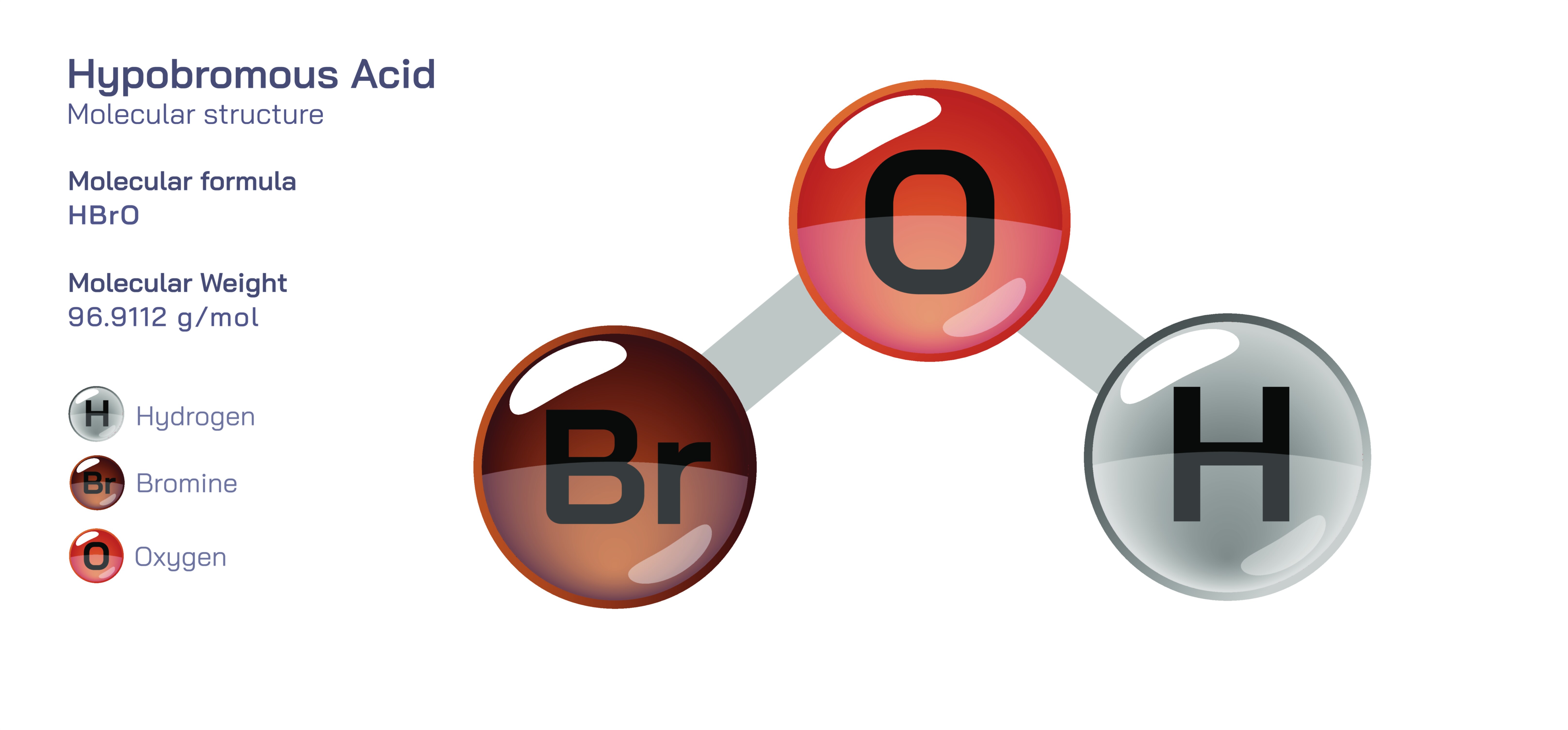
Hypobromous Acid Chemical Structure — HOBr Molecule Diagram and Molecular Behavior Explained
Hypobromous acid, represented by the chemical formula HOBr, is one of the most intriguing molecules in inorganic and biological chemistry because its simplicity conceals a powerful combination of oxidizing capability, disinfectant strength, and biochemical relevance. At its structural core, hypobromous acid is composed of a hydrogen atom bonded to an oxygen atom, which in turn is bonded to a bromine atom, forming the sequence H–O–Br. This arrangement makes HOBr an oxoacid of bromine, belonging to the family of hypohalous acids along with hypochlorous acid (HOCl) and hypoiodous acid (HOI). The molecule is asymmetric and highly polar, with electron density drawn strongly toward the oxygen and bromine atoms while hydrogen carries a partial positive charge. In aqueous solution, HOBr behaves as a weak acid, but the weakness in acidity does not reflect weakness in reactivity; it is an aggressive oxidizing and antimicrobial species due to the unique ability of the bromine atom to accept electrons and participate in redox reactions. The structural relationships within the molecule, the positioning of chemical bonds, and the electron distribution across the atoms all contribute to the extraordinary reactivity of hypobromous acid in biological systems, water treatment chemistry, halogen reactions, and environmental processes.
