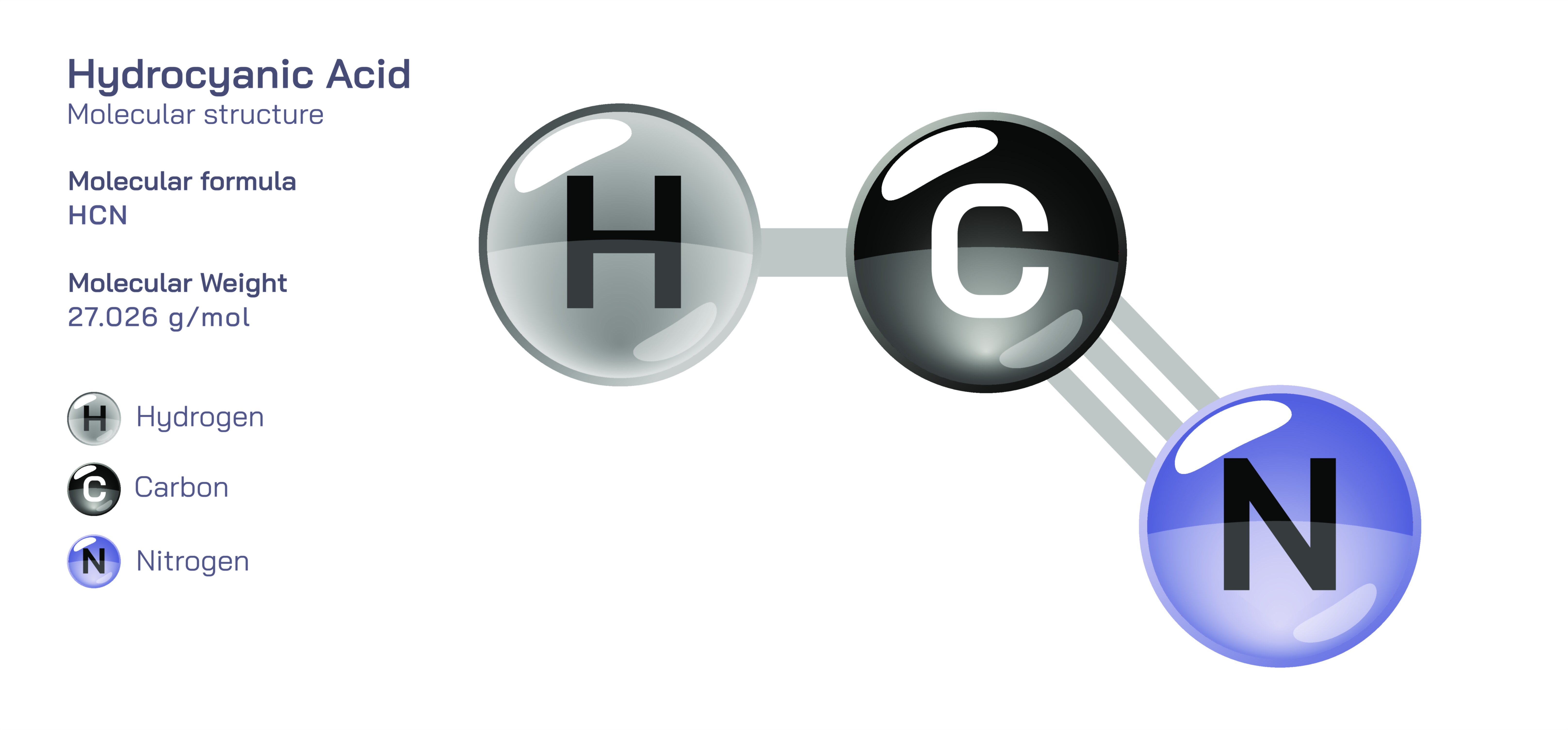
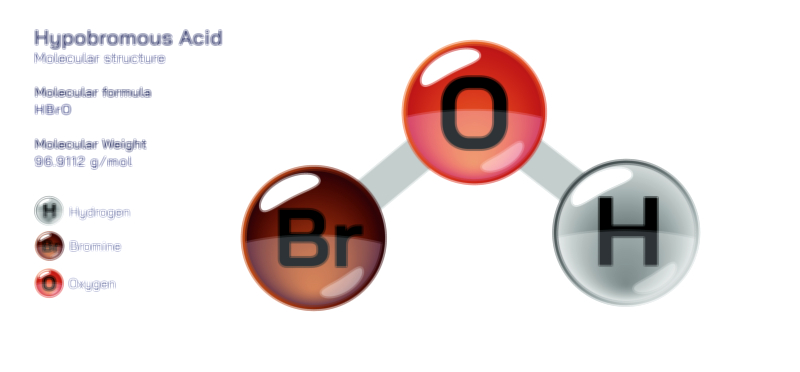
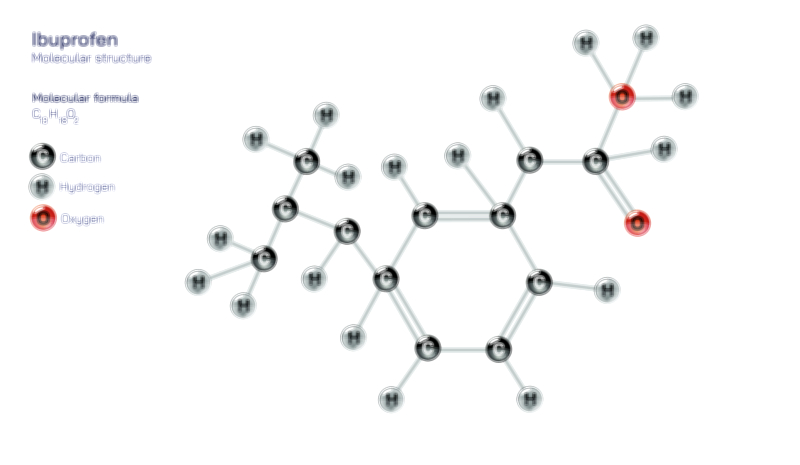
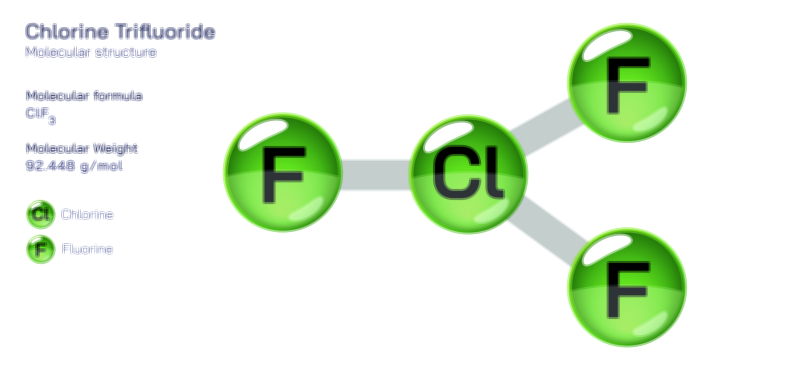
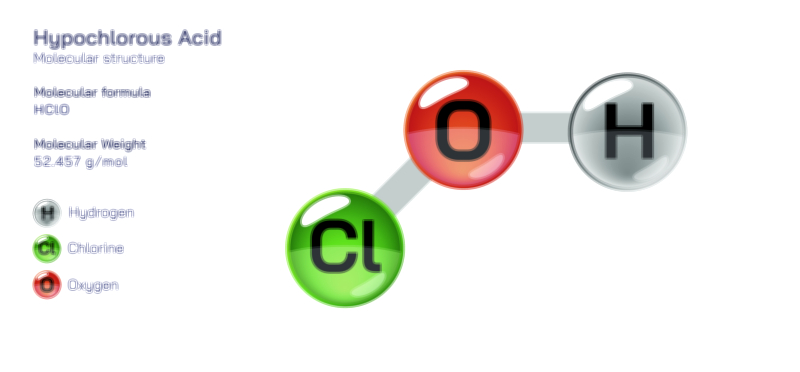
Hydrocyanic Acid — Chemical Structure, Toxic Compound Safety, Industrial Uses, and Scientific Relevance
Hydrocyanic acid, also known as hydrogen cyanide in its gaseous form, occupies a unique and unsettling position in chemistry because it combines an extremely simple molecular composition with a lethal level of toxicity that demands caution, respect, and strict controlled handling. This compound is formed from atoms of hydrogen, carbon, and nitrogen, and its structure gives rise to powerful chemical reactivity as well as the biological danger for which it is widely known. Despite its simplicity, hydrocyanic acid has had a major impact on industrial chemistry, metallurgical extraction, agricultural formulations, and the history of toxicology. It exists as a colorless liquid at lower temperatures and as a highly volatile gas at room temperature, giving it the ability to spread rapidly in air and affect exposed biological systems quickly. The compound is faintly acidic in solution, but its most defining characteristic is not its acidity; rather, it is the intense binding affinity of cyanide ions toward biological molecules that are essential for cellular respiration. This affinity results in rapid disruption of critical biochemical processes if inhaled, ingested, or absorbed through bodily tissues, and it explains why hydrocyanic