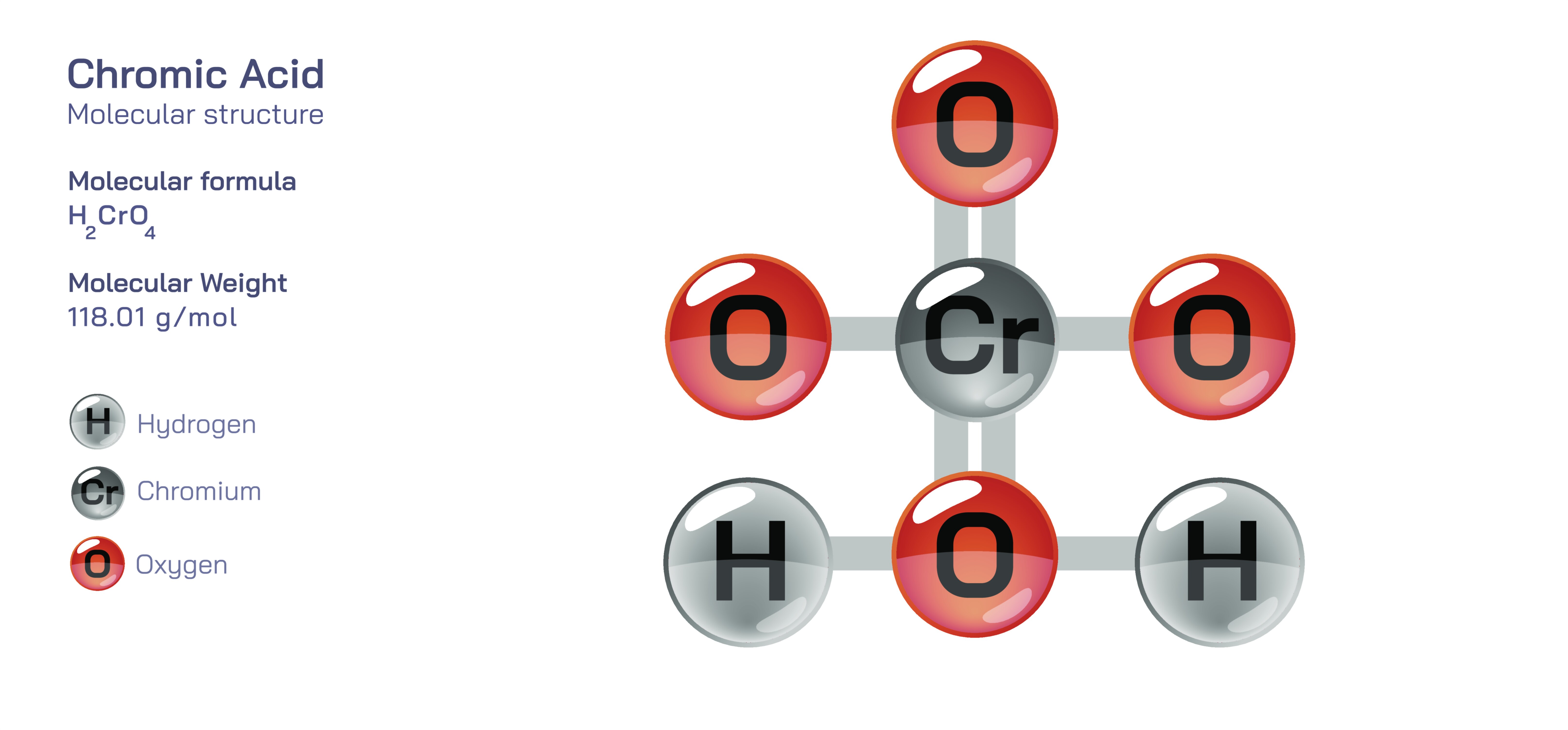
Chromic Acid — Strong Oxidizing Chemical Compound
Chromic acid stands as one of the most powerful, versatile, and historically significant oxidizing chemical compounds in inorganic chemistry. Though the name “chromic acid” is often used loosely in laboratories and industry, the term refers not to a single stable molecule but to a family of related species generated when chromium(VI) compounds such as chromium trioxide dissolve in water. In its idealized form, chromic acid corresponds to H₂CrO₄, but in reality, it quickly equilibrates with other chromium(VI) oxyanions including dichromic and polychromic acids depending on concentration, pH, and surrounding chemical conditions. What remains consistent across all of these forms is the presence of chromium in its hexavalent oxidation state—a state that endows chromic acid with extraordinary oxidizing strength, high reactivity, and a broad capacity to transform organic and inorganic substances. Its vivid colors, shifting from yellow to orange to deep red depending on structural changes, reflect the electronic transitions characteristic of chromium(VI), visually signaling the potent chemistry contained within this compound. Understanding chromic acid requires exploring its structure, reactivity, industrial uses, environmental implications, biological toxicity, and the deep historical footprint it has left across chemical processes used for generations.
