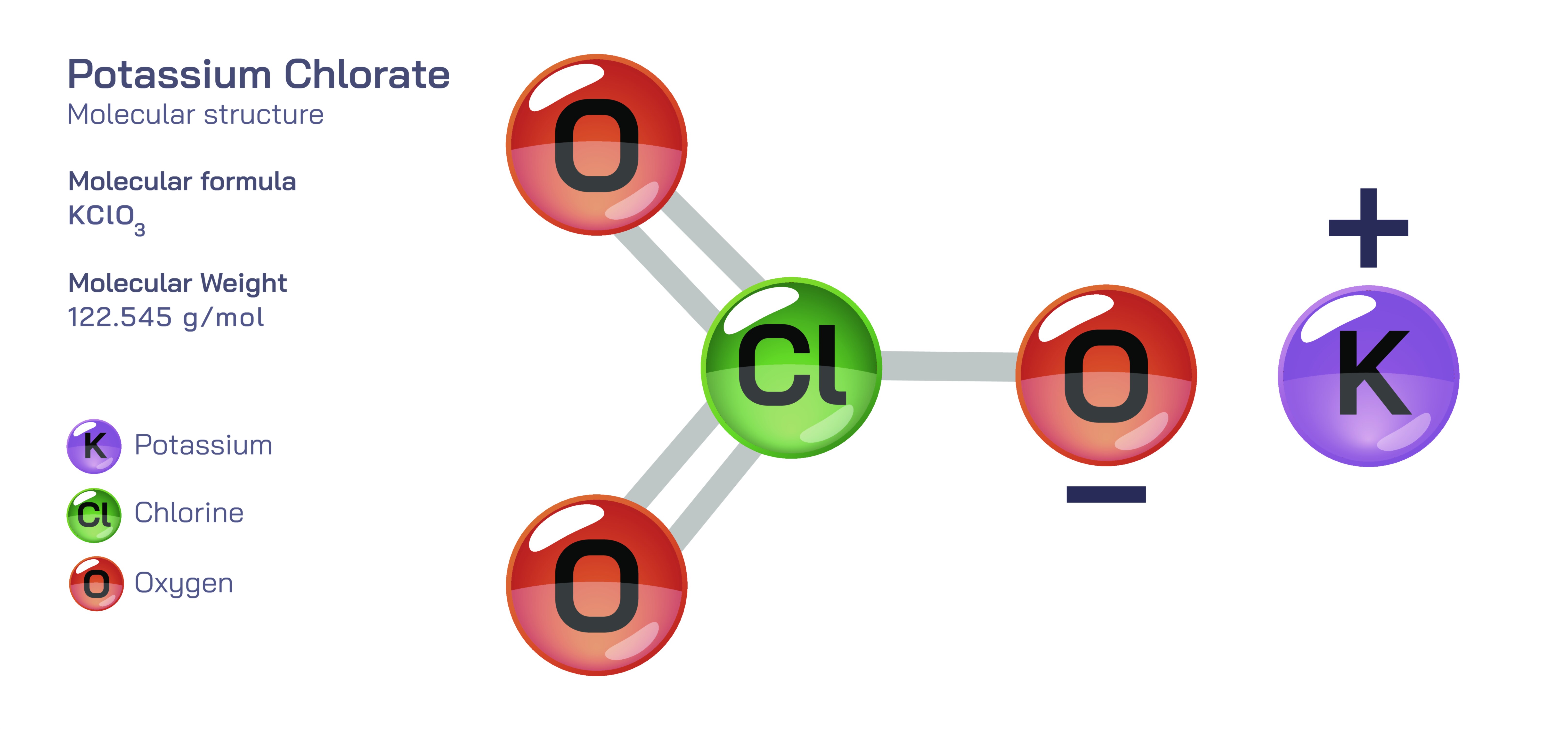
Potassium Chlorate — Chemical Properties, Oxidizing Behavior, Industrial Uses, and Safety Considerations
Potassium chlorate is a highly reactive inorganic compound widely recognized for its strong oxidizing capabilities and its long-standing role in manufacturing, industrial chemistry, pyrotechnics, laboratory experiments, and even early educational demonstrations involving oxygen generation. It appears as a white crystalline solid that dissolves moderately in water and releases oxygen readily when heated or when brought into contact with powerful reducing agents. Its chemical identity is defined by a combination of potassium, chlorine, and oxygen atoms arranged in a stable chlorate ion structure paired with a potassium cation. While the compound itself is stable under controlled storage, its oxidizing nature makes it capable of sustaining combustion, intensifying flames, and triggering rapid reactions when it encounters fuels, organic matter, powdered metals, or other chemicals capable of electron donation. These characteristics place potassium chlorate among the most important oxidants used for ignition, signaling, propulsion, and controlled oxygen release. At the same time, they demand careful handling, strict storage guidelines, and thoughtful risk awareness to ensure that the compound remains a tool of industrial and scientific value rather than a source of accidental ignition or unsafe release of energetic reactions.