acid is treated with maximum precaution in laboratories, industrial facilities, and emergency response scenarios. When the subject of hydrocyanic acid appears in educational or scientific illustrations, those vector diagrams normally emphasize not only its simple structure but also the surrounding context of hazard markings, controlled laboratory storage, and safety guidelines that ensure it is never handled casually.
The toxicity of hydrocyanic acid arises from the way cyanide ions interfere with oxygen utilization in living organisms. Oxygen may be present in the blood, but cellular machinery becomes unable to use it when cyanide binds to the component of cells responsible for energy production. This blockage prevents energy generation, leading to rapid systemic failure. Because this happens at the biochemical level rather than through external damage or corrosive action, the victim may not initially feel pain even though the body is entering a state of crisis. That characteristic is what makes the compound particularly dangerous: high volatility enables inhalation exposure before a person recognizes any hazard, and the onset of symptoms may be extremely swift. Training for personnel who work with hydrocyanic acid therefore emphasizes not only safe handling but also real-time monitoring of air quality using chemical sensors and alarms rather than relying on human senses. In the presence of moisture, the gas dissolves readily and can disperse into liquid handling systems or open environments, which is another reason why pressure-sealed storage and closed reaction systems are mandatory in industrial applications. The compound must be kept away from incompatible substances that could initiate unintended reactions or release hydrogen cyanide suddenly into the air.
Despite the risks, hydrocyanic acid has a highly significant role in industrial processes that cannot be fulfilled easily by alternative compounds. One of its most important uses is in metal extraction and mineral processing, especially in the recovery of precious metals such as gold and silver. Cyanide-based solutions can dissolve metallic forms of these elements and allow them to be separated and purified efficiently from ore. Due to the economic importance of these metals, cyanide processes have been used for more than a century, although modern operations are subject to strict environmental controls to prevent leakage into ecosystems. In electroplating industries, hydrocyanic acid contributes to creating surface coatings on metals because cyanide salts formed from the acid promote stable metal complexes required for smooth and uniform plating. In the chemical manufacturing world, hydrocyanic acid is a starting point for synthesizing a wide range of organic intermediates used to produce plastics, synthetic fibers, pharmaceuticals, agricultural chemicals, and nitrile-based polymers. Many compounds used in material science trace their origins to hydrocyanic acid through multi-step reactions that convert cyanide functionality into diverse structures suited for industrial performance.
