ØÉüØÉÜØɽØÉóØÉ«ØÉ” ØÉłØÉ©ØÉØØÉóØÉØØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉÜØɦØÉØ ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×, ØÉāØÉ×ØÉ×ØÉ® ØÉŗØÉ©ØɦØÉĀ ØÉäØÉ▒ØÉ®ØÉźØÉÜØɦØÉÜØÉŁØÉ©ØɽØÉ▓ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
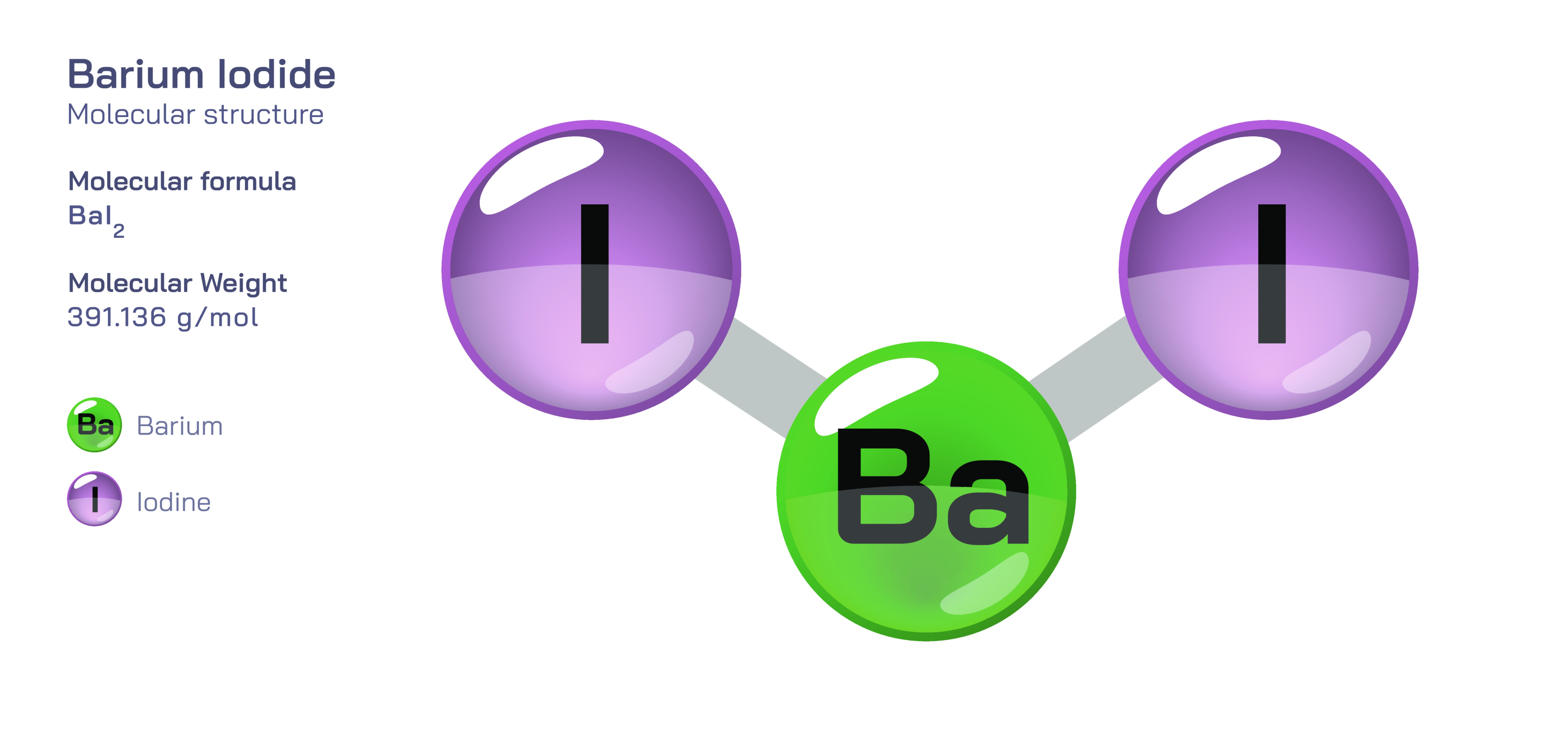
Barium iodide is an inorganic compound known primarily for its highly ionic nature, distinctive crystalline characteristics, and strong solubility in polar solvents. With the chemical formula BaIŌéé, it consists of a divalent barium cation (Ba┬▓Ōü║) bonded to two monovalent iodide anions (IŌü╗). The compound exists in both an anhydrous form and a hydrated crystalline form, most commonly as barium iodide dihydrate (BaIŌéé┬Ę2HŌééO). At first glance, barium iodide appears to be a simple metal halide, but its molecular and structural characteristics reveal important insights into chemical bonding, crystal organization, solvation behaviour, and interactions between metal ions and halogens. The structure is largely governed by the strong electrostatic attraction between barium and the iodide ions surrounding it, giving the compound high stability in ionic lattices yet allowing it to dissolve readily in water because hydration effectively separates and stabilizes the charged species.
