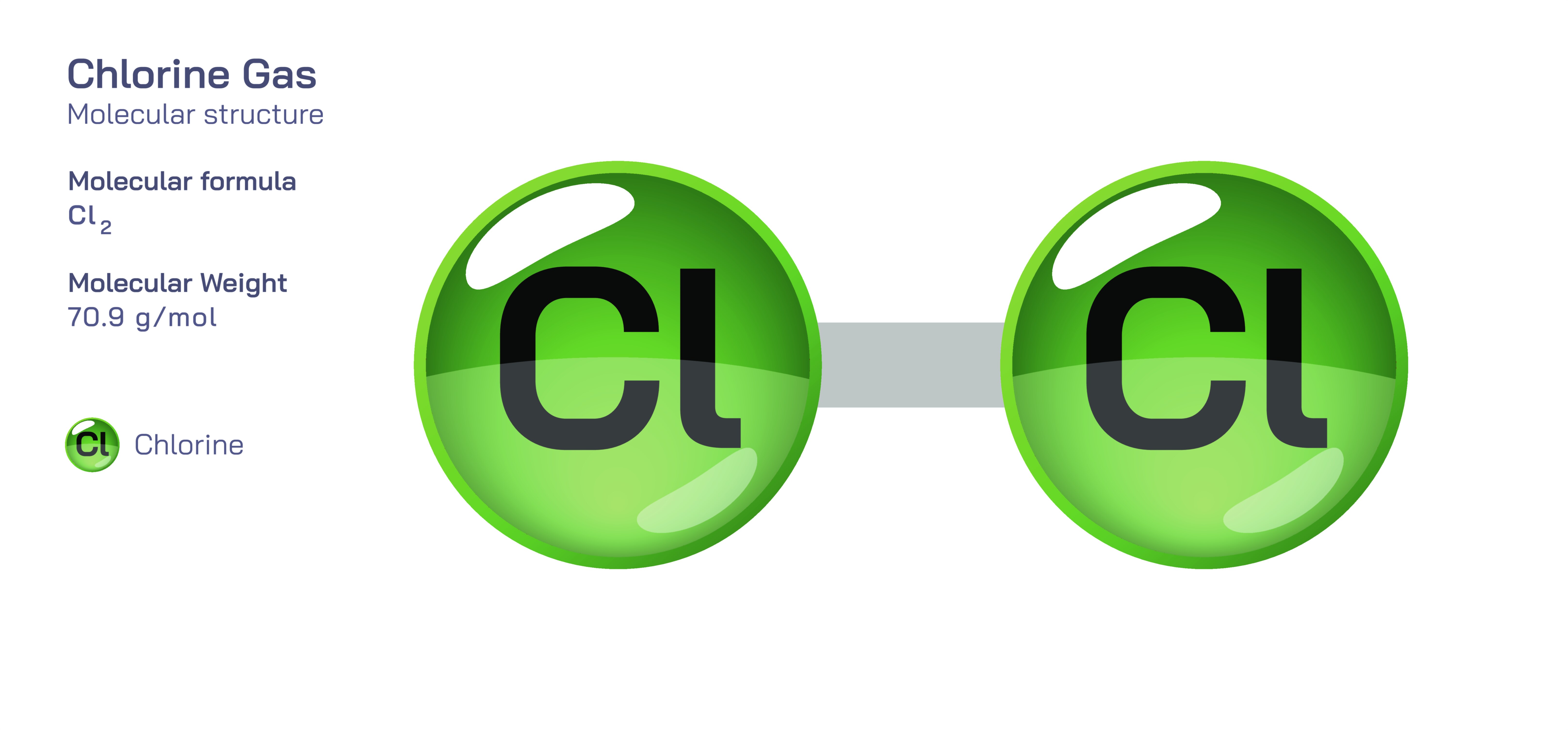
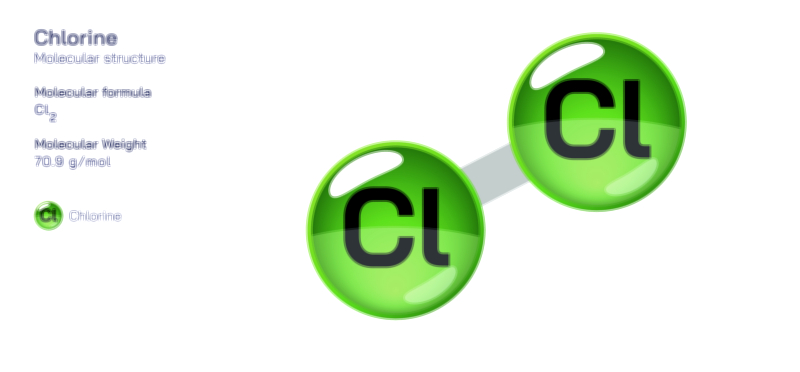
Chlorine Gas — Chemical Element and Molecular Structure
Chlorine gas is one of the most distinctive and influential elemental substances in chemistry, known for its sharp, penetrating odor, its greenish-yellow appearance, and its profound reactivity that shapes environmental processes, industrial production, biological systems, and public health. Although chlorine exists as a simple diatomic molecule composed of two identical chlorine atoms, this molecule’s chemical behavior is anything but simple. It stands at the intersection of natural geochemical cycles and human technological advancement, serving simultaneously as an essential purifying agent, a powerful industrial reagent, a naturally occurring environmental component, and a historically significant chemical with both beneficial and hazardous properties. To understand chlorine gas in its full depth, one must explore its molecular structure, its reactive tendencies, its environmental interactions, its industrial relevance, and its biological impacts—all of which reveal how one elemental substance wields immense influence across multiple domains of human life and the planet at large.
At the molecular level, chlorine gas exists as Cl₂, a diatomic molecule in which two chlorine atoms share a single covalent bond. Because chlorine is a halogen—a member of the highly reactive Group 17 elements—each atom has seven electrons in its outer shell and needs one more to achieve a stable configuration. By forming a bond with another chlorine atom, both achieve stability, producing a molecule with symmetric charge distribution. This symmetry makes chlorine gas nonpolar despite the high electronegativity of its atoms, and it contributes to the molecule’s moderate solubility in water. When dissolved, chlorine undergoes disproportionation reactions that produce hypochlorous acid and chloride ions, a transformation central to its disinfecting power. Chlorine’s strong oxidizing ability, derived from its tendency to gain electrons and form chloride, underpins nearly every application of this gas—from water purification to bleaching to the synthesis of industrial chemicals.
