Rising Water Experiment: Understanding Air Pressure and Oxygen Consumption in a Simple Science Activity Explained
The rising water experiment is one of the most elegant classroom demonstrations for showing how air pressure and oxygen consumption interact to create visible movement of liquids. At first glance, the experiment looks like a simple trick: a candle burns in a shallow dish of colored water, a glass jar is placed over it, and as the flame goes out, the water rises into the glass seemingly as if the flame has “pulled” the water upward. For a child observing it for the first time, the effect feels magical. For a scientist, it becomes a window into the invisible behaviors of gases, pressure differences, and combustion. Every part of the setup — the flame, the air trapped inside the container, the pressure surrounding the dish, and the water climbing upward — plays a meaningful role. To fully understand what is happening, one must break the process down into its physical and chemical events and observe the way they unfold in sequence.
The experiment begins with an ordinary open dish filled with a thin layer of water, often colored with dye for better visibility. A small candle is placed upright in the water and lit. As soon as the burning candle is covered with a transparent upside-down glass or jar, the flame continues to burn, but the environment around the flame becomes isolated from the outside atmosphere. Initially, nothing dramatic happens. The flame burns brightly for several seconds, illuminating the inside of the jar, evaporating tiny droplets of water, and heating the air trapped inside. The candle consumes oxygen and produces carbon dioxide and water vapor in the process of combustion, but at first the flame has sufficient oxygen supply to remain stable. During this early stage, the flame actually warms the gases inside the jar. Heating causes air to expand, so the pressure inside the jar becomes slightly higher than the pressure outside. For a moment, this pressure difference pushes some of the air out from under the rim of the jar, usually forming a stream of bubbles through the water at the base. This bubbling phase is subtle but crucial because it later determines the amount of air available for contraction.
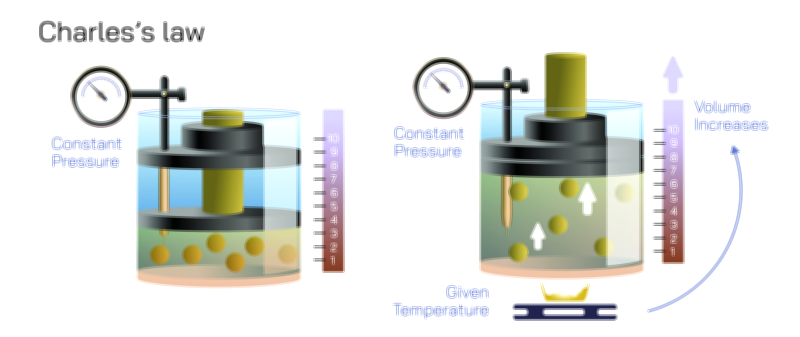
Once some of the heated air escapes and the flame continues to burn, the supply of oxygen enclosed in the jar begins to drop. The flame relies on oxygen to sustain combustion, and when the concentration becomes too low to support the chemical reaction, the flame dims and finally extinguishes. This is the key transition of the experiment. The moment the flame goes out, the heat source inside the jar suddenly disappears. Without continuous burning, the air inside begins to cool quickly. Cooling reduces the kinetic energy of the gas molecules and causes them to occupy a smaller effective volume. This contraction creates an internal pressure lower than the atmospheric pressure on the outside. The surrounding air cannot reenter the jar because the rim is sealed against the layer of water in the dish, so the external atmosphere pushes on the water instead. To equalize the pressure difference, water rises into the jar until the pressure inside and outside the jar reach equilibrium again.
The result looks simple — water climbs upward into the container — but it represents two interwoven physical processes: the cooling and contraction of trapped heated air and the partial removal of oxygen through combustion. Many people assume the water rises solely because oxygen was “used up” and therefore the internal air volume shrank. But the reality is more subtle. Oxygen consumption alone cannot account for all the observed water movement because oxygen makes up only about 21% of air. Even if oxygen disappeared completely, the internal reduction in volume would not fully match the height to which the water rises. What actually drives the majority of the water movement is the cooling of the air immediately after the flame extinguishes. Hot air expands; cold air contracts. Because the candle heated the air while burning and that heated air partly escaped as bubbles, the remaining gas cools dramatically after extinction and creates a partial vacuum. Atmospheric pressure outside the jar pushes the water upward to fill the reduced internal space.
The experiment becomes even more interesting when variations are introduced. For example, if a very tall glass is used, the burning time increases, allowing more hot air to escape as bubbles and producing a larger pressure difference when cooling occurs. This leads to a greater rise in water level. If multiple candles are used under the same jar, the water rises even higher because more heat warms the trapped air and more air escapes before oxygen depletion extinguishes the flames. Placing the jar prematurely over a candle that has not had time to warm the air will produce a much smaller rise because the pressure difference is not strong enough. The experiment therefore becomes a practical demonstration of the relationship between temperature and pressure in enclosed gases.
Water vapor also plays a meaningful role. During combustion, a candle produces water vapor, which expands when hot and condenses into liquid droplets as the jar cools. Condensation removes gaseous molecules from the air inside, further reducing gas pressure and contributing to water rise. This subtle effect is often overlooked, but it showcases another scientific principle: when a gas condenses into a liquid, it occupies far less space, making the remaining gas pressure drop even further.
Although designed as a child-friendly experiment, this activity embodies several fundamental principles found in physics and chemistry. It demonstrates that air has weight and pressure; that heat causes gas expansion and cooling causes contraction; that oxygen is essential for combustion; and that atmospheric pressure causes water movement rather than suction or vacuum pulling. This same concept appears in real-world systems such as barometers, vacuum sealing, fuel combustion engines, and fire suppression. Every situation where heated or cooled gases change pressure reflects the same principles seen in the rising water experiment.
The experiment can also help learners understand oxygen consumption more intuitively. People tend to think that oxygen is abundant and constant, but this demonstration reveals how quickly a flame depletes oxygen in an enclosed space. The flame brightens while oxygen is plentiful, weakens as oxygen becomes scarce, and extinguishes when the gas becomes insufficient to sustain combustion. This same pattern is critical in fire safety, atmospheric science, and human respiration, showing that oxygen availability controls combustion and metabolic processes alike.
Ultimately, the rising water experiment is memorable not because of its simplicity but because it translates hidden physical forces into something visible. It reveals that air pressure is not an abstract idea — it is a real force pressing on every surface around us. It shows that temperature changes can alter pressure dramatically even when nothing appears to be happening. It demonstrates that gases are active participants in chemical reactions rather than passive backgrounds. Most importantly, it teaches that science can be observed with ordinary materials: a candle, a dish of water, and an inverted glass become a powerful model of atmospheric physics. When the water climbs, it is a reminder that the natural world follows precise principles. Even an activity that fits on a small tabletop is governed by the same laws that control weather systems, engines, and biological respiration. Through the rising water experiment, learners witness invisible forces made visible — a vivid lesson in the physics of gases, air pressure, heat, and oxygen consumption.