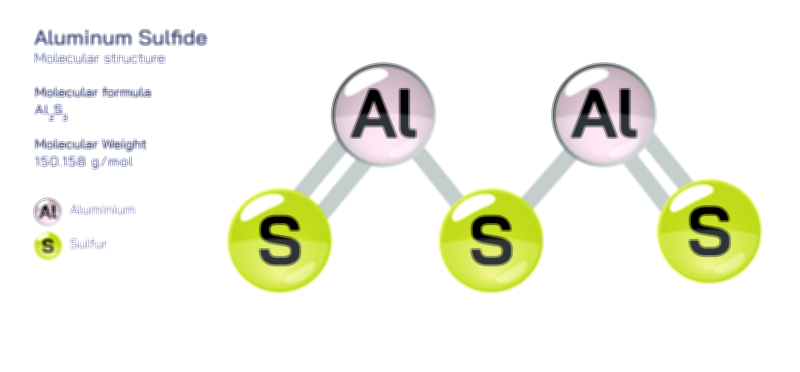
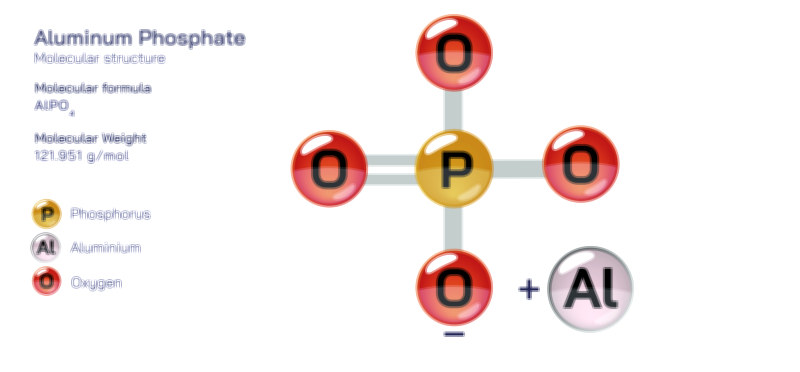
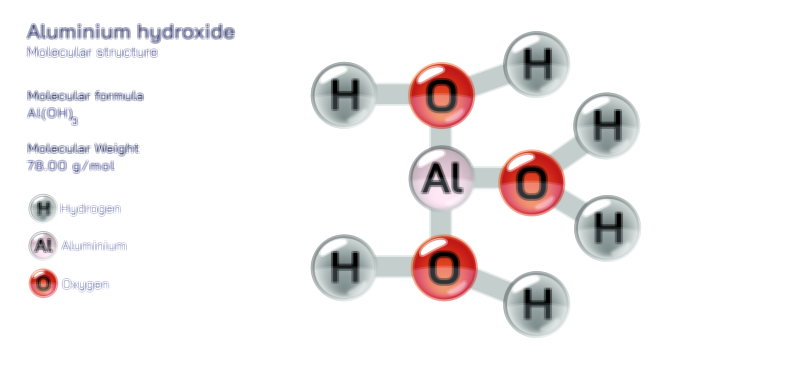
Aluminum molecule chemistry, especially when explored through the lens of atomic structure and bonding, reveals a unique interplay of metallic characteristics, electron configuration, and compound formation that makes aluminum one of the most versatile elements in both natural and industrial contexts. Although aluminum is often discussed as a metal rather than in the form of discrete “molecules,” the term can be interpreted in the broader chemical sense to include the atomic-scale interactions, clusters, ionic compounds, and coordination structures formed by aluminum atoms with other elements. Understanding aluminum at this level requires beginning with its fundamental atomic structure. Aluminum, with atomic number 13, has an electron configuration of 1s² 2s² 2p⁶ 3s² 3p¹, meaning it possesses three electrons in its outermost shell. These valence electrons determine how aluminum participates in bonding, as the atom tends to lose all three to achieve a stable neon-like configuration. This predisposition to form Al³⁺ ions influences nearly all of its chemical behavior, including its ability to create ionic lattices, covalent frameworks, metallic bonds, and complex coordination geometries depending on the chemical environment.