Chlorine gas has a long natural history intertwined with Earth’s environment. It is produced naturally in trace amounts through reactions involving volcanic emissions, the evaporation of seawater aerosols, and certain biological and geochemical processes. Chloride ions, the reduced form of chlorine, are abundant in oceans and are essential for many life forms. While chlorine gas itself is not naturally abundant, halogens in general play significant roles in atmospheric chemistry. In certain conditions, reactive halogen species—including chlorine radicals—participate in reactions that influence ozone levels in the lower atmosphere. In coastal regions, sea spray and sunlight can lead to the formation of minute amounts of reactive chlorine compounds that contribute to environmental transformations. Although these processes occur at very small scales compared to human industrial use, they highlight chlorine’s inherent reactivity and its natural presence in Earth’s chemical cycles.
The industrial production of chlorine gas revolutionized modern chemistry and technology. Historically, chlorine was first recognized in the 18th century and later identified as an element. By the 19th century, its bleaching properties and disinfecting capabilities had made it indispensable. Today, chlorine is manufactured primarily through the electrolysis of brine, a process that splits sodium chloride into chlorine gas, sodium hydroxide, and hydrogen gas. This electrochemical method lies at the heart of the chlor-alkali industry, one of the most important industrial sectors in the world. The materials produced—chlorine, caustic soda, and hydrogen—serve as raw materials for thousands of products, ranging from plastics to pharmaceuticals, textiles, cleaning agents, paper, adhesives, paints, and countless chemical intermediates. Perhaps the most well-known chlorine-derived product is polyvinyl chloride (PVC), a widely used polymer found in pipes, flooring, wiring insulation, medical tubing, and consumer goods. Without chlorine gas, PVC’s global production and the many industries it supports would not be possible.
