Diffusion and Effusion — Gas Movement Concept
Diffusion and effusion represent two fundamental concepts in the study of gases, describing how gas particles move, mix, and escape through openings in ways that reveal the underlying behavior of matter at the microscopic level. Although these ideas are often introduced in chemistry and physics classes through simple examples, their conceptual depth extends far into scientific, biological, industrial, and environmental systems. Both diffusion and effusion arise from the natural, constant motion of gas particles, which move freely, rapidly, and randomly in all directions. This motion represents the fundamental characteristic of gases: their molecules are widely spaced, experience minimal intermolecular forces, and therefore disperse spontaneously. Understanding diffusion and effusion conceptually offers insight into why smells travel across a room, how gases mix in the atmosphere, how lungs exchange oxygen and carbon dioxide, why balloons slowly deflate, how industrial separators work, and how the motion of particles reflects deeper physical principles such as randomness, distribution, and energy.
To begin understanding diffusion, picture a scenario in which a gas is released into a space filled with air. As soon as the gas molecules enter that space, they start spreading out, mixing with the surrounding molecules until the mixture becomes uniform. This process does not require stirring, external forces, or mechanical movement. Instead, diffusion happens because each gas particle is in constant thermal motion, colliding with air molecules and bouncing away in new directions. Every collision redirects a molecule, and over time, these countless random movements cause the molecules to disperse and spread evenly. Even if two different gases start in separate regions, they will eventually mix completely and uniformly. This natural mixing happens because particles tend to move from regions where they are more concentrated to regions where they are less concentrated, gradually evening out the distribution. Importantly, diffusion is not driven by “intent” or any directional force; it results purely from random, microscopic motion that produces macroscopic effects. This concept underlies a huge portion of natural and technological processes, demonstrating the power of particle movement in shaping the world we experience.
One of the most relatable examples of diffusion comes from everyday sensory experiences. When someone opens a bottle of perfume or when food cooks in a kitchen, the scent molecules gradually spread throughout the room. Even though only a small amount of the scented substance is released initially, the molecules disperse far from their source, filling the entire space with detectable aroma. This dispersal happens because molecules constantly collide, rebound, and migrate in all directions. Eventually, their concentration becomes even across the room, explaining why the smell becomes noticeable everywhere. Similar diffusion processes occur when smoke spreads, when gas leaks disperse in enclosed spaces, and when air freshener sprays distribute their fragrance. All of these are visible or sensory illustrations of microscopic collisions creating large-scale effects.
Diffusion is not limited to gases; it also occurs in liquids and even solids, although far more slowly because particles are more restricted. However, gases exhibit the fastest diffusion because they have the greatest freedom of motion. This high mobility underlies environmental processes such as the mixing of atmospheric gases, the dispersal of pollutants, the spread of volcanic emissions, and the distribution of oxygen in aquatic environments. The atmosphere itself is a giant demonstration of diffusion: nitrogen, oxygen, carbon dioxide, water vapor, and trace gases all mix uniformly around the planet due to constant molecular movement. Without diffusion, the atmosphere would form layers of separate gases rather than the well-mixed air essential for life.
Diffusion is equally vital in biology. Every breath a person takes relies on diffusion to exchange gases in the lungs. Oxygen diffuses from the air inside the alveoli into the bloodstream, while carbon dioxide diffuses in the opposite direction. This life-sustaining transfer happens because differences in concentration drive gas movement across thin biological membranes. In cells, diffusion guides the movement of nutrients, signaling molecules, and waste products. Plants depend on diffusion for carbon dioxide intake during photosynthesis and for oxygen release. Animals depend on diffusion for transport across cell membranes, within tissues, and between bodily fluids. Even processes like nutrient absorption in the intestines and neurotransmitter movement across synapses are guided by diffusion principles. These biological examples show how deeply the concept of diffusion is woven into the most essential operations of living systems.