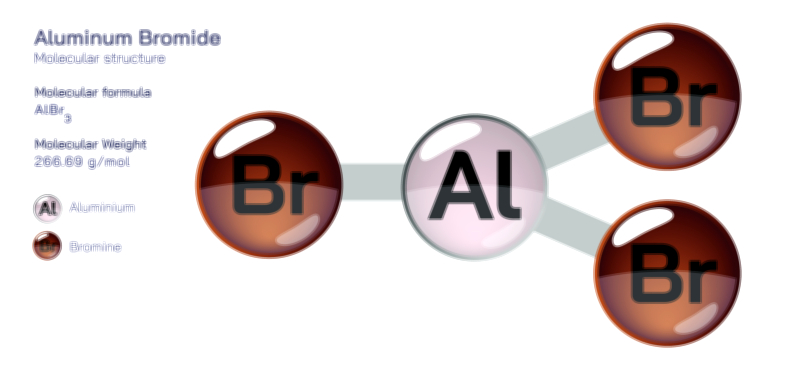
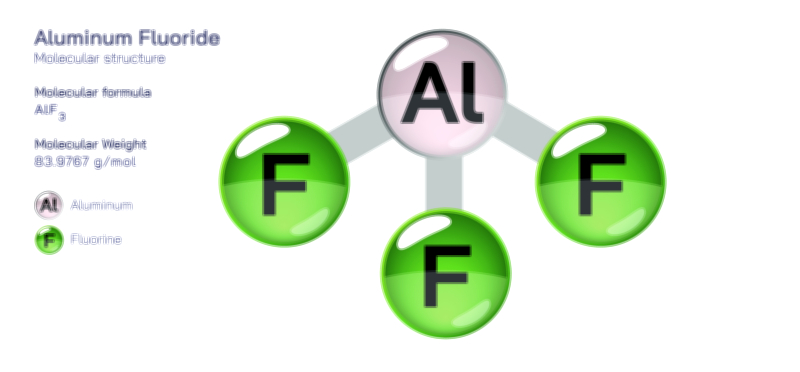
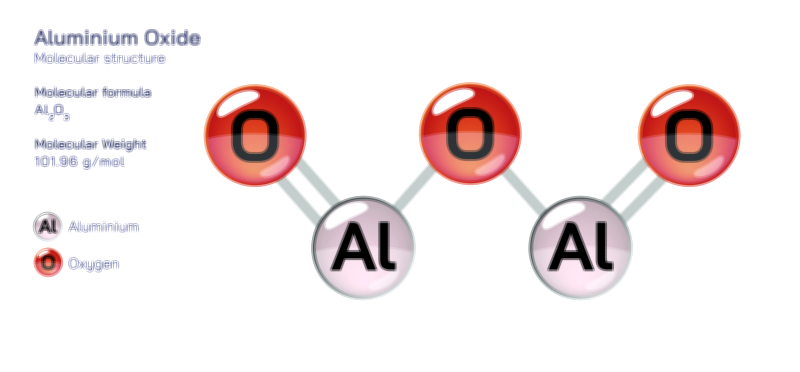
When aluminum atoms are examined individually, their metallic bonding tendencies are rooted in the delocalization of electrons. However, when we shift toward compound chemistry, the picture becomes richer and more varied. In ionic compounds such as aluminum oxide, the aluminum cation strongly attracts electronegative oxygen atoms, forming a structure that is both rigid and extraordinarily stable. This explains why aluminum oxide naturally forms a passive protective layer over metallic aluminum surfaces, preserving the metal underneath from further oxidation. This oxide layer showcases the dual nature of aluminum bonding: although aluminum atoms themselves favor metallic bonding, their interaction with nonmetals produces durable ionic networks. In other contexts, aluminum forms covalent or partially covalent bonds, particularly when bonded to elements like chlorine, fluorine, or carbon. Aluminum chloride, for example, is known for its interesting behavior; in its solid state it adopts an ionic lattice, but in gaseous or molten states it exists as a covalent dimer, Al₂Cl₆, illustrating how aluminum’s bonding character shifts fluidly depending on temperature and phase.
The concept of aluminum “molecules” becomes even more intriguing when considering aluminum clusters—groups of aluminum atoms bonded in metallic but molecule-like forms. These clusters exhibit properties distinct from bulk aluminum metal and demonstrate that aluminum can participate in bonding patterns that bridge the gap between metallic solids and molecular chemistry. Aluminum hydrides further extend this chemical diversity. Compounds such as alane (AlH₃) show covalent characteristics, featuring networks of aluminum and hydrogen bound in electron-deficient frameworks. These electron-sharing patterns differ significantly from the purely ionic behavior observed in aluminum salts, revealing aluminum’s ability to adopt various bonding arrangements depending on electron availability and external forces.
In coordination chemistry, aluminum frequently acts as a central metal ion surrounded by electron-donating ligands. Because Al³⁺ is small and highly charged, it strongly attracts lone-pair-bearing atoms such as oxygen, nitrogen, or fluorine. This results in the formation of octahedral complexes where six ligands stabilize the aluminum ion. These complexes are vital to both biological and industrial systems. In aqueous environments, the Al³⁺ ion becomes surrounded by water molecules, forming hexaaquaaluminum complexes that influence the acidity, solubility, and reactivity of aluminum-containing solutions. This same coordination behavior explains why aluminum salts are widely used as flocculating agents in water purification.
Studying aluminum’s bonding patterns also illuminates its reactivity. Because the Al³⁺ cation is so stable, aluminum often participates in reactions that involve electron donation or acceptance leading to the formation or dissolution of this ion. This chemical property is central to aluminosilicate minerals, the most abundant group of minerals in the Earth’s crust, where aluminum atoms integrate into complex three-dimensional frameworks with silicon and oxygen. These structures demonstrate how aluminum’s preference for threefold or fourfold coordination reshapes entire mineral families and geological processes.
Altogether, aluminum molecule chemistry provides an expansive view of how a single element can manifest diverse bonding behaviors—from metallic delocalization to ionic attraction, from covalent electron sharing to intricate coordination networks. By examining its atomic structure and the behavior of its valence electrons, we see why aluminum forms such an extraordinary range of compounds and materials that influence everything from construction alloys to catalysts, electronics, environmental processes, and even planetary geology.