Chromic acid’s identity begins with the chromium(VI) ion, a highly electron-deficient center surrounded by oxygen atoms arranged in characteristic tetrahedral geometries. This electron distribution imparts strong oxidizing behavior, as chromium(VI) eagerly accepts electrons to transition into the more stable chromium(III) state. This oxidation–reduction transformation lies at the heart of chromic acid’s function. When chromic acid encounters organic compounds, it can strip electrons from carbon–hydrogen or carbon–oxygen bonds, converting alcohols into aldehydes or ketones, and aldehydes into carboxylic acids. These reactions have played an essential role in classical organic chemistry, particularly before milder or greener oxidants became available. Chromic acid’s aggressive oxidizing nature extends beyond organic molecules; it can attack metals, break down complex materials, and even destroy biological tissues, making it both a powerful tool and a dangerous substance requiring rigorous control. Its color-changing behavior—shifting toward shades of green as chromium(VI) reduces to chromium(III)—gives chemists a visual indicator of reaction progress and has made chromic acid mixtures iconic in traditional laboratory settings.
The reactivity of chromic acid is intimately tied to environmental conditions. In acidic solutions, dichromate species dominate, while in neutral or alkaline environments, chromate ions appear more prominently. This equilibrium affects chromic acid’s oxidizing strength, with acidic conditions enhancing its ability to drive redox reactions. The compound’s behavior in aqueous solution is also relevant to industrial processes, where chromic acid mixtures are often prepared using chromium trioxide and concentrated sulfuric acid. These solutions are capable of dissolving organic residues, removing coatings, and etching materials, and their application requires careful management of concentration and acidity to achieve precise outcomes. Chromic acid’s capacity to transition between chromate, dichromate, and various protonated species reflects its flexibility but also adds complexity to its safe handling and environmental behavior.
Historically, one of chromic acid’s most important applications lay in metal finishing and surface treatment. Chromic acid plays a crucial role in chrome plating, a process in which a layer of chromium metal is deposited onto metal objects to provide corrosion resistance, hardness, and aesthetic appeal. Chromic acid baths, combined with electrical currents, enable the controlled reduction of chromium(VI) to chromium metal on the surface of substrates such as steel or copper alloys. This transformation produces a shiny, durable chromium coating widely used in automotive parts, industrial tools, appliances, hardware, and decorative fixtures. The properties of chromium coatings—resistance to oxidation, wear, and corrosion—stem directly from the unique chemistry of chromium itself, but chromic acid supplies the essential precursor that makes electroplating possible. Although emerging technologies and environmental regulations now promote alternatives such as trivalent chromium plating, chromic acid remains foundational to understanding the evolution of materials engineering.
Chromic acid has also been used extensively in cleaning and etching applications. In laboratories, chromic acid cleaning solutions were once the standard for removing stubborn organic residues from glassware. These solutions, combining chromium trioxide and concentrated sulfuric acid, possess extraordinary oxidizing capability, decomposing organic contaminants and leaving surfaces free of microscopic films that might interfere with precise analytical work. While effective, this practice has been largely abandoned due to safety and environmental concerns, replaced by detergents, peroxides, or modern cleaning agents that reduce the risk associated with chromium(VI) exposure. Nevertheless, the historical use of chromic acid in laboratories serves as a reminder of its unmatched power to degrade organic material and its longstanding importance in laboratory technique.