On the structural level, barium iodide consists of a barium ion positioned at the centre of a three-dimensional ionic framework surrounded symmetrically by iodide ions. The Ba┬▓Ōü║ ion is relatively large among alkaline earth metals, and the IŌü╗ ion is the largest halide ion, which influences the geometry of the ionic lattice. In the solid state, anhydrous barium iodide adopts a layered arrangement in which each barium ion is coordinated by several iodide ions in a geometry optimized for minimizing lattice energy. The large ionic radii of both species mean that the packing is less densely compact than seen in compounds involving smaller halides such as chloride or fluoride. The symmetry of this structure ensures even distribution of electrostatic forces throughout the lattice, which is why the solid has a well-defined crystalline appearance and high melting strength. In its hydrated form, water molecules enter the lattice and form hydrogen-bond networks with iodide ions while also interacting with the barium cation, which leads to a slightly altered lattice geometry without compromising the compoundŌĆÖs core ionic identity. Hydration stabilizes the structure by lowering lattice energy and improving solubility.
The ionic nature of barium iodide explains its dissolution behaviour in water and other polar solvents. When BaIŌéé crystals come into contact with water, the strong dipole of the water molecules surrounds and separates the ions: water molecules orient oxygen toward the barium ion because of partial negative charge, while hydrogen atoms orient toward the iodide ions because of partial positive charge. This hydration process weakens and ultimately breaks the ionic bonding forces holding the lattice together. As a result, barium iodide dissociates completely into free Ba┬▓Ōü║ and IŌü╗ ions in aqueous solution. Once dissolved, the ions behave independently and do not remain clustered as intact BaIŌéé molecular units. This dissociation is important chemically because the behaviour of the compound in solutionŌĆöits reactivity, conductivity, and interaction with other ionsŌĆödepends on the free availability of Ba┬▓Ōü║ and IŌü╗ rather than the compound as a whole. Solutions of barium iodide conduct electricity strongly, demonstrating the mobility of charged species.
The structural composition of barium iodide also influences its chemical reactivity. The iodide ion is easily oxidized, meaning it can undergo electron-transfer reactions with oxidizing agents. Barium iodide therefore becomes a valuable precursor for generating elemental iodine in laboratory reactions: when treated with oxidizing substances, IŌü╗ ions release electrons and convert into molecular iodine, which appears visibly as a purple vapour or a brownish solution. Meanwhile, the Ba┬▓Ōü║ ion is chemically reactive toward sulfate, carbonate, and chromate ions because the resulting barium salts of these anions are insoluble. This property has analytical value, especially in precipitation reactions where the formation of characteristic white barium sulfate crystals helps identify the presence of sulfate ions. Thus, the molecular identity of barium iodide in solution is not passive: it actively participates in ionic exchange, precipitation, redox processes, and salt conversion, reflecting a deeper connection between structure and behaviour.
The steric features and ionic arrangement of barium iodide define its optical behaviour and materials-science relevance as well. Barium halides including barium iodide have been explored as components of scintillation detectors due to their optical transparency and sensitivity to radioactive energy when fabricated into doped crystals. The transparency arises from well-ordered ionic lattices that minimize defects that scatter light, while the iodine component contributes to efficient absorption of high-energy photons. Although barium iodide is not the most common scintillator material, understanding its structure has contributed to advancements in crystal engineering across related compounds. Its high solubility also makes it an important precursor in synthesis of other barium-based materials and catalytic systems.
Biologically and environmentally, the structural attributes of barium iodide influence toxicity and handling. The Ba┬▓Ōü║ ion has toxic effects on the nervous and muscular system when present in free form because it interferes with potassium ion channels that regulate muscle contraction. However, toxicity depends on the solubility of the barium compound: highly insoluble salts like barium sulfate pass harmlessly through the body, but soluble salts like barium iodide can release Ba┬▓Ōü║ ions readily into blood and tissues if ingested. This difference demonstrates that toxicity is an extension of chemical structureŌĆöthe same element can be safe or hazardous depending on how tightly it is bound in the compound. Iodide ions themselves are physiologically important in controlled amounts as they support thyroid hormone synthesis. But in barium iodide, the association between the ions and their dissociation in body fluids means that ingestion poses a risk due to the free barium ions released rather than the iodide component. Therefore, the compound requires careful handling in laboratory or industrial settings, even though iodide salts of other metals may be benign.
Industrial and synthetic applications of barium iodide are tied to its structure-driven chemical roles. It acts as a convenient source of iodide ions for organic and inorganic synthesis, allowing substitution reactions, halogenation processes, and redox transformations that rely on high iodide reactivity. It is also used as a precursor to produce highly pure barium compounds for ceramic manufacturing, specialty glasses, and electrical materials. In research, the compound serves as a reagent in molecular spectroscopy and analytical chemistry due to the predictable behaviour of iodide in light absorption and photochemical reactions. Each of these applications traces back to the strong ionic identity of the compound, demonstrating how a basic arrangement of Ba┬▓Ōü║ and IŌü╗ can influence reactivity across many scientific fields.
The molecular structure of barium iodide therefore explains each aspect of the compoundŌĆöits crystalline form, reactivity, solubility, chemical roles, optical functions, and toxicity. The pairing of a large alkaline earth metal ion with a large halide ion produces an ionic lattice that is stable yet readily dissociated, reactive yet predictable, and industrially valuable yet biologically risky if not controlled. The bariumŌĆōiodide bond is not a single pair of shared electrons but an electrostatic attraction maintained through the precise geometric organization of ions, which is why the compound is more accurately understood as an ionic network rather than a discrete molecular unit. Every behaviour of the compoundŌĆöfrom the structure of its crystals to the ion exchange reactions in solutionŌĆöemerges from this fundamental ionic architecture. Barium iodide serves as a vivid example of how the arrangement of atoms determines a compoundŌĆÖs role in chemistry and in the world.