Agricultural chemistry has historically included hydrocyanic acid as a building block for certain insecticides and fumigation treatments. Its toxicity toward living organisms creates a means of controlling harmful pests that might threaten harvests or stored products. However, applications of this sort are highly controlled today, and many earlier formulations have been replaced with safer compounds to reduce risk to workers and the environment. In plant biology and natural ecology, interestingly, hydrocyanic acid is not exclusively a synthetic compound; some plants produce cyanogenic glycosides that release small quantities of cyanide when damaged, functioning as a natural defensive mechanism. This fact creates an unexpected scientific relevance: the same compound that industrial chemists handle under strict safety protocols also plays a biochemical role in the evolutionary survival strategies of organisms across the plant kingdom.
From the standpoint of emergency safety, hydrocyanic acid has prompted some of the most well-defined response procedures in occupational health and hazardous materials management. Workers in environments where accidental exposure is conceivable receive specialized training on engineering controls, personal protective equipment, air monitoring, and emergency medical protocols. Laboratories equipped to handle the compound include fume hoods with negative pressure systems, gas detectors, and secured chemical cabinets designed to prevent accidental release. Because hydrogen cyanide gas disperses easily, any facility storing the liquid form must have ventilation and multiple backup containment systems. Personnel learn that in the event of a cyanide leak, evacuation, ventilation shutdown, respirator use, and medical intervention are not optional but immediate requirements. Even the transportation of hydrocyanic acid is governed by international regulations that dictate container design, route monitoring, labeling standards, and accident reporting systems. These precautions are not meant to discourage the legitimate use of the compound, but rather to ensure that industries that depend on it protect both workers and surrounding communities effectively.
Hydrocyanic acid also plays a major role in the teaching of chemical hazards and risk assessment because it represents a compound where multiple safety dimensions intersect. It is volatile, toxic, reactive, and capable of entering the body quickly, meaning that handling protocols must account for the combined risks rather than taking a single protective measure. Educational material about the compound helps students understand how chemistry demands responsibility, not fear. Lessons on hydrocyanic acid often include scientific case studies showing the difference between controlled industrial practices and accidental exposure scenarios. These lessons reinforce that the dangerous nature of a substance does not diminish its usefulness; instead, it amplifies the need for proper stewardship. Vector illustrations that accompany these lessons sometimes depict molecular bonds along with hazard pictograms, ventilation diagrams, and secure laboratory storage arrangements. This visual connection reminds viewers that the small-scale composition of a compound affects large-scale outcomes ranging from product manufacturing to workplace safety.
Industries that are committed to environmental sustainability continue to refine how hydrocyanic acid is used to reduce ecological impact. Waste treatment processes are designed to convert cyanide into less harmful substances before release. Continuous monitoring ensures that wastewater streams do not introduce cyanide into rivers or soil systems. Scientists have developed alternative metal extraction techniques and greener pathways in polymer synthesis, aiming to reduce dependence on cyanide-based chemistry without sacrificing efficiency. Yet in many cases, hydrocyanic acid remains the most effective option available, so innovation focuses on safer and more localized handling instead of elimination. Regulations and technological improvements have transformed a compound once linked almost exclusively to hazard into one safely integrated into modern industrial cycles with managed environmental outcomes.
When considering hydrocyanic acid from a broader scientific perspective, it becomes clear that its story is not defined solely by its toxicity but by its ability to alter raw materials, fuel manufacturing systems, and shape entire technological sectors while simultaneously demanding a high level of responsibility from those who work with it. The compound serves as a dramatic example of how chemistry combines power and risk: the same structural features that make it a valuable industrial building block are also responsible for its biological danger. The ultimate lesson for science students, industrial professionals, and policymakers is the importance of knowledge, regulation, and respect rather than fear. Hydrocyanic acid is not simply a compound to avoid, but one to understand deeply so that the benefits it provides in medicine, material science, manufacturing, and mining can be utilized under conditions that protect people and the environment.
In this way, hydrocyanic acid symbolizes an ongoing principle of modern chemistry: powerful substances can advance society, but only when paired with disciplined safety practices, technological vigilance, and continuous scientific scrutiny. Its legacy is built not only on its extreme toxicity, but also on the sophistication of the systems humans have designed to use it constructively without allowing danger to surpass control.