In industry, chromic acid contributes to numerous surface treatment processes beyond plating. Chromate conversion coatings, for example, rely on chromic acid solutions to form protective layers on aluminum, zinc, and magnesium alloys. These coatings offer corrosion resistance, improve paint adhesion, and provide durability under harsh environmental conditions, making them valuable in aerospace, automotive, military, and marine applications. The longevity and reliability of chromate-based coatings have made them difficult to replace fully, although environmental and health regulations continue to drive the search for chromium-free alternatives. Chromic acid etching also plays a role in preparing materials for bonding or composite fabrication, particularly in advanced manufacturing sectors where adhesion performance is critical.
Chromic acid has also been significant in organic synthesis. Classical oxidation methods such as the Jones oxidation or the use of pyridinium chlorochromate (PCC) and pyridinium dichromate (PDC) evolved from chromic acid’s underlying chemistry. These oxidizing agents allowed chemists to convert alcohols into more oxidized functional groups with high reliability, enabling the synthesis of pharmaceuticals, fragrances, dyes, and specialty organic compounds. Chromic acid’s predictable redox behavior made it a staple reagent in chemical research for decades. Though many laboratories now prefer milder or more environmentally benign oxidants, traditional chromic acid-based methods remain central to the historical understanding of organic reaction mechanisms.
However, chromic acid’s impressive chemical utility is matched by significant health and environmental hazards. Chromium(VI) compounds, including chromic acid, are classified as carcinogenic, mutagenic, and capable of causing severe respiratory, skin, and systemic toxicity. Exposure can lead to tissue damage because chromium(VI) readily penetrates biological membranes and generates reactive intermediates that harm DNA, proteins, and cellular structures. Industrial workers handling chromic acid must employ stringent protective measures—ventilation, gloves, eye protection, and air monitoring systems—to prevent inhalation or skin contact. Regulatory agencies strictly control permissible exposure levels due to the compound’s ability to cause long-term health effects. This recognition has led to widespread safety reforms, the adoption of alternative chemicals, and ongoing research into chromium(VI) waste management.
Environmental concerns further shape the modern use of chromic acid. When released into water or soil, chromium(VI) compounds exhibit high mobility, traveling through ecosystems and posing risks to human and ecological health. Reducing chromic acid to chromium(III), a more stable and less toxic form, is the primary strategy for environmental remediation. This reduction can occur through chemical agents like sulfite or ferrous iron, or through biological pathways involving specialized microorganisms that metabolize chromium(VI). Remediation efforts often involve complex interactions between geochemistry, hydrology, and microbial ecology, reflecting how deeply chromic acid’s behavior intersects with natural systems. Modern waste treatment facilities employ controlled reduction processes and strict containment to prevent chromium(VI) contamination, acknowledging the challenges associated with managing such a potent oxidizing agent.
Despite these risks, chromic acid continues to serve as a reference point for evaluating oxidation strength, corrosion protection strategies, and transition metal chemistry. Its role in educational settings—now largely theoretical rather than practical—helps students grasp redox principles, oxidation states, ligand interactions, and transition-metal behavior. The vivid color changes associated with chromic acid and its reduction products offer intuitive, visual insights into chemical transformations, reinforcing core concepts in inorganic chemistry. Even as safer alternatives become more prevalent, the conceptual importance of chromic acid persists, bridging the historical foundations of chemistry with modern advancements in green chemistry, environmental protection, and material science.
In many ways, chromic acid symbolizes the dual nature of chemistry itself: a compound of remarkable utility and equally remarkable danger. It powered industrial and scientific progress for decades, enabling innovations in manufacturing, analysis, and synthesis. At the same time, its hazards have informed regulatory changes, environmental awareness, and a rethinking of chemical practices. Chromic acid’s story is one of power, transformation, responsibility, and adaptation. It shows how deeply chemical substances can influence technology, how essential safety becomes when dealing with strong oxidizers, and how evolving understanding can reshape even the most established materials and methods.
Ultimately, chromic acid remains a compound defined by intensity—of color, reactivity, and scientific impact. Its role in oxidation chemistry, metal finishing, and industrial processing illustrates the extraordinary capabilities of chromium(VI) compounds, while its environmental and biological risks underscore the need for caution and innovation. Through understanding chromic acid, we gain insight into the profound interplay between chemical structure, reactivity, utility, and responsibility, a relationship that lies at the heart of modern chemical science.