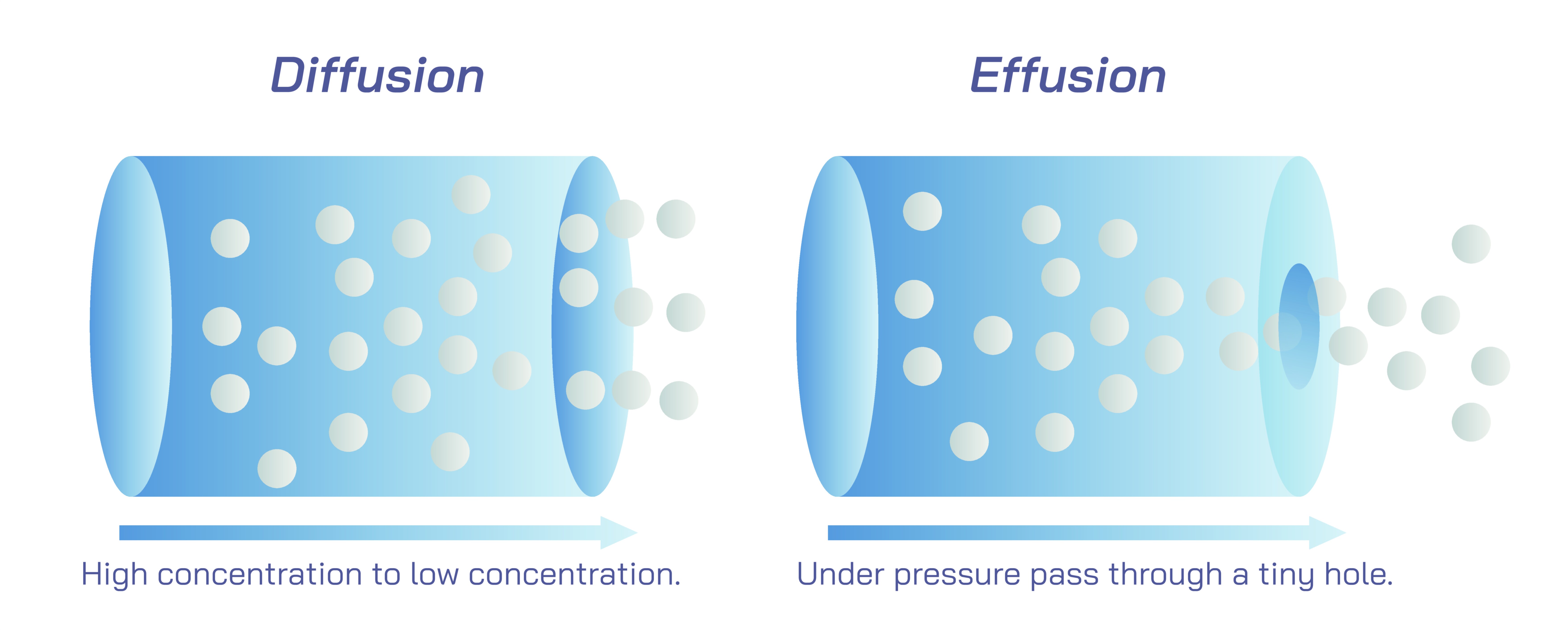
Effusion, although related to diffusion, refers to a different movement behavior. While diffusion describes how gas molecules spread throughout a space, effusion describes how gas molecules escape through a small opening into an empty or less pressurized area. For example, when a gas leaks slowly from a small hole in a balloon, or when molecules escape through tiny pores in a membrane, this process is effusion. It occurs because gas molecules, always in motion, occasionally collide with an opening and pass through it. Since only a very small hole is involved, gas molecules must reach it individually; they cannot push through in large groups. Once a molecule passes through the opening, if the other side is nearly empty or under lower pressure, the molecule moves freely into the new space. Over time, many such individual escapes result in significant loss of gas. This explains why even well-sealed balloons eventually lose air or helium: the gas slowly effuses through microscopic pores in the rubber material. The same principle explains the slow deflation of tires, the escape of gases from containers, and the removal of vacuum in systems where seals are imperfect.
