Lithium Phosphate — Chemical Structure, Physical and Chemical Properties, Industrial Relevance, Biological Considerations, and Expanding Role in Modern Energy and Materials Technology
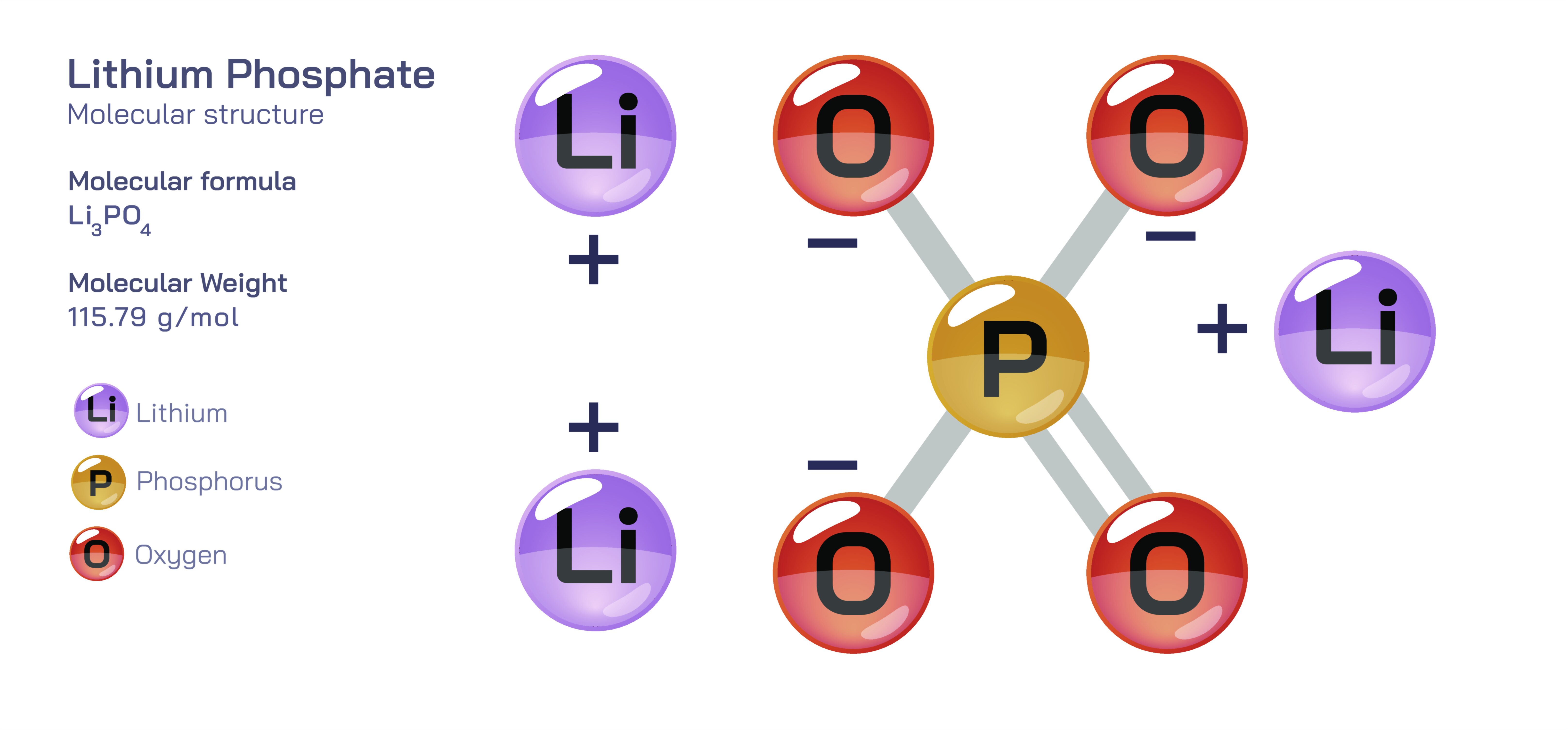
Lithium phosphate is an inorganic compound with the formula Li₃PO₄, consisting of three lithium cations (Li⁺) electrostatically bonded to a phosphate anion (PO₄³⁻). Although it may appear to be a simple crystalline salt at first glance, its behaviour places it at the intersection of chemistry, materials science, environmental technology, electrochemistry, and biomedical research. The pairing of the lightest alkali metal with one of the most structurally influential anions in inorganic chemistry produces a compound that is remarkably stable, chemically resilient, and capable of participating in advanced industrial and technological systems. Lithium phosphate exemplifies how the underlying ionic framework and lattice geometry of a compound can allow it to function as both a passive structural material and an active component in chemical, electrical, and biological environments.
In the solid state, lithium phosphate exists as a crystalline ionic network in which phosphate tetrahedra (PO₄³⁻) serve as rigid units surrounded by lithium ions that occupy charge-balancing positions in the lattice. This geometry produces a highly stable structure with strong covalent bonding within the phosphate groups and ionic bonding between phosphate tetrahedra and lithium ions. The compound is white, odorless, and thermally robust, remaining stable at high temperatures without decomposition and resisting dissolution in many common solvents. Unlike many lithium salts that are highly soluble and hygroscopic, lithium phosphate is only sparingly soluble in water. This relatively low solubility is the result of strong ionic packing and the triply charged PO₄³⁻ group, which anchors lithium ions into a network that is not easily disrupted by hydration forces. This stability defines many of its industrial and technological uses because it creates an environment where lithium is immobilized, predictable and resistant to unwanted release.
