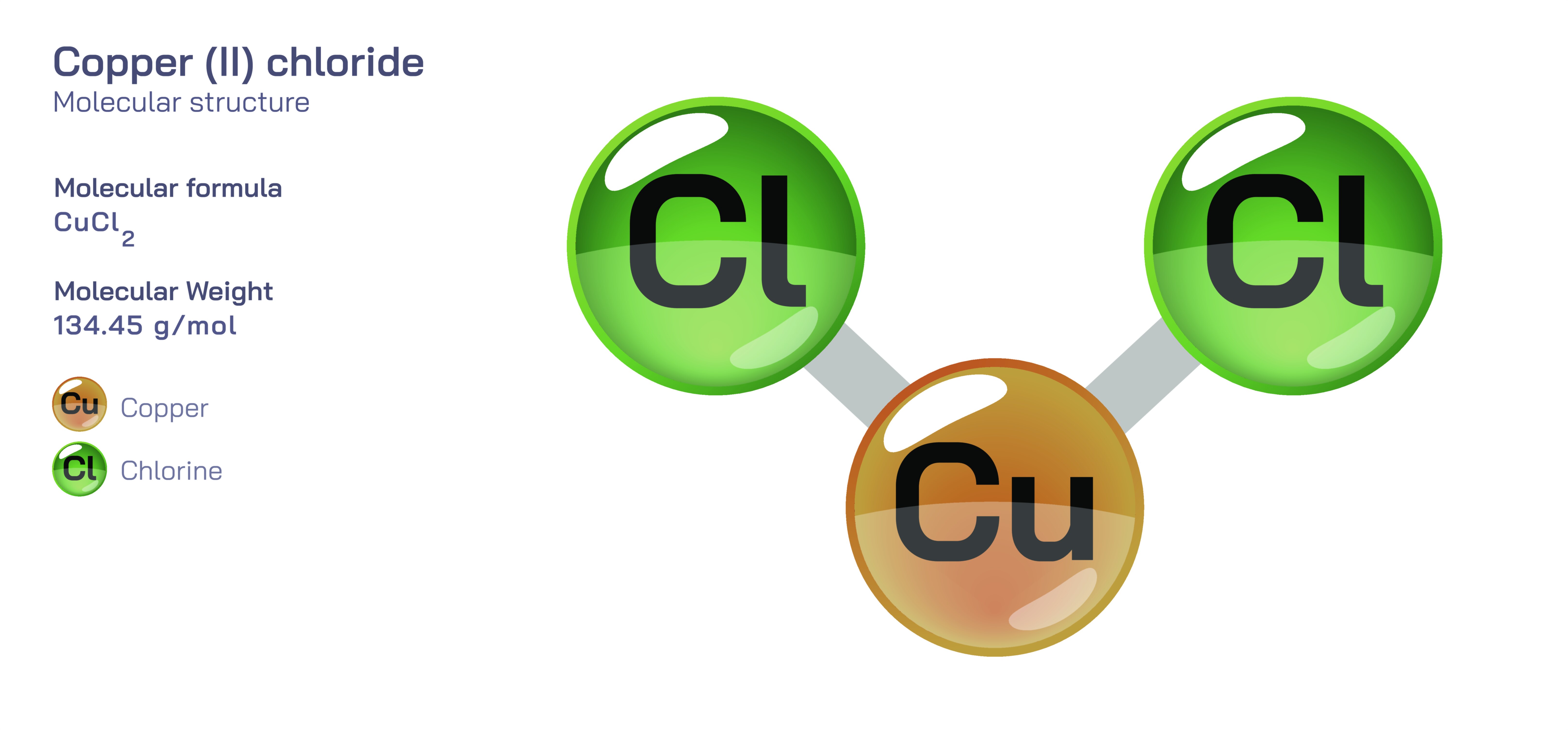
Copper (II) Chloride — Inorganic Chemical Compound
Copper (II) chloride is an important inorganic compound that stands out not only because of its vivid color but also because of its versatile chemical behavior, structural properties, and broad applications across industry, laboratories, environmental science, and materials chemistry. Recognized for its striking blue-green appearance in hydrated form and its dark brown crystalline appearance when anhydrous, this compound demonstrates how a single substance can express different physical identities depending on its molecular surroundings. Copper (II) chloride represents a clear example of how transition metals engage with ligands, environments, and reactive conditions to produce dynamic chemistry. Its behavior reveals the principles of coordination, oxidation, hydration, and complex formation, making it a valuable material for teaching as well as applied science. Although simple in formula, copper (II) chloride embodies the intricate interplay between structure and function that characterizes the chemistry of transition metal salts.
The compound exists primarily in two forms: the anhydrous variant and the more commonly encountered dihydrate. The anhydrous form appears as a brown or yellowish solid with a strong affinity for moisture, rapidly absorbing water from the atmosphere to form the dihydrate. This hydrated form is visually striking, typically presenting an emerald-to-turquoise coloration that reflects the coordination of water molecules around the copper ions. These distinct forms illustrate the strong connection between hydration and electronic structure in transition metal compounds. When water molecules coordinate with copper ions, they alter the energies of the d-orbitals, leading to different ways light is absorbed and reflected, essentially changing the compound’s color. This property demonstrates how coordination chemistry directly influences optical characteristics, a principle found throughout inorganic materials used in pigments, sensors, and catalysts.