Effusion also helps illustrate deeper principles about particle motion. Because gas molecules in motion do not follow a predetermined path, only those that randomly come into contact with the tiny opening will pass through. This “chance collision” nature reveals how microscopic randomness governs macroscopic behavior. Effusion offers a window into the idea that lighter gas molecules generally move faster than heavier ones because their smaller mass gives them higher average speeds. As a result, lighter gases tend to effuse more quickly. This is why helium balloons deflate more rapidly than air-filled ones: helium molecules, being smaller and lighter, escape through material pores more easily. Although this observation has a strong scientific basis, it comes directly from understanding molecular motion rather than from relying on mathematical formulas.
In industrial and scientific applications, both diffusion and effusion play important roles. Gas separation techniques often exploit effusion by allowing lighter gases to pass through porous barriers while heavier ones remain behind. This principle contributed historically to uranium enrichment processes, where gaseous uranium compounds passed through membranes to gradually increase the proportion of lighter isotopes. Similarly, diffusion is used in chemical reactors, environmental monitoring systems, semiconductor fabrication, and controlled-atmosphere packaging. In these contexts, controlling how quickly gases mix or escape allows engineers and scientists to design processes that optimize reaction rates, preserve materials, or maintain safety.
In environmental science, diffusion governs how pollutants spread in air and water. Once a gas is released into the environment, its molecules distribute outward through diffusion, influencing air quality and ecological impacts. Volatile organic compounds, industrial emissions, refrigerant leaks, and natural gases all spread through diffusion. Understanding how diffusion works helps environmental scientists model patterns of pollution, predict concentrations in urban areas, and design ventilation systems that ensure safe indoor air. Effusion also contributes to environmental processes, particularly in soil and rock formations where gases escape from underground pockets or migrate through porous materials. Natural gas seepage, volcanic emissions, and methane release from wetlands all involve effusion-like movement through narrow passages.
Human technology likewise relies on diffusion in unexpected ways. For instance, the ink inside a marker diffuses through porous materials to produce writing. Scents from cleaning products diffuse through fabrics and air. Even cooking relies on diffusion when flavors spread through food or when gases escape from dough during baking. In the human-built world, diffusion helps metals absorb carbon during hardening processes, allows semiconductor components to be doped with specific elements, and enables controlled-release medications to disperse active ingredients within the body over time.
The conceptual difference between diffusion and effusion boils down to the environment in which the gas moves. Diffusion involves movement throughout a space that may already contain other gases, allowing molecules to spread and mix. Effusion involves movement through a narrow opening into a region of lower pressure or near vacuum. Both processes rely on the same underlying feature—constant molecular motion—but manifest differently because of the physical context. This shared foundation illustrates a central idea in the kinetic theory of gases: gas behavior emerges from countless random motions that produce predictable overall patterns.
Perhaps the most profound conceptual takeaway from studying diffusion and effusion is the realization that large-scale patterns—such as the mixing of gases, the spread of scents, or the gradual deflation of a container—arise from microscopic randomness. Every gas molecule moves unpredictably, yet together they create orderly, measurable behavior. This idea reflects a universal scientific theme: complex outcomes emerge from simple, repeated actions. Diffusion and effusion demonstrate how natural processes continuously seek balance, spreading particles until uniformity is reached or until equilibrium is achieved between two spaces. These principles guide both natural systems and engineered technologies, revealing how deeply the motion of particles influences the physical world.
Ultimately, diffusion and effusion provide essential frameworks for understanding gas behavior in science, biology, industry, and everyday life. They show how gases mix, how they escape, how they respond to concentration differences, and how random molecular movement leads to predictable and meaningful outcomes. Through these concepts, one can appreciate the elegance of gas behavior and the remarkable ways in which microscopic motion shapes everything from human respiration to atmospheric dynamics, industrial design, and the smallest details of daily experience.