The production of lithium phosphate typically involves the neutralization of lithium hydroxide or lithium carbonate with phosphoric acid, yielding Li₃PO₄ as a precipitate. The process is straightforward but requires precise control of temperature and pH to ensure phase purity. Because the compound is chemically durable and non-hygroscopic, it remains stable in dry air and does not absorb moisture like lithium chloride or lithium bromide. It melts only at high temperatures, showing resistance to thermal shock and structural breakdown. At elevated temperatures, however, lithium phosphate can participate in complex silicate or ceramic phase reactions, a behaviour that underpins its importance in glass-ceramic technology and industrial kiln materials. In such systems, Li₃PO₄ acts as a fluxing agent, reducing melting temperatures and increasing fluidity, but unlike lithium oxide or lithium carbonate, it moderates thermal expansion while enhancing structural stability. This makes lithium phosphate valuable for producing heat-resistant glass, enamels, glazes, and ceramic components that must maintain dimensional stability across wide temperature ranges.
One of the most transformative applications of lithium phosphate appears in electrochemical energy storage, particularly in the context of lithium iron phosphate (LiFePO₄ or LFP) batteries. In contemporary battery technology, Li₃PO₄ is not typically used directly as the cathode material, but it serves as a precursor, source of phosphate, and stabilizing additive in the synthesis of LFP. Lithium iron phosphate—created by integrating Li₃PO₄ with iron sources under controlled sintering conditions—forms a cathode material famous for its excellent thermal stability, long cycle life, high current-handling ability, and intrinsic safety. Unlike cobalt- or nickel-based cathodes, LiFePO₄ is resistant to thermal runaway and degradation at elevated temperatures, making it especially valuable for electric vehicles, grid-scale energy storage, solar battery banks, and consumer electronics that require long-term reliability. Lithium phosphate’s structural stability and phosphate-rich composition are essential for achieving the olivine crystal framework that gives LFP batteries their durability and low risk of combustion. As global energy systems shift toward electrification and renewable storage, lithium phosphate has become a strategically important industrial compound even if it does not appear in the final cathode formulation.
Lithium phosphate also finds application in solid electrolytes and ion-conducting ceramic systems, where modified forms—such as Li₃PO₄ doped with halogens or metal ions—facilitate lithium-ion movement at high ionic conductivity. These engineered materials appear in research prototypes for solid-state lithium batteries, where the electrolyte must withstand high voltages while preventing dendrite growth and maintaining physical contact with the electrode surfaces. Phosphate-based solid electrolytes are valued for their electrochemical stability windows, nonflammability, and mechanical rigidity. Although commercialization of solid-state systems is still developing, lithium phosphate remains a critical research material in electrolyte formulation, interphase coatings, and cathode protective layers designed to improve battery cycle life.
Beyond energy storage, Li₃PO₄ is used in metallurgical processes and corrosion control. When incorporated into surface treatments, phosphate compounds form protective coatings that reduce metal oxidation and enhance adhesion of paints or thermal barrier layers. Lithium phosphate is sometimes included in specialty corrosion-inhibiting formulations where the lithium ion enhances coating uniformity and improves crack resistance. Additionally, in certain lithium-based glasses and optical materials, Li₃PO₄ contributes to controlled refractive index modification and enhanced resistance to devitrification, allowing production of optical lenses, fiber materials, and precision ceramic substrates.
In biomedical and biochemical contexts, lithium phosphate participates in a different kind of significance. Lithium compounds have long been recognized for their neurological effects, particularly in controlling mood disorders. While lithium carbonate is the most common therapeutic lithium salt, lithium phosphate has historically been investigated as a controlled-release form of lithium due to its lower solubility and gradual ionic dissociation. Although it is not widely used clinically today, research continues to examine how Li₃PO₄ can be incorporated into biomaterials to slowly release lithium ions for neural protection, controlled delivery, and scaffold integration in tissue engineering. The phosphate component also aligns with biological systems, as phosphates are a natural part of bone and cellular energy pathways. For this reason, lithium phosphate has been studied in the development of bioactive ceramics and bone-regeneration materials, where controlled lithium release may promote osteogenesis and influence cellular signalling pathways.
Lithium phosphate is generally safe to handle when used in industrial settings due to its low solubility and chemical stability. However, it is still an alkaline salt, and contact with eyes or prolonged contact with sensitive skin may cause irritation. Ingestion of excessive amounts can introduce lithium ions into the body at hazardous levels, requiring controlled handling when used in laboratory or chemical environments. In energy and ceramics manufacturing, Li₃PO₄ is handled using sealed containers to prevent dust migration, and appropriate ventilation is implemented during high-temperature synthesis because of the potential release of reaction vapours.
What makes lithium phosphate scientifically and technologically remarkable is the way its inherent chemical traits scale into real-world impact. The rigid phosphate structure creates stability that can be transferred into industrial glass and ceramic systems. The immobilized lithium ions within the lattice become mobile under engineered conditions, enabling ion-conducting battery materials. The low solubility of Li₃PO₄ becomes an advantage in biological and pharmacological applications that require gradual lithium release. The high-temperature robustness of lithium phosphate supports metallurgical and catalytic processes that demand thermal reliability.
In a world where electrification, materials longevity, and sustainable engineering shape global infrastructure, lithium phosphate—despite its chemical simplicity—holds a place of profound strategic importance. Whether serving as a precursor for world-leading LFP battery cathodes, a stabilizing agent in engineered ceramics and optical materials, a functional participant in next-generation solid-state electrolytes, or a controlled-delivery source of lithium ions in biomedical applications, Li₃PO₄ exemplifies how a single inorganic compound can influence multiple fields of science and technology. Its value lies not only in what it is, but in what it enables: safer batteries, stronger materials, efficient industrial systems, and emerging innovations that rely on stability, ion coordination, and controlled reactivity at both molecular and macroscopic scales.