Copper (II) chloride is composed of copper ions in the +2 oxidation state paired with chloride ions, forming an ionic compound whose structure is shaped by electrostatic interactions and coordination preferences. Transition metals like copper often display multiple geometries depending on the ligands surrounding them, and copper (II) chloride is a classic study of how ligand environment affects structure. In water, the compound dissociates into copper and chloride ions, enabling it to participate in further reactions that produce complex ions such as tetrachlorocuprate species when chloride concentration is high. These complexes often show deep blue or green coloration, further illustrating how coordination sphere changes influence chemical and physical identity. The ability to form such complexes makes copper (II) chloride a key reagent for investigating ligand exchange reactions, equilibrium processes, and the principles of coordination chemistry in laboratory settings.
One of the most fascinating aspects of copper (II) chloride is its dynamic behavior when heated or exposed to reactive conditions. When heated strongly, the compound can decompose or participate in redox reactions depending on the environment. In reducing conditions, copper (II) ions can be converted to metallic copper, a process exploited in certain synthesis pathways and demonstrations that reveal redox principles. In oxidizing conditions, copper (II) chloride may interact with oxygen-rich environments, displaying transformations that illustrate how copper moves between oxidation states. This redox versatility lies at the heart of much of copper’s chemical importance, with implications for catalysis, corrosion, and metallurgical processing. Copper salts including copper (II) chloride play significant roles in electrochemical systems as well, where copper ions participate in electron transfer pathways critical to battery behavior, plating processes, and corrosion studies.
In manufacturing and industry, copper (II) chloride has long been valued for its catalytic properties. It participates in organic transformations such as chlorination reactions and acts as a catalyst in the manufacturing of vinyl chloride, which is a precursor to polyvinyl chloride (PVC). This industrial use demonstrates the compound’s ability to mediate reactions involving electron transfer and halogen redistribution. Its catalytic capabilities stem from the copper ion’s ability to alternate between oxidation states, allowing it to engage in reaction cycles that regenerate active forms repeatedly. This makes copper (II) chloride not just a simple salt but a functional material capable of accelerating chemical processes that drive large-scale production.
In the realm of environmental science, copper (II) chloride has dual significance. On one hand, it serves as a test reagent and indicator in laboratory analyses. On the other hand, its presence in the environment must be carefully monitored because copper compounds, while essential micronutrients in small amounts, can become harmful to plants and aquatic life when concentrations rise too high. Its solubility and mobility in water enable it to interact with soils, biological tissues, and ecosystems. Understanding how copper (II) chloride dissolves, migrates, and transforms in environmental settings contributes to broader knowledge about heavy metal cycles, contamination pathways, and remediation strategies. Copper’s tendency to bind with organic matter, minerals, and biological molecules means that copper (II) chloride plays a role in complex environmental chemistry where redox conditions influence distribution and impact.
In laboratory settings, copper (II) chloride is widely used as an educational tool. Its vivid color changes, hydration behavior, and ability to form complex ions allow students to observe key chemical concepts directly. Whether in demonstrating Le Chatelier’s principle using chloride-rich solutions that shift equilibrium toward complex formation, or in showcasing redox reactions where copper metal precipitates out of solution, this compound provides clear visual evidence of fundamental chemical processes. Its dehydrated and hydrated forms also help illustrate reversibility and the influence of water coordination on crystalline structure. These teaching applications highlight how simple inorganic compounds can serve as accessible gateways to deeper chemical understanding.
Beyond classroom use, copper (II) chloride plays a role in etching and metal finishing. In printed circuit board production, the compound helps remove metallic copper from surfaces, enabling the creation of intricate conductive patterns. This etching ability derives from copper (II) ions’ readiness to accept electrons from metallic copper, dissolving it and allowing controlled removal. The process demonstrates the compound’s strong redox character and its relevance in the precision manufacturing of electronic components. Even though alternative etchants exist, copper (II) chloride remains an important material in certain fabrication workflows due to its effectiveness and reusability in closed-loop systems.
Biological interactions with copper (II) chloride also illustrate the deep connection between inorganic chemistry and living systems. Copper ions are vital micronutrients necessary for enzymes involved in respiration, antioxidant activity, and cellular energy production. However, when present in excess, copper can promote oxidative stress, leading to cellular damage. Copper (II) chloride in laboratory studies helps researchers explore how cells regulate metal ions, how transport proteins control copper uptake, and how organisms respond to varying metal concentrations in their environments. These studies contribute to advances in biochemistry, toxicology, and medical research, illustrating the compound’s relevance beyond strictly chemical domains.
The compound’s vibrant colors and structural versatility also find use in artistic and material applications. Historically, copper salts contributed to pigments, glazes, and dyes, imparting blue-green tones valued in ceramics and artwork. Though modern pigments rely on more stable compounds, copper (II) chloride remains a point of reference for understanding how metal salts influence coloration in minerals and materials. Its ability to produce colored complexes continues to inform research in advanced materials where transition metals contribute to optical, magnetic, or electronic properties for use in sensors, coatings, and functional composites.
Ultimately, copper (II) chloride embodies the dynamic and adaptable nature of transition metal chemistry. From its hydrated and anhydrous forms to its roles in catalysis, complex formation, metal finishing, environmental behavior, and biological relevance, the compound demonstrates how a single inorganic substance can participate in a wide range of chemical processes shaped by its coordination environment and electron configuration. It serves as both a practical industrial material and a compelling scientific example of how structure, environment, and reactivity intertwine. Through this compound, one sees clearly the interconnectedness of disciplines—chemistry, biology, materials science, and environmental studies—all woven together by the versatile behavior of copper and its chloride partners.