a very light alkali metal with a very heavy halogen creates chemical, physical and industrial characteristics that differ sharply from other lithium halides, making LiI especially valuable to energy storage science, optical engineering, and specialized analytical systems.
In its crystalline state, lithium iodide adopts a structure typical of ionic halide salts but with particular features governed by size mismatch between lithium and iodide ions. The solid exists mainly in the rock-salt (NaCl-type) cubic lattice, where each lithium ion sits at the centre of an octahedron of six iodide ions, and each iodide ion is surrounded similarly by lithium ions. Because Li⁺ has one of the smallest cationic radii in the periodic table, while I⁻ is one of the largest common anions, the lattice spacing is governed more by the size of the iodide ions than the lithium ions. This creates a structural arrangement that is compact yet flexible, allowing the lattice to crack or cleave with relative ease and making LiI sensitive to hydration. Exposure to atmospheric moisture triggers rapid deliquescence, meaning the solid absorbs water until it dissolves. This behaviour reflects the strong hydration energy of lithium ions and the ability of iodide ions to polarize surrounding molecules. As a result, lithium iodide must be stored in airtight containers to prevent spontaneous moisture uptake.
Lithium iodide dissolves readily in water and in various polar organic solvents. When dissolved, the ions do not behave as passive dissociated species; rather, lithium becomes tightly surrounded by multilayer hydration shells while iodide remains strongly polarizable. The dissolution process releases heat due to solvation of the small, highly charged lithium cation. While simple to dissolve, LiI also participates in complex equilibria involving polyiodide species when excess iodine is present, forming compounds like LiI₃ and LiI₅ that display unique optical and electrochemical properties. This tunability between simple ionic salt and polyiodide network materials contributes to lithium iodide’s utility in batteries, spectroscopy, and certain optical devices.
One of the most consequential modern applications of lithium iodide is in battery and electrolyte science, especially in solid-state lithium-iodine batteries and lithium-air battery research. Lithium iodide can serve as a solid electrolyte because of the mobility of lithium ions within its lattice at elevated temperatures and its ability to support ion transport when engineered into crystalline or polymer-rich composite matrices. In lithium-iodine batteries—used historically in long-life medical implants such as pacemakers—the iodine cathode reacts with lithium to form lithium iodide progressively during discharge. Because LiI itself becomes the solid electrolyte, the battery maintains stable internal resistance for extremely long periods, allowing decade-scale functionality in implanted devices where battery failure is unacceptable. The extreme reliability of these systems pushed lithium iodide to the forefront of biomedical energy storage before newer rechargeable chemistries emerged. Even today, lithium-iodine battery designs are studied for ultra-long-life and low-drain applications, especially in remote or implantable systems.
Lithium iodide has also been identified as an important component in next-generation rechargeable energy research. In lithium-air batteries, iodine-based redox mediators—including lithium iodide—help overcome sluggish oxygen reduction and evolution reactions, improving rechargeability and decreasing electrode passivation. Additionally, LiI incorporated into polymer electrolyte matrices enhances ionic conductivity and mechanical stability at room temperature. Although these technologies remain developmental, lithium iodide continues to be investigated because its large anion and strong polarizability mediate electron transfer in ways that smaller halides (like fluorides or chlorides) cannot replicate.
Another significant area of application is radiation detection, particularly in scintillation detectors. When doped with europium or thallium, crystalline lithium iodide becomes a scintillating material capable of converting incident X-ray and gamma radiation into detectable visible light. These detectors are valued in geophysical surveying, medical imaging instrumentation, nuclear safety systems, and astrophysical research because of their sensitivity and energy resolution. Because lithium contains the isotope ⁶Li, which is highly efficient at absorbing thermal neutrons, LiI-based detectors are also employed for nuclear monitoring and neutron measurement systems.
Lithium iodide holds considerable importance in optics and infrared technology. It has one of the highest refractive indices among alkali halides and excellent transparency in both visible and infrared spectral regions. These characteristics make LiI valuable in IR windows and prisms, laser systems, atmospheric spectroscopy and certain types of thermal imaging equipment. However, its strong hygroscopicity restricts use to protected environments, limiting its role to sealed optical modules or controlled laboratory systems rather than open-air field devices.
In organic synthesis, lithium iodide serves as a selective and versatile reagent. The iodide ion is a soft nucleophile, and its large size facilitates substitution reactions, particularly in converting alkyl chlorides and alkyl bromides into more reactive iodides. LiI can also act as a catalyst in acylation and condensation reactions, especially in polar aprotic solvents where lithium coordinates to carbonyl oxygen atoms while iodide activates electrophilic centres. These dual ionic behaviours allow the reagent to accelerate transformations used in pharmaceutical syntheses, polymer chemistry and fine-chemical manufacturing. In addition, lithium iodide influences solubility of cellulose and polysaccharides in amide-based solvents, supporting research and product development in biopolymers, nanocellulose, film formation and material science.
Lithium iodide also appears in heat-storage mixtures and molten-salt systems. When blended with lithium chloride or lithium bromide, it forms eutectic salts with low melting points and large energy-storage capacities suitable for thermal management in solar-thermal systems, high-temperature heat-transfer loops and certain industrial heating units. Its use in such systems depends on its ionic heat capacity and molten-phase conductivity, properties that stem from the mobility of lithium ions and the polarizability of iodide.
Handling lithium iodide requires careful attention because the compound absorbs moisture rapidly and becomes corrosive in concentrated solution. Prolonged contact with skin or eyes can cause irritation, and ingestion or inhalation of dust is harmful due to lithium’s reactivity and biological effects. Industrial uses therefore involve sealed circulation systems, corrosion-resistant materials and controlled atmospheres when high purity is required for battery or optical applications. Proper storage, ventilation and protective equipment prevent unexpected deliquescence and chemical exposure.
Viewed as a whole, lithium iodide is a compound whose global importance is defined not only by its structure but by its ability to transition across multiple scientific and technological domains. The Li⁺ ion contributes strong solvation, ionic mobility and electrolyte behaviour; the I⁻ ion contributes softness, polarizability and optical response. Together, these characteristics enable lithium iodide to support long-life energy storage, neutron and gamma detection, infrared optics, redox mediation, synthetic chemistry and molten-salt energy systems. Through the interaction of its ions, its hygroscopic nature, its electrochemical versatility and its role in high-precision industrial applications, lithium iodide stands as one of the most valuable halide compounds in advanced science and modern technology — demonstrating how atomic-level behaviour expands into global material innovation and energy progress.