Barium Hydroxide – Chemical Structure, Properties, and Industrial Significance
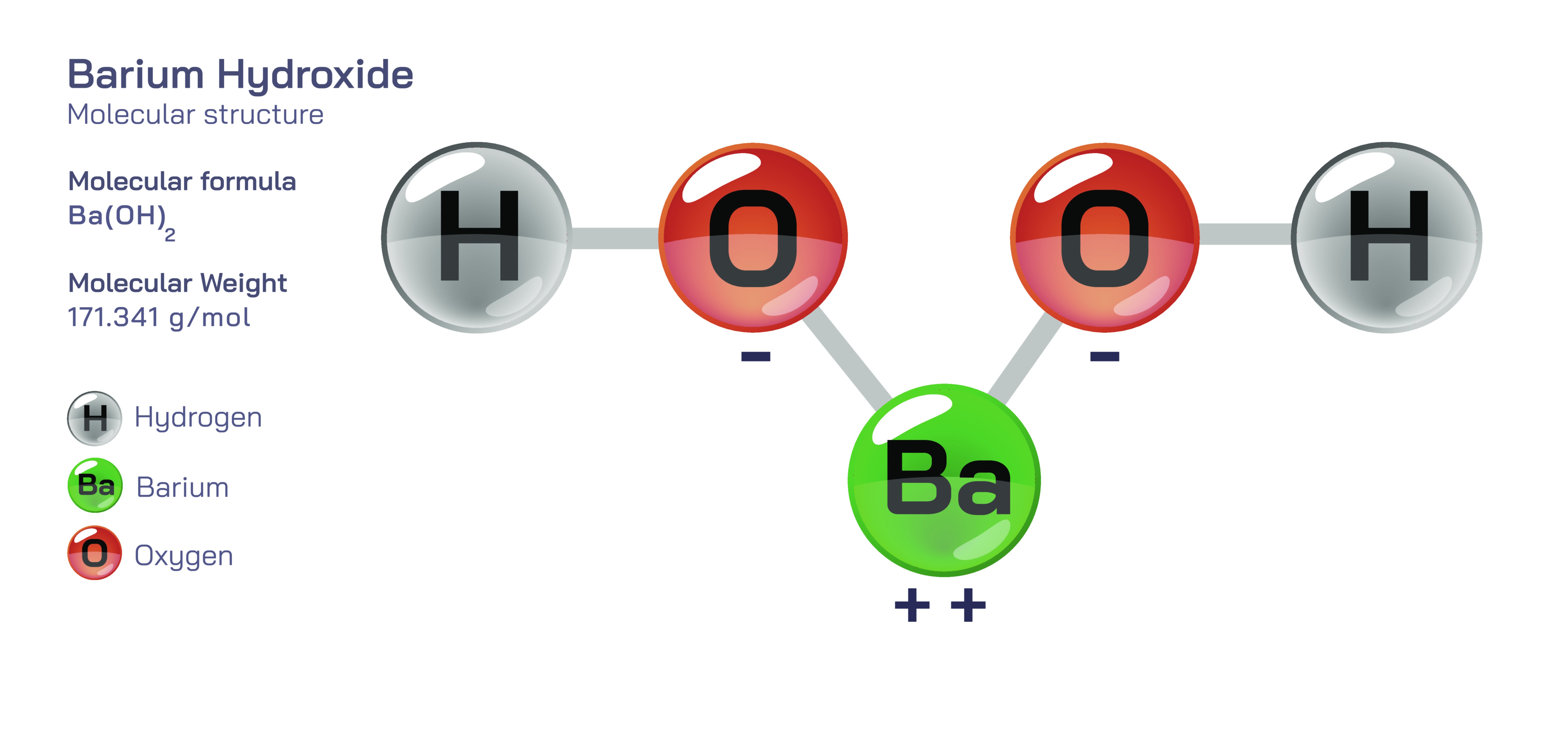
Barium hydroxide, widely represented by the formula Ba(OH)₂, is an essential inorganic compound whose behavior, reactivity, and practical importance stretch far beyond its simple appearance as a metal hydroxide. When visualized as a chemical structure vector illustration, the compound is most commonly depicted with a central barium ion bonded symmetrically to two hydroxide groups. In such molecular graphics, the heavy alkaline earth metal atom occupies the center, and the hydroxide ions extend outward, emphasizing the ionic character of the compound and the distinctive geometry caused by the strong electrostatic attraction between Ba²⁺ and OH⁻. These vector representations are not merely decorative depictions; they serve as precise scientific languages that communicate composition, bonding arrangement, atomic positions, and chemical identity with scalable clarity suitable for both educational and industrial documentation. Every line, spatial relationship, and bond angle delivers information that allows researchers and learners to visualize how structure relates to function, and in the case of barium hydroxide, structure plays a defining role in the material’s behavior, reactivity, and applications.
Barium hydroxide exists in several forms, but one of the most widely encountered variants is barium hydroxide octahydrate, a crystalline solid that incorporates water molecules into its structure. This hydrated form appears as white, highly crystalline flakes that dissolve readily in water, producing strongly alkaline solutions. When water evaporates or when specific thermal conditions are applied, the compound can convert into the anhydrous form, a more reactive and thermally resilient material. Regardless of hydration state, the compound maintains the same structural core: a large barium ion stabilized by electrostatic interactions with hydroxide groups. The ionic nature of this structure explains why the compound is so soluble in water and why its solutions exhibit strong basicity. Every OH⁻ ion present dissociates easily, increasing the concentration of hydroxide in solution and giving rise to pronounced alkalinity. This reactivity is central to both the compound’s value and the need for careful handling wherever it is used.