Chlorine’s disinfecting properties perhaps form its most visible role in daily life. Chlorine-based compounds keep drinking water clean, prevent the spread of waterborne diseases, and maintain hygiene in swimming pools. When chlorine gas dissolves in water, it forms hypochlorous acid, a potent antimicrobial agent capable of destroying bacteria, viruses, and other harmful organisms by disrupting their cell walls and metabolic processes. The introduction of chlorine disinfection into municipal water systems in the early 20th century dramatically reduced outbreaks of cholera, typhoid, and other deadly diseases. Public health experts widely regard water chlorination as one of the greatest medical interventions in history due to its immense impact on human survival and sanitation. Chlorine-based disinfectants remain essential tools in food processing, hospital sterilization, sanitation systems, and emergency response situations where clean water is scarce.
Beyond sanitation and industry, chlorine gas plays a role in organic chemistry and synthetic processes. Its ability to replace hydrogen atoms in hydrocarbons makes it valuable for producing chlorinated intermediates used in pharmaceuticals, agrochemicals, solvents, and polymers. These compounds often serve as stepping-stones in the creation of more complex molecules. For example, chlorine chemistry contributes to the synthesis of anesthetics, antiseptics, herbicides, dyes, and flame retardants. In metallurgy, chlorine is used to refine metals and extract essential elements from ores. Chlorine gas also plays a role in producing high-purity silicon for electronics, as well as in forming specialized materials used in aerospace and advanced manufacturing.
In the environment, chlorine’s reactivity means that free chlorine gas rarely persists for long. It tends to react quickly with organic matter or reduce to chloride ions, which are stable and relatively harmless in small quantities. However, industrial chlorine use requires careful handling, as accidental releases can pose risks to human and ecological health. Chlorine gas is toxic when inhaled, and exposure to high concentrations can irritate the respiratory system, eyes, and skin. Despite this, its risks are managed through strict industrial safety protocols that include containment systems, ventilation, detectors, training, and protective equipment. The combination of high reactivity and potential toxicity makes chlorine a substance requiring respect and responsible use.
Chlorine gas has a complex legacy, not only as a useful industrial and public health tool but also as a chemical once misused in warfare. During World War I, chlorine was deployed on the battlefield as one of the first large-scale chemical weapons. Its choking effects, borne from its reactivity with moisture in the lungs, represented a tragic misuse of chemical science. Although international agreements now prohibit the use of chlorine as a weapon, historical memory reinforces the importance of regulating chemical substances responsibly. This duality—chlorine as both lifesaving disinfectant and harmful agent—illustrates how context determines whether a substance serves humanity or endangers it.
Natural biological systems also depend on chlorine-derived compounds. Chloride ions are essential to human physiology, contributing to the balance of electrolytes, the maintenance of fluid levels, nerve transmission, stomach acid formation, and overall metabolic health. Every cell in the human body requires chloride ions for proper functioning. This shows that while chlorine gas itself is hazardous, its basic ionic form is vital to life. The digestive system relies on hydrochloric acid, formed using chloride ions, to break down food and protect the body from ingested pathogens. The chloride shift within red blood cells helps transport carbon dioxide from tissues to the lungs. The widespread biological role of chloride demonstrates that elemental chlorine, once transformed into its stable ionic form, becomes indispensable to living organisms.
Chlorine gas also influences material science and conservation. It is used to bleach paper and textiles, to treat wood, and to produce safe medical materials. In food preservation, chlorine compounds help sanitize equipment and surfaces used in packaging, processing, and storage, preventing contamination and spoilage. In environmental chemistry, chlorine participates in reactions that influence atmospheric trace gases, though its direct presence in the environment is usually brief and highly reactive. Researchers study chlorine chemistry to understand pollution, ozone formation, and atmospheric oxidative cycles.
Culturally, chlorine’s presence has shaped modern expectations about sanitation, cleanliness, and public health. The comforting scent of a chlorinated pool, the assurance that tap water is safe to drink, and the availability of clean facilities all trace back to chlorine’s disinfecting power. Industrial societies rely on chlorine-based chemistry to support manufacturing, healthcare, shipping, agriculture, and countless other fields. Even households depend on chlorine-derived cleaning products and disinfectants to maintain hygiene.
The story of chlorine gas demonstrates how a single molecular structure can produce cascading effects across science, technology, medicine, and society. Its reactive nature allows it to purify water, synthesize materials, eliminate pathogens, and drive industrial production. Its hazards demand respect, safety, and regulation. Its ionic form sustains biological life. And its historical and modern uses reflect humanity’s evolving understanding of chemistry. Chlorine gas, with its greenish glow and sharp scent, remains a powerful example of how the elemental world shapes human civilization, enabling progress while requiring careful stewardship.