The structure of potassium chlorate revolves around the chlorate ion, in which one chlorine atom forms bonds with three oxygen atoms in a trigonal pyramidal configuration. The relatively high electron deficiency of the chlorine center gives the ion a strong affinity for reactions that transfer electrons. When the compound is exposed to heat, strain, or catalytic substances such as manganese dioxide, the chlorate ion decomposes to produce oxygen gas and potassium chloride. This decomposition reaction releases oxygen in quantities sufficient to sustain and accelerate combustion even without direct atmospheric contribution. Unlike ordinary combustion that requires oxygen from the air, reactions fueled by potassium chlorate can support burning in enclosed spaces because oxygen is generated internally from the breaking of chemical bonds. This makes the compound valuable wherever a steady supply of oxygen is needed under controlled circumstances, but it also highlights why its use cannot be taken lightly around heat sources, friction, incompatible materials, or contaminants that might initiate decomposition unpredictably.
Despite these energetic properties, potassium chlorate has been and remains a cornerstone chemical in several industries. One of its most prominent historical uses has been in match production. Safety matches traditionally rely on a controlled ignition surface that transfers energy to a small pellet of potassium chlorate in the match head. When struck, the heat and friction convert the chlorate into oxygen rapidly, igniting sulfur and combustible binders to produce a clean flame. Without potassium chlorate, this instantly reliable ignition mechanism would not function with the same speed and consistency. Modern match technology has diversified, but potassium chlorate continues to be involved in ignition systems due to its predictable reactivity and non-toxic combustion products when used correctly. This same oxygen-releasing ability also makes the compound valuable in pyrotechnics. In fireworks, signal flares, and smoke-producing cartridges, potassium chlorate serves as a central oxidizer that enables the combustion of metallic powders, organic fuels, and color-producing compounds. The compound helps regulate flame temperature, burn time, and energy release, supporting vibrant and sustained visual displays that characterize pyrotechnic shows and military signaling.
In laboratory environments, potassium chlorate plays a foundational role in both research and education. It is frequently used in experiments that demonstrate oxygen generation, allowing students to observe gas evolution from a solid compound in a controlled setting. When heated with a catalyst, it produces oxygen smoothly, making it convenient for lessons on gas collection, stoichiometry, and reaction kinetics. Beyond instructional demonstrations, potassium chlorate is used in analytical chemistry where strong oxidizing conditions are required to break down complex samples or convert chemical species into identifiable forms. It has also found relevance in niche synthetic applications where the oxidizing power of the chlorate ion supports reactions that other milder oxidants cannot drive to completion. The compound’s predictability in the presence of catalysts and thermal activation makes it highly versatile, though modern laboratory practice emphasizes that every use must be coupled with strict precautions to avoid uncontrolled reactions.
The industrial role of potassium chlorate extends into sectors associated with oxygen-dependent processes. Water treatment plants have historically used chlorate-derived chemistry to control microbial growth and break down undesirable organic contaminants, although more efficient sanitation systems have gradually reduced reliance on chlorate compounds. In mining, potassium chlorate has contributed to controlled explosion technologies and separation of ores under highly regulated conditions. In the production of certain dyes, disinfectants, and specialty chemicals, potassium chlorate serves as an intermediate oxidizing agent that initiates or sustains reaction paths that simpler oxidants cannot support. It has also been employed in the manufacture of gas-generating systems for emergency breathing devices and life-saving equipment, where the compound supplies oxygen rapidly in situations where access to breathable air is compromised. These industrial applications highlight the compound’s ability to release large amounts of oxygen reliably and compactly when triggered intentionally.
Safety considerations are central to all uses of potassium chlorate because the same characteristics that make it useful can also create hazards if mishandled. The compound is not explosive on its own, but it becomes dangerous when combined with fuels, dusts, sulfur, strong acids, or finely divided organic materials. Even gentle impacts or friction can trigger vigorous reactions in mixtures containing potassium chlorate and combustible substances. Exposure to heat or contamination by reducing agents can accelerate decomposition unpredictably. For this reason, industries that use the compound store it separately from fuel sources, maintain high purity standards to prevent impurities from altering reaction behavior, and employ environmental controls that avoid static discharge or accidental mixing. Personal protective equipment, antistatic handling tools, sealed storage containers, and continuous monitoring of temperature and humidity are standard in environments where the compound is used at scale. In educational settings as well, handling protocols emphasize limited quantity use, appropriate catalysts, proper disposal of reaction residues, and supervision by experienced personnel.
Even though safer and more stable oxidizing agents now exist for many applications, potassium chlorate continues to hold a prominent position because of its unmatched combination of availability, reactivity, energy density, and oxygen-generating capability. Its enduring presence demonstrates that industrial chemistry evolves not only through the discovery of new compounds but also through responsible management of known substances that possess exceptional utility. Potassium chlorate illustrates the principle that powerful reactions can be harnessed safely when manufacturing systems, laboratory practices, and educational methods are matched with risk-aware design. The compound teaches a lesson broader than chemistry: technology advances most effectively when reactive materials are used with respect for their strengths and their dangers.
Understanding potassium chlorate therefore requires seeing both sides of its identity — an invaluable oxidizer that powers ignition, pyrotechnics, oxygen generation, and chemical synthesis, and a compound that demands deliberate handling to prevent accidental energetic reactions. Its story spans the earliest days of ignition technology through modern industrial science, linking chemistry to manufacturing, engineering, and education. Whether supplying oxygen to a flame in a match head, sustaining combustion in a firework, driving complex reactions in a factory, or providing students a firsthand view of chemical gas evolution, potassium chlorate serves as a vivid reminder of how controlled chemical reactivity shapes the systems and materials that support daily life.