Chemically, barium hydroxide behaves as a strong base, and this property makes it indispensable in laboratories and industrial systems that rely on alkaline agents. In titration and analytical chemistry, barium hydroxide solutions serve as reliable alternatives to sodium or potassium hydroxides when carbonate-free conditions are required. Carbonate contamination can disrupt measurements and alter reaction outcomes, so the lower tendency of barium hydroxide to absorb atmospheric carbon dioxide makes it uniquely valuable. Its predictable dissociation in water also makes it useful for preparing solutions of known alkalinity for quantitative analytical work. With each ion dissociation event, the compound demonstrates its structural purpose: the binding arrangement shown in chemical vector illustrations translates directly into real-world chemical function.
Beyond laboratory chemistry, the compound plays a pivotal role in industrial manufacturing environments. Barium hydroxide participates significantly in the production of lubricating additives, where its reactivity allows it to form barium-based lubricants used to resist thermal breakdown and oxidation in engines and machinery subjected to harsh workloads. In polymer and plastic industries, it contributes to stabilizing formulations that would otherwise degrade under heat, pressure, or aging. Its alkaline properties also make it useful in the purification of organic compounds, where it can remove unwanted acidic components during synthesis. In the textile and soap industries, it functions as a caustic agent to regulate pH during complex manufacturing steps. Across all of these applications, it is the inherent ionic structure of barium hydroxide — the same one conveyed by vector graphics — that grants the compound its industrial power, because ionic hydroxide groups provide the reactive foundation upon which the compound’s diverse chemical behaviors are built.
Barium hydroxide also holds practical significance in gas purification and environmental control. When used in scrubbing and filtration systems, the compound reacts with sulfur dioxide and other acidic gases to neutralize them. This makes it an important tool in controlling emissions and preventing industrial pollutants from entering the atmosphere. The compound performs this role with both efficiency and predictability because the hydroxide ions are ready to neutralize acidic species, forming less harmful by-products. Once again, the vector illustration of the molecular structure reflects this behavior symbolically: the ionic bond environment makes OH⁻ readily available for reaction, and this availability manifests in environmental chemistry as a mechanism of industrial emissions control.
The structural identity of barium hydroxide also influences its thermal behavior and phase transitions, which matter greatly in extended high-temperature processes. When heated strongly, the compound decomposes into barium oxide and water vapor, shifting from hydroxide chemistry to oxide chemistry in a transformation that depends on the ionic structure of the parent compound. This transformation is reversible under correct conditions, allowing the material to take part in complex chemical cycles that alternate between hydroxide and oxide forms. Understanding this change requires an appreciation of the atomic framework at the heart of the compound, which is precisely what structural illustrations communicate: the composition of the solid does not exist in isolation — it dictates thermal reactivity, phase behavior, and chemical tendencies.
Despite its usefulness, barium hydroxide must be handled with strict attention to safety. The solubility of the compound means barium ions can enter biological systems if the substance is ingested or improperly handled, and soluble barium salts are toxic to humans and animals. Proper laboratory and industrial procedures rely on gloves, protective eyewear, ventilation, and controlled disposal. Contact with skin or eyes can cause chemical burns due to both causticity and ionic activity, so trained handling is essential. However, when kept in controlled chemical environments — whether in educational laboratories, manufacturing plants, or research facilities — barium hydroxide remains an indispensable compound whose benefits are realized safely through adherence to professional standards. Safety considerations do not diminish its importance; they demonstrate that even powerful materials can be managed responsibly when structural understanding and informed caution converge.
The vector illustration of barium hydroxide plays a subtle but meaningful role in enabling that structural understanding. Unlike static printed graphics that blur when enlarged, a vector illustration scales infinitely, meaning every hydroxide connection, every spatial orientation, and every atomic representation remains just as precise regardless of whether the image appears on a tiny datasheet or an oversized educational display. This clarity matters because chemistry depends heavily on visualizing relationships between atoms. The ionic attraction that binds Ba²⁺ to OH⁻ governs the compound’s solubility, basicity, thermal reactivity, and industrial functionality. When displayed visually in scalable form, the chemistry becomes intuitive even to someone encountering it for the first time. In teaching environments, this ensures that the structural reality of the compound is not lost in the simplicity of symbolic formulas. In professional settings, this means digital documents, safety manuals, technical catalogues, and material specification sheets can depict the compound without distortion or ambiguity.
Viewed more broadly, barium hydroxide embodies a powerful lesson about the intersection of atomic structure and applied chemistry. At the microscopic level, its identity reduces to a barium ion surrounded and stabilized by hydroxide groups. Yet from this basic ionic architecture emerges a compound capable of altering industrial manufacturing pathways, enabling precision analytical chemistry, purifying gases, stabilizing polymers, participating in lubrication systems, and transforming under heat into an important industrial oxide. Every property it displays — solubility, alkalinity, reactivity, phase change behavior — is rooted in the invisible chemical structure that vector diagrams make visible. The illustration is not merely a symbol; it is a representation of the foundation that determines how the compound behaves in laboratories, factories, and environmental systems.
In the end, Barium Hydroxide stands as an example of how a seemingly modest inorganic chemical can influence multiple scientific and industrial sectors through the power of fundamental ionic interactions. And in the context of a chemical structure vector illustration, the compound becomes not just a scientific entry but an educational bridge: a visual reminder that behind every reaction, every solution preparation, every industrial formulation, and every environmental process lies a lattice of atoms arranged with purpose. By turning microscopic structure into visible form, the vector representation deepens understanding, strengthens safety, enriches instruction, and supports the wide range of practical applications that make barium hydroxide an enduring and significant compound in modern chemistry.