The molecular structure of HOBr is best understood by considering its electron distribution and bond polarities. Oxygen, being more electronegative than bromine and hydrogen, acts as the central electron-withdrawing atom in the molecule. Its presence between hydrogen and bromine results in two distinctly different bonds: the O–H bond has strong polarity with hydrogen carrying partial positive charge, while the O–Br bond connects oxygen to a larger, more diffuse halogen atom that has a high electron-accepting capacity. The effect of this arrangement is that the proton (H⁺) is weakly held, allowing HOBr to act as an acid, but the bromine center becomes highly reactive toward electron-rich substances, making the molecule an oxidant even in dilute solutions. This dual nature — proton donation and redox ability — explains why HOBr disrupts organic molecules such as cellular membranes, protein side chains, and nucleic acids. Even though the molecule dissociates only partially in water, the equilibrium between HOBr and its conjugate base, the hypobromite ion (OBr⁻), gives the solution a dynamic oxidative power that depends on pH, temperature, and the presence of catalysts. The structural weakness of the H–O bond relative to the O–Br bond also means that HOBr decomposes under certain conditions, yielding bromine-containing species that can participate in further chemical reactions.
One of the most important features of hypobromous acid is its role as a biological disinfectant. In seawater and in many physiological environments, HOBr is formed when bromide ions (Br⁻) are oxidized by chlorine-based disinfectants or biological enzymes. Marine organisms, including some immune-specialized white blood cells, deliberately produce HOBr as part of their antimicrobial defense systems because its oxidizing strength can destroy bacterial cell walls and viral components. Hypobromous acid can neutralize pathogens by halogenating amine groups in proteins, disrupting metabolic enzymes, oxidizing sulfhydryl groups, and damaging membrane lipids. This biochemical action is highly effective but must be tightly controlled by organisms because excess HOBr can also cause oxidative stress to host tissues. The ability of HOBr to react quickly with biological molecules reflects the electron demand of the bromine atom, which tends to form new covalent bonds with nucleophilic centers in living systems. In other words, the same structural feature that fights pathogens can, without regulation, threaten the organism that produces it, highlighting a delicate balance between chemical utility and biological hazard.
In industrial and environmental water chemistry, hypobromous acid plays an essential role because it forms naturally and efficiently when bromide-containing water is disinfected with oxidizing agents. In swimming pools, hot tubs, and seawater sanitation systems, HOBr often becomes the dominant active disinfectant when bromide salts are present. Compared to hypochlorous acid (HOCl), HOBr reacts more rapidly with organic contaminants and nitrogen-containing biomolecules, which is why bromine-based pools retain disinfecting efficiency at higher temperatures. The structural behavior of HOBr explains this heightened reactivity: the larger bromine atom polarizes the O–Br bond, making bromine readily available for electrophilic attack. However, this same mechanism contributes to the formation of brominated organic byproducts when hypobromous acid reacts with natural organic matter, illustrating that the molecule’s environmental effects are not limited to pathogen control but extend to water safety and chemical fate. HOBr can convert into bromamines in the presence of ammonia, undergo disproportionation to form bromate or bromide depending on oxidation conditions, and participate in halogen-exchange reactions that shape the broader halogen cycle in aquatic ecosystems.
The structural and chemical properties of hypobromous acid provide insight into its function in oxidation and reduction pathways. HOBr is strong enough to oxidize many inorganic and organic species but mild enough to operate selectively compared with more aggressive oxidants. The reactivity flows largely from the electrophilic bromine atom, which tends to accept electrons by inserting into chemical bonds or abstracting hydrogen from substrates. This is why HOBr can bleach pigments, degrade biological residues, and break down staining compounds through oxidative halogenation. At the same time, the proton-donating capacity of HOBr allows it to interact with bases, forming hypobromite salts that can participate in reversible oxidation cycles depending on pH. These reversible chemical paths contribute to the versatility of HOBr as both a reactant and an intermediate in halogen reaction networks.
The stability of hypobromous acid is highly dependent on environmental conditions, reflecting the delicately balanced energy of its molecular structure. HOBr exists most readily in mildly acidic to neutral aqueous environments; at lower pH, it shifts toward formation of molecular bromine species, and at higher pH, it converts into the hypobromite ion. Light, heat, and catalytic impurities accelerate decomposition by weakening the O–Br bond, which is less stable than the equivalent O–Cl bond in hypochlorous acid. Thus, the molecule’s chemical power is inherently tied to its structural fragility — the very bond that gives HOBr its oxidizing strength can be broken by the same reactive patterns it promotes. This instability is not a flaw but a reflection of the reactive role HOBr plays in ecological and engineered systems: it does its work and then decays, preventing accumulation but ensuring rapid chemical impact.
Safety considerations surrounding hypobromous acid also stem directly from its structure. Because the molecule aggressively reacts with organic compounds, concentrated HOBr or solutions rich in hypobromite ions can damage skin, eyes, and mucous membranes through oxidative protein modification and lipid peroxidation. Inhalation of aerosols containing HOBr can irritate respiratory tissue for the same reasons. Despite these hazards, the molecule is invaluable when controlled carefully because its reactivity leaves no harmful residue after decomposition, transitioning into bromide and other lower-energy species. The challenge is not whether HOBr is safe or dangerous by nature, but whether humans manage its reactivity in a thoughtful and informed way.
Ultimately, the story of hypobromous acid illustrates how molecular structure determines function in both chemical and biological contexts. The simple sequence of atoms — H linked to O linked to Br — encodes electron distribution, bond strengths, reactivity patterns, and chemical lifetimes that shape real-world behaviors ranging from immune system activity and seawater chemistry to water disinfection and environmental oxidation pathways. HOBr is powerful not because of complexity but because of precision: every property it exhibits emerges from the electronegativity contrast between hydrogen, oxygen, and bromine, and from the resulting polar, asymmetric molecular configuration. Understanding hypobromous acid means recognizing that even the smallest shift of electrons within a molecule can give rise to processes that shape microscopic cell defense, global water chemistry, and technological methods for controlling microbial life. Through this lens, HOBr stands as a profound example of how structure directs chemical destiny and how a seemingly simple molecule can have outsized influence across nature and human innovation.